Advanced Electrochemical Synthesis of FCAA: Scaling High-Purity Vitamin A Intermediates
Advanced Electrochemical Synthesis of FCAA: Scaling High-Purity Vitamin A Intermediates
The global demand for high-purity vitamin A derivatives and carotenoids necessitates robust, sustainable supply chains for critical intermediates like 2-methyl-4-acetoxy-2-butenoic acid (FCAA). Recent advancements in green chemistry have highlighted the potential of electrochemical methods to replace traditional, hazardous oxidation processes. Specifically, patent CN116005179A introduces a groundbreaking methodology for the electrochemical in-situ oxidation of 2-methyl-4-acetoxy-2-butenal (FCA) to FCAA. This technology leverages a specialized electrode system to generate reactive oxygen species (ROS) directly within the reaction medium, bypassing the need for external chemical oxidants. For R&D directors and procurement specialists in the fine chemical sector, this represents a significant shift towards safer, more controllable, and economically viable manufacturing protocols. By utilizing a modified graphite felt gas diffusion electrode as the cathode and an inert anode separated by a cation exchange membrane, the process achieves exceptional conversion rates and selectivity. This report analyzes the technical merits of this innovation, providing a strategic overview for stakeholders seeking a reliable pharmaceutical intermediate supplier capable of delivering complex molecules with stringent purity specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of FCA to FCAA has relied heavily on conventional chemical oxidation routes involving strong oxidizing agents such as hydrogen peroxide (H2O2) or sodium chlorite (NaClO2). While these methods can achieve reasonable conversion, they are plagued by significant operational drawbacks that impact both safety and cost efficiency. The use of halogen-containing oxidants introduces severe corrosion risks to reactor equipment, necessitating expensive materials of construction and frequent maintenance. Furthermore, these reactions often require rigorous temperature control, typically needing cryogenic conditions to prevent runaway exotherms, which drastically increases energy consumption. Post-reaction processing is equally burdensome; excess oxidants must be quenched with reducing agents, generating substantial volumes of saline wastewater that require complex treatment before disposal. The presence of residual halogens and metal ions often complicates the purification of the final API intermediate, potentially affecting the impurity profile of downstream vitamin A derivatives. These factors collectively create a bottleneck in the supply chain, increasing lead times and reducing the overall sustainability of the manufacturing process.
The Novel Approach
In stark contrast, the electrochemical method described in patent CN116005179A offers a paradigm shift by utilizing electricity as the primary reagent. This approach eliminates the need for hazardous chemical oxidants entirely, replacing them with electrons and oxygen. The core of this innovation lies in the use of a modified graphite felt carbon material gas diffusion electrode as the cathode. This specialized electrode facilitates the oxygen reduction reaction (ORR), generating active oxygen species in situ that selectively oxidize the aldehyde group of FCA to the carboxylic acid. 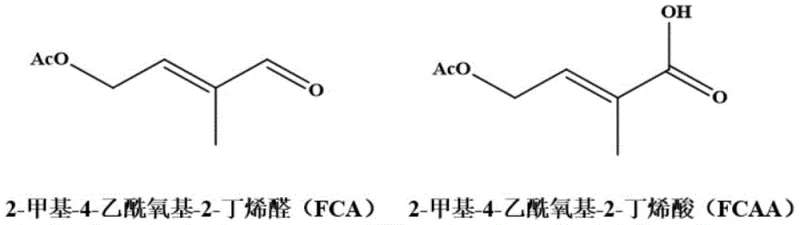 As illustrated in the structural transformation, the process preserves the sensitive acetoxyl and alkene functionalities while targeting the aldehyde moiety with high precision. The implementation of a cation exchange membrane physically isolates the anode compartment, preventing the substrate from undergoing direct anodic oxidation, which is a common cause of over-oxidation and byproduct formation in direct electrolysis. This indirect oxidation mechanism ensures that the reaction proceeds under mild conditions, typically between 20°C and 40°C, significantly reducing thermal stress on the equipment and the molecule. The result is a streamlined workflow that minimizes waste generation and simplifies the isolation of the final product, aligning perfectly with the principles of green chemistry and modern industrial safety standards.
