Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Scalable Chiral Amine Production
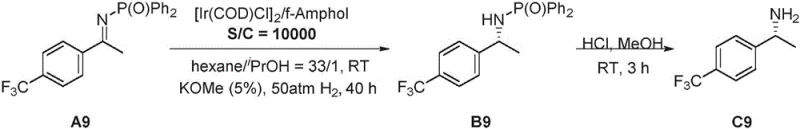
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral centers, particularly for active pharmaceutical ingredients (APIs) where stereochemical purity is non-negotiable. Patent CN112142784B introduces a groundbreaking asymmetric reduction method for nitrogen-phosphono-protected imines, addressing critical challenges in chiral amine synthesis. This technology utilizes a sophisticated metal catalyst system derived from an iridium complex and a specialized nitrogen-phosphorus chiral ligand known as f-Amphol. The process operates under a hydrogen atmosphere to convert prochiral imines into high-value chiral amines with exceptional efficiency. What sets this invention apart is its ability to achieve yields exceeding 90% and enantiomeric excess (ee) values greater than 99% for a vast array of substrates. Furthermore, the catalyst demonstrates remarkable turnover numbers (TON) reaching up to 10,000, signifying a highly economical use of precious metals. This capability is not merely theoretical; the patent explicitly validates the method through the successful synthesis of Cinacalcet, a vital drug for treating secondary hyperparathyroidism in chronic kidney disease patients, proving its direct applicability in high-stakes medicinal chemistry.
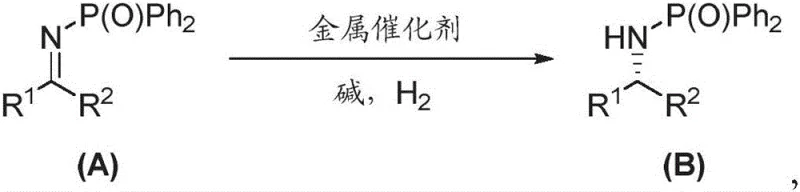
Traditional methods for synthesizing chiral amines often rely on resolution techniques or less efficient catalytic systems that struggle with substrate generality and stereocontrol. Conventional approaches frequently necessitate harsh reaction conditions, expensive stoichiometric chiral auxiliaries, or multiple purification steps that erode overall yield and increase waste generation. Many existing catalytic hydrogenation protocols fail to maintain high enantioselectivity when faced with sterically hindered or electronically diverse imine substrates, leading to inconsistent batch quality and increased production costs. In contrast, the novel approach detailed in this patent leverages the unique electronic and steric properties of the nitrogen-phosphono protecting group. This specific protection strategy stabilizes the imine functionality while facilitating precise coordination with the iridium catalyst. By employing the f-Amphol ligand, the system creates a highly defined chiral environment that directs hydrogen addition with superior fidelity. This results in a streamlined process that eliminates the need for extensive downstream purification to remove unwanted enantiomers, thereby simplifying the manufacturing workflow and enhancing the overall sustainability of the production line for fine chemical intermediates.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the intricate interplay between the metal precursor and the chiral ligand. The catalyst is generated in situ by reacting the iridium dimer [Ir(COD)Cl]2 with the f-Amphol ligand, which features a ferrocene backbone and specific bulky substituents like 3,5-di-tert-butylphenyl groups on the phosphorus atom. This structural architecture, as illustrated in the ligand diagram, is crucial for inducing asymmetry. The nitrogen-phosphono group on the substrate acts not just as a protector but as a directing group that coordinates with the metal center, locking the imine into a specific orientation relative to the chiral pocket of the catalyst. Upon exposure to hydrogen gas at pressures ranging from 40 atm to 50 atm, the activated catalyst facilitates the transfer of hydride and proton to the carbon-nitrogen double bond. The rigidity of the ligand framework ensures that this addition occurs exclusively from one face of the planar imine, resulting in the observed >99% ee. This level of control is essential for meeting the stringent purity specifications required by global regulatory bodies for pharmaceutical intermediates.
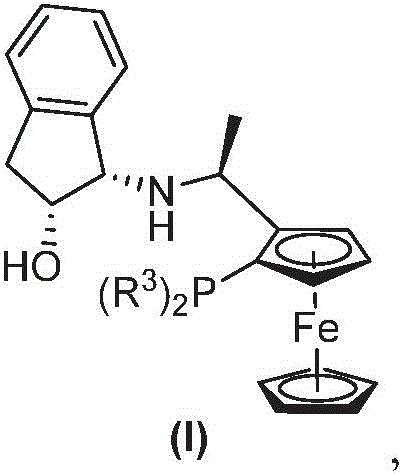
Impurity control is another critical aspect where this mechanism excels. In chiral synthesis, the formation of the wrong enantiomer or over-reduced byproducts can be detrimental to the safety profile of the final drug. The high selectivity of this iridium-f-Amphol system minimizes the formation of such impurities at the source. The phosphono group remains stable under the reduction conditions but can be easily removed in a subsequent step using mild acidic hydrolysis, as demonstrated in the patent examples. This orthogonal stability ensures that the chiral integrity established during the hydrogenation step is preserved throughout the workup process. For R&D directors, this means a cleaner reaction profile that reduces the burden on analytical teams and lowers the risk of batch rejection due to out-of-specification chiral purity. The ability to tune the reaction by adjusting parameters like base concentration (e.g., using KOMe) and solvent polarity further allows for optimization against specific impurity profiles, ensuring a robust and reliable synthetic route for complex molecules.
