Optimizing Cinacalcet Production: A Technical Analysis of Novel Intermediate Synthesis and Commercial Scalability
Introduction to Advanced Cinacalcet Intermediate Technology
The global demand for effective treatments for Secondary Hyperparathyroidism (SHPT) has placed significant pressure on the supply chain of key active pharmaceutical ingredients (APIs), particularly Cinacalcet. Patent CN103664577A introduces a groundbreaking preparation method for a critical Cinacalcet intermediate, specifically (Z)-3-chloro-3-[3-(trifluoromethyl)phenyl]-2-acrylic acid. This technological advancement addresses long-standing inefficiencies in traditional synthesis routes by utilizing 3-(trifluoromethyl)acetophenone as a robust starting material. The structural integrity of the final API, N-[(1R)-1-(1-naphthyl)ethyl]-3-[3-(trifluoromethyl)phenyl]propyl-1-amine, relies heavily on the stereochemical purity and yield of its precursors. By optimizing the condensation, chlorination, and oxidation steps, this patent offers a pathway that is not only chemically elegant but also industrially viable.
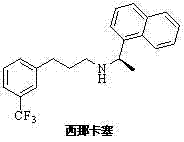
For R&D directors and procurement specialists, understanding the nuances of this synthesis is crucial for securing a reliable pharmaceutical intermediates supplier. The patent details a method that circumvents the use of hazardous reagents found in earlier iterations, such as titanium tetraisopropylate and sodium cyanoborohydride. Instead, it leverages a sequence involving N-substituted acetals and selective catalytic hydrogenation. This shift represents a paradigm change in how we approach the commercial scale-up of complex pharmaceutical intermediates, ensuring that the final product meets stringent purity specifications required by regulatory bodies worldwide while maintaining economic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cinacalcet has been plagued by significant technical and safety hurdles that impact both cost and supply continuity. Early methodologies, such as those disclosed in US6211244, relied on the condensation of 1-acetonaphthone with 3-(trifluoromethyl)phenethylamine using titanium tetraisopropylate. This approach is fundamentally flawed for large-scale manufacturing due to the high cost of the titanium catalyst and the generation of substantial metal-containing waste. Furthermore, the subsequent reduction step utilizing sodium cyanoborohydride introduces severe toxicity concerns, necessitating specialized handling protocols and expensive waste treatment infrastructure.
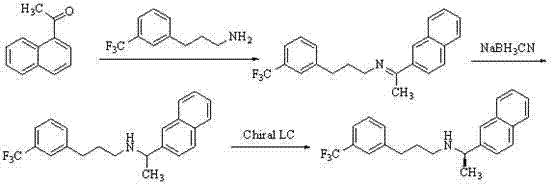
Alternative routes, such as the one described in US7250533, attempted to utilize Heck coupling reactions with ethyl acrylate. However, ethyl acrylate is a known carcinogen and is highly unstable, posing unacceptable risks for industrial environments. Additionally, these conventional pathways often suffer from low selectivity, leading to difficult purification processes and reduced overall yields. The presence of side products like polymers or deamination byproducts, as seen in CN102307845, further complicates the isolation of the target molecule. These factors collectively drive up the cost of goods sold (COGS) and create bottlenecks in the supply chain reliability for high-purity Cinacalcet.
The Novel Approach
The methodology presented in CN103664577A offers a transformative solution by establishing a new synthetic lineage centered around the (Z)-3-chloro-acrylic acid intermediate. This novel approach begins with the condensation of 3-(trifluoromethyl)acetophenone with an N-substituted acetal, followed by chlorination and oxidation to generate the key unsaturated acid derivative. This route eliminates the need for expensive transition metal catalysts in the initial stages and avoids the use of highly toxic reducing agents until the final, controlled amidation reduction. The strategic introduction of the chlorine atom allows for a unique dual-function reduction step later in the process.
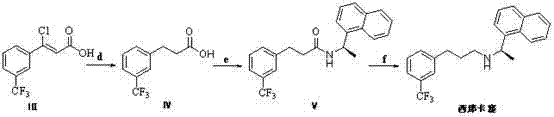
By employing catalytic hydrogenation under optimized conditions (2-10 kg/cm² pressure, 10-30°C), the process achieves simultaneous reduction of the carbon-carbon double bond and hydrodechlorination. This single-step transformation converts the chloro-intermediate directly into the saturated propionic acid derivative with high efficiency. Subsequent amidation with (1R)-1-(1-naphthyl)ethylamine and final amide reduction yield the target API. This streamlined sequence drastically simplifies the operational workflow, reduces the number of unit operations, and enhances the overall atom economy. For manufacturers, this translates to a more robust process capable of delivering high-purity pharmaceutical intermediates with consistent quality batch after batch.
