Advanced Soluble Electron-Transporting Red Light Materials for Commercial OLED Production
Advanced Soluble Electron-Transporting Red Light Materials for Commercial OLED Production
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only offer superior photophysical properties but also enable cost-effective manufacturing processes. Patent CN101280187A introduces a groundbreaking class of soluble electron-transporting electroluminescent materials designed specifically to address these dual challenges. By utilizing a 4,7-bis(4-substituted thiophene)-2,1,3-benzothiadiazole core connected to rigid end groups, this technology achieves a delicate balance between solubility for solution processing and the amorphous stability required for high-performance devices. This innovation represents a significant leap forward for manufacturers seeking to transition from expensive vacuum deposition to scalable solution-based fabrication methods without compromising on efficiency or color purity.
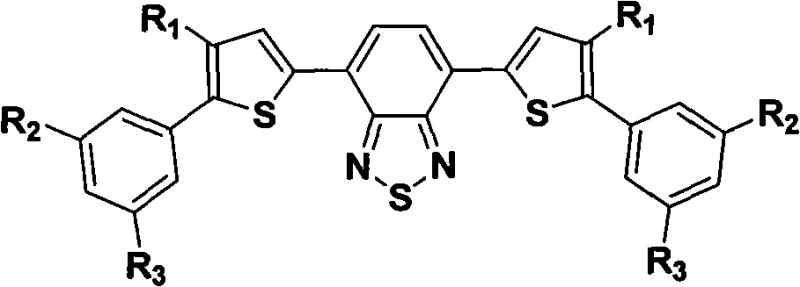
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of high-efficiency small molecule OLEDs has relied heavily on vacuum thermal evaporation (VTE), a process that is inherently capital-intensive and limited in scalability. Conventional red-emitting materials often suffer from poor solubility, necessitating complex vacuum equipment and resulting in low material utilization rates. Furthermore, standard red emitters frequently exhibit concentration quenching and lack balanced charge transport properties, forcing device engineers to incorporate additional functional layers, such as hole-blocking or electron-transport layers, to maintain efficiency. These extra layers increase the complexity of the device architecture, raise production costs, and introduce more interfaces where failure can occur, ultimately impacting the long-term stability and yield of the final display panels.
The Novel Approach
The novel approach detailed in this patent overcomes these hurdles by engineering a Donor-Acceptor-Donor (D-A-D) molecular structure that intrinsically possesses electron-transporting capabilities. By strategically introducing solubilizing alkyl or alkoxy groups at the 4-position of the thiophene rings and attaching rigid aromatic end groups, the material achieves excellent solubility in common organic solvents like toluene and chloroform. This solubility enables the use of low-cost solution processing techniques such as spin-coating or inkjet printing. Moreover, the inherent electron mobility of the benzothiadiazole core simplifies the device structure by potentially eliminating the need for separate electron transport layers, thereby reducing the total number of processing steps and enhancing the overall manufacturability of red OLED components for large-area displays.
Mechanistic Insights into Palladium-Catalyzed Suzuki Coupling
The synthesis of these advanced materials relies on the robust and versatile palladium-catalyzed Suzuki-Miyaura cross-coupling reaction, which allows for the precise construction of the conjugated carbon skeleton under relatively mild conditions. The mechanism involves the oxidative addition of a palladium(0) catalyst to an aryl halide, followed by transmetallation with an organoboron species and subsequent reductive elimination to form the new carbon-carbon bond. In the context of this patent, the reaction is typically conducted at temperatures ranging from 70°C to 110°C under an inert nitrogen atmosphere to prevent catalyst deactivation. The use of tetrakis(triphenylphosphine)palladium as the catalyst ensures high turnover numbers, facilitating the coupling of sterically hindered rigid end groups with the thiophene-benzothiadiazole core while maintaining high regioselectivity.
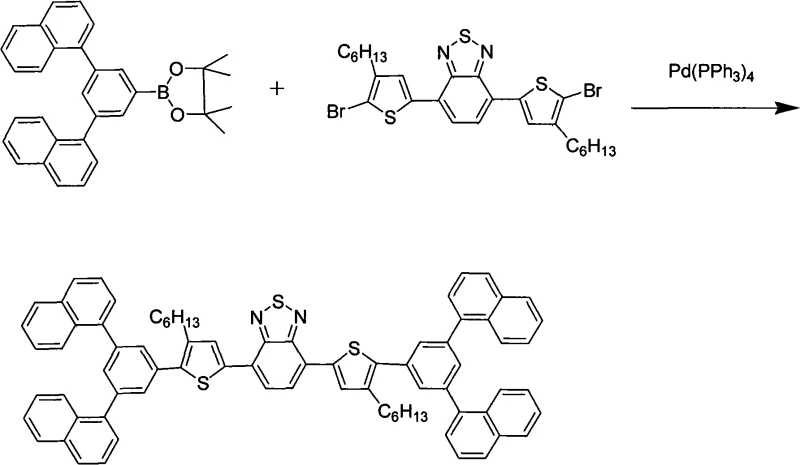
Impurity control is paramount in electronic grade chemicals, and this synthetic route offers distinct advantages in managing byproduct profiles. The stepwise nature of the synthesis, involving the isolation of intermediate boronic esters, allows for rigorous purification via silica gel column chromatography and recrystallization before the final coupling step. This modular approach prevents the accumulation of homocoupling byproducts or unreacted starting materials in the final product. Additionally, the crystallinity and solubility differences between the target molecule and potential impurities are exploited during the workup phase, where specific solvent systems like petroleum ether and dichloromethane are used to selectively precipitate the high-purity red solid, ensuring the material meets the stringent purity specifications required for commercial electroluminescent applications.