How to Synthesize Chiral Amines Efficiently
Implementing this asymmetric reduction protocol requires careful attention to catalyst preparation and reaction conditions to maximize the benefits of the high TON capability. The process begins with the activation of the iridium precursor in a suitable solvent, followed by the introduction of the substrate and base under an inert atmosphere before pressurizing with hydrogen. The patent provides detailed embodiments showing that the reaction can proceed effectively at room temperature, which significantly reduces energy consumption compared to thermal methods. The versatility of the system allows for the use of various solvents such as hexane, alcohols, or chlorinated hydrocarbons, offering flexibility for process engineers to select options that align with safety and environmental guidelines. For those looking to adopt this technology for the commercial scale-up of complex pharmaceutical intermediates, understanding the precise stoichiometry and mixing parameters is vital to replicate the high yields and selectivity reported in the laboratory examples.
- Prepare the metal catalyst by reacting [Ir(COD)Cl]2 with the f-Amphol ligand in a first solvent like isopropanol at room temperature for 1 to 4 hours.
- Combine the catalyst solution with the nitrogen-phosphono protected imine substrate, a base such as potassium methoxide, and a second solvent like n-hexane.
- Conduct the asymmetric reduction under a hydrogen atmosphere of 40 to 50 atm at room temperature for 1 to 12 hours, followed by purification to isolate the chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic reduction of catalyst loading requirements due to the high turnover numbers achieved. Since the catalyst can facilitate up to 10,000 cycles, the amount of expensive iridium and chiral ligand needed per kilogram of product is minimized, leading to significant cost reduction in pharmaceutical intermediates manufacturing. This efficiency translates directly into lower raw material costs and reduced dependency on precious metal supply chains, which are often volatile. Furthermore, the mild reaction conditions eliminate the need for specialized high-temperature equipment, allowing production to occur in standard stainless steel reactors commonly found in multipurpose facilities. This compatibility enhances supply chain reliability by enabling flexible manufacturing across different sites without requiring capital-intensive retrofitting.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the exceptional catalytic efficiency and the elimination of costly resolution steps. By achieving high enantioselectivity directly, the process avoids the 50% yield loss typically associated with classical resolution techniques, effectively doubling the output from the same amount of starting material. Additionally, the ability to remove the phosphono protecting group under mild conditions simplifies the workup procedure, reducing solvent usage and waste disposal costs. The qualitative improvement in process mass intensity (PMI) makes this route highly attractive for cost-sensitive generic drug production where margin optimization is critical for market competitiveness.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions and the broad substrate scope. The method tolerates a wide range of functional groups, meaning that variations in starting material quality or slight changes in substrate structure do not necessitate a complete re-optimization of the process. This flexibility ensures consistent delivery schedules even when sourcing raw materials from different suppliers. Moreover, the use of common solvents and bases reduces the risk of supply bottlenecks associated with exotic reagents. For supply chain planners, this translates to a more resilient production schedule with reduced lead time for high-purity pharmaceutical intermediates, ensuring that downstream API synthesis is never delayed by intermediate shortages.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety and environmental challenges, but this method mitigates many of those risks. The reaction operates at room temperature and moderate hydrogen pressures, which are well within the safety limits of standard industrial hydrogenation units. The high atom economy and reduced waste generation align with green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. The successful demonstration of gram-scale synthesis in the patent indicates a clear path to ton-scale production. This scalability ensures that as demand for the final drug product grows, the manufacturing capacity can be expanded seamlessly without compromising on quality or safety standards, supporting long-term business growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reduction technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing solutions. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios. The answers reflect the practical realities of running high-efficiency catalytic reactions in a regulated environment.
Q: What represents the key innovation in this asymmetric reduction method?
A: The key innovation lies in the use of nitrogen-phosphono protected imines combined with a specific iridium complex and f-Amphol chiral ligand, achieving exceptionally high enantioselectivity (>99% ee) and turnover numbers up to 10,000 under mild conditions.
Q: Can this method be scaled for industrial pharmaceutical production?
A: Yes, the patent demonstrates successful gram-level synthesis and high TON experiments, indicating robust scalability. The mild reaction conditions (room temperature) and high catalyst efficiency make it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What types of substrates are compatible with this catalytic system?
A: The method exhibits broad substrate scope, successfully reducing imines with various aryl, heteroaryl, and alkyl substituents, including those with electron-withdrawing groups like trifluoromethyl and halogens, maintaining high yields and stereoselectivity across diverse structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinacalcet Supplier
The potential of this asymmetric reduction technology to transform the production of chiral amines is immense, particularly for high-value drugs like Cinacalcet. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are successfully translated into industrial reality. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee >99% ee as required by this patent. We understand the critical nature of supply consistency in the pharmaceutical sector and have established robust protocols to maintain quality across large batches. Our team is ready to leverage this advanced iridium-catalyzed methodology to deliver cost-effective and high-quality intermediates that meet your exacting standards.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Whether you are in the early stages of process development or looking to secure a long-term supply partner for commercial manufacturing, NINGBO INNO PHARMCHEM is committed to providing the technical expertise and production capacity necessary to support your success in the global market.