Mechanistic Insights into Selective Catalytic Hydrogenation and Amidation
The core innovation of this patent lies in the precise control of the catalytic hydrogenation step, which serves as the pivot point for the entire synthesis. In the conversion of Formula III (the chloro-acrylic acid) to Formula IV (the propionic acid), the reaction mechanism involves the adsorption of the substrate onto the surface of a heterogeneous catalyst, such as palladium on carbon (Pd/C) or nickel. Under mild hydrogen pressure, the catalyst facilitates the addition of hydrogen across the alkene double bond while simultaneously cleaving the carbon-chlorine bond. This dual reactivity is highly sensitive to reaction parameters; the patent specifies that maintaining the temperature between 10-30°C and pressure between 2-10 kg/cm² is critical. Deviations from these windows can lead to over-reduction or incomplete dechlorination, generating impurities that are difficult to separate.
Following the formation of the propionic acid, the amidation step utilizes activated acid halides (generated in situ using thionyl chloride or oxalyl chloride) to react with the chiral amine. The use of bases like triethylamine or pyridine scavenges the generated acid, driving the equilibrium towards the amide product (Formula V). The final reduction of the amide to the amine is achieved using borane complexes or sodium borohydride-iodine systems. This specific choice of reductant is mechanistically superior to lithium aluminum hydride in terms of safety and selectivity, as it minimizes the risk of reducing other sensitive functional groups that might be present. The rigorous control over these mechanistic steps ensures that the impurity profile remains within tight limits, a key requirement for reducing lead time for high-purity pharmaceutical intermediates during regulatory filings.
How to Synthesize (Z)-3-chloro-3-[3-(trifluoromethyl)phenyl]-2-acrylic acid Efficiently
The synthesis of the key intermediate requires careful attention to reaction stoichiometry and solvent selection to maximize yield and minimize byproduct formation. The process initiates with the condensation of the ketone starting material in polar aprotic solvents like DMF or dioxane at elevated temperatures to ensure complete conversion to the enaminone. Subsequent chlorination must be managed to prevent over-chlorination, followed by a delicate oxidation step using systems like Sodium phosphate dibasic monohydrate/Textone. The detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are critical for process engineers.
- Condense 3-(trifluoromethyl)acetophenone with an N-substituted acetal to form an enaminone intermediate.
- React the enaminone with a chlorinating agent like phosphorus oxychloride to introduce the chlorine atom.
- Oxidize the resulting aldehyde using an oxidant system to obtain the key (Z)-3-chloro-acrylic acid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic advantages beyond mere technical feasibility. The primary benefit is the substantial reduction in raw material costs and hazard management expenses. By eliminating the need for titanium tetraisopropylate and sodium cyanoborohydride, manufacturers can avoid the premium pricing associated with these specialty reagents and the costly disposal fees for heavy metal and cyanide waste. This shift directly contributes to cost reduction in pharmaceutical intermediates manufacturing, allowing for more competitive pricing structures in the final API market without compromising on quality standards.
- Cost Reduction in Manufacturing: The new route utilizes commodity chemicals such as 3-(trifluoromethyl)acetophenone and common solvents like toluene and methanol, which are readily available in the global market. The replacement of expensive catalysts with reusable heterogeneous catalysts like Pd/C further drives down operational expenditures. Additionally, the simplified purification steps reduce solvent consumption and energy usage, leading to a leaner manufacturing process that significantly lowers the total cost of ownership for the production line.
- Enhanced Supply Chain Reliability: Dependence on hazardous or scarce reagents creates vulnerability in the supply chain. By switching to a route based on stable, non-toxic intermediates, suppliers can guarantee continuous production even during market fluctuations. The robustness of the catalytic hydrogenation step ensures high throughput and consistent batch cycles, minimizing downtime. This reliability is essential for maintaining the inventory levels required by major pharmaceutical clients who demand just-in-time delivery of critical API intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations suitable for multi-ton production. The avoidance of carcinogenic ethyl acrylate and toxic cyanides simplifies environmental permitting and reduces the regulatory burden on manufacturing sites. This alignment with green chemistry principles not only mitigates legal risks but also enhances the corporate sustainability profile, a growing priority for stakeholders in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the process capabilities.
Q: What are the safety advantages of this new Cinacalcet synthesis route compared to prior art?
A: Unlike previous methods utilizing toxic sodium cyanoborohydride or carcinogenic ethyl acrylate, this patent employs safer catalytic hydrogenation and standard chlorination reagents, significantly reducing occupational health risks and waste disposal complexity.
Q: How does the selective hydrogenation step improve product purity?
A: The process utilizes controlled catalytic hydrogenation (e.g., Pd/C) at mild pressures (2-10 kg/cm²) and temperatures (10-30°C) to simultaneously reduce the double bond and remove the chlorine atom, minimizing side reactions and simplifying downstream purification.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the route avoids expensive catalysts like titanium tetraisopropylate and uses readily available starting materials like 3-(trifluoromethyl)acetophenone, making it highly cost-effective and scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinacalcet Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex therapeutics like Cinacalcet depends on a partnership grounded in technical excellence and supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot plant to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards, providing you with the confidence needed to navigate the regulatory landscape.
We invite you to collaborate with us to leverage this advanced synthetic route for your Cinacalcet supply needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and drive value for your organization.