How to Synthesize Soluble Electroluminescent Material Efficiently
The synthesis protocol outlined in the patent provides a reproducible pathway for generating these high-value electronic chemicals, emphasizing the importance of stoichiometric control and atmospheric protection. The process begins with the preparation of rigid end-group precursors, followed by their coupling to substituted thiophenes, and concludes with the central assembly around the benzothiadiazole unit. Each step requires careful monitoring of reaction times, which can range from 8 to 24 hours depending on the steric bulk of the substituents. For a detailed breakdown of the specific reagents, molar ratios, and workup procedures required to achieve optimal yields and purity, please refer to the standardized synthesis guide below.
- Perform initial Suzuki coupling between rigid end groups and substituted thiophene bromides to establish the molecular periphery.
- Execute lithiation followed by borylation at the thiophene 5-position to generate the reactive boronic ester intermediate.
- Conduct the final Suzuki coupling with 4,7-dibromo-2,1,3-benzothiadiazole to form the complete conjugated system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this soluble electron-transporting material translates into tangible operational efficiencies and risk mitigation strategies. The ability to purify these materials via solution methods rather than sublimation significantly lowers the barrier to entry for scaling production, as it utilizes standard chemical processing equipment rather than specialized vacuum hardware. This shift reduces the dependency on complex physical vapor deposition infrastructure, allowing for a more flexible and responsive supply chain capable of adapting to fluctuating market demands for OLED components. Furthermore, the simplified device architecture enabled by the material's electron-transport properties reduces the number of distinct raw materials required per device, streamlining inventory management and reducing the complexity of the Bill of Materials (BOM).
- Cost Reduction in Manufacturing: The transition to solution-processable materials fundamentally alters the cost structure of OLED manufacturing by enabling high-throughput coating techniques. Eliminating the need for high-vacuum evaporation chambers drastically reduces energy consumption and capital expenditure associated with facility setup. Additionally, the removal of auxiliary layers like hole-blocking layers from the device stack reduces material consumption per unit area. These factors combine to drive down the overall cost of goods sold (COGS) for red emissive layers, making high-quality full-color displays more economically viable for mass-market applications without sacrificing performance metrics.
- Enhanced Supply Chain Reliability: The synthetic route utilizes widely available commodity chemicals such as thiophenes, benzothiadiazoles, and aryl boronic acids, which mitigates the risk of supply bottlenecks associated with exotic or proprietary precursors. The robustness of the Suzuki coupling chemistry ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term supply contracts with display manufacturers. By establishing a synthesis pathway that relies on stable intermediates and standard catalytic systems, suppliers can guarantee continuous availability of high-purity materials, thereby reducing lead times and preventing production stoppages for downstream clients.
- Scalability and Environmental Compliance: Scaling this chemistry from laboratory grams to metric tons is facilitated by the use of homogeneous catalysis in common organic solvents, which is well-understood in the fine chemical industry. The process avoids the use of highly toxic heavy metals other than the trace palladium catalyst, which can be effectively scavenged and recycled, aligning with increasingly strict environmental regulations. The ability to purify the final product through crystallization rather than energy-intensive sublimation further reduces the carbon footprint of the manufacturing process, supporting corporate sustainability goals while ensuring the material is ready for commercial scale-up of complex organic semiconductors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this soluble electroluminescent technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical characteristics of the material for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this material into existing production lines or new product development pipelines.
Q: Why is solubility critical for this electroluminescent material?
A: Solubility allows for solution-based processing techniques like spin-coating, which significantly reduces manufacturing costs compared to traditional vacuum thermal evaporation methods required for insoluble small molecules.
Q: How does this material simplify OLED device architecture?
A: The material possesses intrinsic electron-transporting properties due to its benzothiadiazole core, eliminating the need for a separate electron transport or hole-blocking layer in the device stack.
Q: What is the primary synthetic strategy used?
A: The synthesis relies heavily on palladium-catalyzed Suzuki-Miyaura cross-coupling reactions, utilizing boronic esters and aryl halides to build the conjugated backbone efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable OLED Material Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of soluble electron-transporting materials in the next generation of display technologies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to mass manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for electronic grade chemicals, including trace metal analysis and particle count verification. We are committed to delivering high-purity OLED material that consistently performs in demanding electroluminescent applications.
We invite you to collaborate with our technical team to explore how this patented chemistry can optimize your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your strategic goals for cost reduction in electronic chemical manufacturing and accelerating your time to market.