As illustrated in the structural transformation, the process preserves the sensitive acetoxyl and alkene functionalities while targeting the aldehyde moiety with high precision. The implementation of a cation exchange membrane physically isolates the anode compartment, preventing the substrate from undergoing direct anodic oxidation, which is a common cause of over-oxidation and byproduct formation in direct electrolysis. This indirect oxidation mechanism ensures that the reaction proceeds under mild conditions, typically between 20°C and 40°C, significantly reducing thermal stress on the equipment and the molecule. The result is a streamlined workflow that minimizes waste generation and simplifies the isolation of the final product, aligning perfectly with the principles of green chemistry and modern industrial safety standards.
Mechanistic Insights into Electrochemical In-Situ Oxidation
The efficacy of this electrochemical system is rooted in the precise engineering of the electrode interface and the controlled generation of reactive intermediates. The cathode, a modified graphite felt, undergoes a rigorous pretreatment process involving acid boiling followed by cyclic voltammetry scanning. This dual-modification strategy serves two critical functions: firstly, the acid treatment grafts oxygen-containing functional groups onto the carbon fiber surface, enhancing its hydrophilicity and affinity for the aqueous electrolyte. Secondly, the electrochemical etching creates a micro-roughened surface topology, drastically increasing the effective electroactive surface area available for the oxygen reduction reaction. When oxygen is continuously supplied to the cathode region, it accepts electrons to form superoxide radicals (O2•−) and hydrogen peroxide (H2O2). These reactive oxygen species act as the actual oxidants, attacking the carbonyl carbon of the FCA substrate. Because these species are generated locally at the electrode surface, their concentration can be finely tuned by adjusting the cathode potential, typically maintained between -1.0V and -1.3V vs Ag/Ag+. This level of control is unattainable with bulk chemical oxidants, where concentration gradients often lead to inconsistent reaction rates.
Furthermore, the separation of the anodic and cathodic compartments via a perfluorosulfonic acid cation exchange membrane is pivotal for maintaining high selectivity. In a standard undivided cell, the organic substrate could migrate to the anode and undergo non-selective direct electron transfer, leading to polymerization or complete mineralization to CO2. By restricting the FCA to the catholyte, the system ensures that oxidation occurs exclusively via the mediated ROS pathway. The supporting electrolyte, such as sodium sulfate (Na2SO4) at concentrations of 300-400ppm, ensures sufficient conductivity without introducing interfering ions that could precipitate or react with the product. The use of a water-acetonitrile co-solvent system further optimizes the solubility of the organic FCA substrate while maintaining the ionic strength required for efficient charge transfer. This intricate balance of mass transport, electron transfer, and chemical kinetics results in a process that consistently delivers FCA conversion rates exceeding 97% with product selectivity greater than 99%, demonstrating a robustness that is essential for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Methyl-4-Acetoxy-2-Butenoic Acid Efficiently
Implementing this electrochemical protocol requires careful attention to electrode preparation and cell configuration to replicate the high yields reported in the patent literature. The process begins with the activation of the graphite felt cathode, a step that is critical for establishing the necessary catalytic activity for oxygen reduction. Following electrode modification, the electrolyte system is prepared by mixing deionized water with acetonitrile and dissolving the supporting electrolyte to ensure uniform conductivity throughout the cell. The FCA substrate is then introduced into the cathode compartment, where it is subjected to a constant potential under a continuous flow of oxygen gas. The detailed standardized synthesis steps, including specific parameters for current density, stirring rates, and reaction duration, are outlined below to assist technical teams in reproducing these results.
- Prepare the modified graphite felt cathode by acid boiling and cyclic voltammetry etching to enhance oxygen reduction activity.
- Configure the electrolyte system using a water-acetonitrile mixture with a supporting electrolyte like Na2SO4.
- Perform constant potential electrolysis at the cathode (-1.0 to -1.3V) under oxygen saturation to generate ROS and oxidize FCA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chemical to electrochemical oxidation offers tangible benefits that extend beyond mere technical performance. The most immediate impact is seen in the simplification of the supply chain for raw materials. By eliminating the need for hazardous oxidants like sodium chlorite and hydrogen peroxide, manufacturers reduce their dependency on volatile chemical markets and mitigate the risks associated with the storage and transport of dangerous goods. This shift not only enhances workplace safety but also significantly lowers the regulatory burden related to hazardous waste management. The absence of halogenated byproducts means that wastewater treatment becomes less complex and costly, contributing to substantial cost savings in environmental compliance. Moreover, the mild reaction conditions reduce the energy load associated with cooling systems, further driving down operational expenditures. These efficiencies translate into a more resilient supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive chemical oxidants and the subsequent quenching agents directly reduces the bill of materials for every batch produced. Traditional methods require stoichiometric amounts of oxidants and additional reagents for neutralization, all of which contribute to the final cost of goods. In this electrochemical process, electrons serve as the clean reagent, and the only consumable is electricity, which is generally cheaper and more stable in price than specialty chemicals. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting material to byproducts, maximizing the atom economy. The simplified downstream processing, which avoids complex extraction and washing steps required to remove inorganic salts and quenching residues, further reduces labor and solvent costs. Collectively, these factors drive a significant reduction in the overall manufacturing cost, making the final FCAA intermediate more competitive in the marketplace.
- Enhanced Supply Chain Reliability: Relying on a process that uses abundant and stable inputs like oxygen, water, and electricity inherently de-risks the supply chain. Chemical oxidants often face supply disruptions due to production issues or transportation regulations, which can halt manufacturing lines. In contrast, the inputs for this electrochemical method are universally available, ensuring continuous operation even during market fluctuations. The robustness of the modified electrode also contributes to reliability; once activated, the graphite felt maintains its activity over extended periods, reducing the frequency of electrode replacement and maintenance downtime. This stability allows for longer production campaigns and more predictable delivery schedules, which is critical for customers managing just-in-time inventory for vitamin A synthesis. The ability to scale this technology from pilot to commercial volumes without fundamental changes to the chemistry further secures the long-term supply continuity.
- Scalability and Environmental Compliance: The modular nature of electrochemical reactors facilitates straightforward scale-up, allowing manufacturers to increase capacity by adding more cells rather than building massive new vessels. This flexibility supports the commercial scale-up of complex pharmaceutical intermediates without the exponential increase in risk often seen with batch chemical reactors. From an environmental perspective, the process aligns with increasingly strict global regulations on emissions and waste. The absence of heavy metal catalysts and halogenated waste streams simplifies the permitting process for new facilities and reduces the liability associated with environmental incidents. The self-quenching nature of the reactive oxygen species means that the reaction mixture is inherently safer to handle post-reaction, reducing the risk of accidents during product isolation. This commitment to sustainability not only meets regulatory requirements but also enhances the brand value of the supplier in the eyes of environmentally conscious multinational clients.
Frequently Asked Questions (FAQ)
Understanding the nuances of this advanced synthesis method is crucial for technical teams evaluating its adoption. The following questions address common inquiries regarding the mechanism, scalability, and quality control aspects of the electrochemical oxidation of FCA. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industrial application. We encourage stakeholders to review these insights to fully appreciate the operational advantages offered by this technology.
Q: How does the electrochemical method improve selectivity compared to chemical oxidation?
A: The method uses a cation exchange membrane to isolate the anode, preventing direct over-oxidation of FCA. Reactive oxygen species generated at the cathode provide controlled, indirect oxidation, achieving selectivity greater than 99%.
Q: What are the key advantages of the modified graphite felt electrode?
A: Acid boiling and CV etching introduce oxygen-containing functional groups and increase surface roughness. This significantly enhances the effective surface area and affinity for dissolved oxygen, improving the efficiency of the oxygen reduction reaction.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process operates under mild conditions (20-40°C) without hazardous chemical oxidants. The elimination of complex quenching and extraction steps simplifies downstream processing, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-4-Acetoxy-2-Butenoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates like FCAA requires more than just a patented process; it demands a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity 2-methyl-4-acetoxy-2-butenoic acid that meets stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific requirements of electrochemical synthesis, including the management of specialized electrode systems and the optimization of electrolyte formulations for maximum efficiency.
We invite you to collaborate with us to leverage this cutting-edge technology for your vitamin A derivative supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can optimize your bottom line. Please contact us to request specific COA data and route feasibility assessments for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you secure a reliable source of high-quality intermediates while contributing to a more sustainable and efficient pharmaceutical manufacturing ecosystem.
