Advanced Electrochemical Synthesis of FCAA for High-Purity Vitamin A Intermediates
The global demand for high-purity carotenoid precursors and Vitamin A intermediates continues to drive innovation in synthetic methodology, particularly where traditional chemical routes face environmental and efficiency bottlenecks. Patent CN116288433A, published in June 2023, presents a transformative approach to synthesizing 2-methyl-4-acetoxy-2-butenoic acid (FCAA) from its aldehyde precursor, 2-methyl-4-acetoxy-2-butenal (FCA). This technology leverages a sophisticated cathode-anode cooperative electrochemical oxidation system that operates without the need for ion exchange membranes or hazardous chemical oxidants. By utilizing modified metal catalytic electrodes and ionic liquid solvents, the process achieves exceptional current efficiency and selectivity, addressing critical pain points in the manufacturing of complex pharmaceutical intermediates. For R&D directors and procurement strategists, this patent signifies a shift towards greener, more economically viable production methods that align with modern sustainability goals while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of FCA to FCAA has relied heavily on chemical oxidation methods, such as those disclosed in prior art like CN102863367A, which utilize inorganic oxidants including hydrogen peroxide and sodium chlorite. These traditional processes are inherently problematic due to their violent exothermic nature, necessitating strict low-temperature control throughout the reaction to prevent runaway scenarios and ensure safety. Furthermore, the reliance on halogen-based oxidants introduces significant downstream challenges, including the generation of large volumes of chloride-containing wastewater that require complex and costly treatment protocols to meet environmental discharge standards. The post-reaction workup is equally burdensome, often requiring quenching agents and multiple extraction steps to isolate the product, which inevitably leads to yield losses and increased operational expenditures. From a supply chain perspective, the dependency on stoichiometric amounts of hazardous chemicals creates volatility in raw material sourcing and elevates the risk profile of the manufacturing facility.
The Novel Approach
In stark contrast, the novel electrochemical method described in the patent circumvents these issues by employing electricity as the primary reagent, thereby eliminating the need for external chemical oxidants and their associated quenching agents. The core innovation lies in the synergistic operation of a modified metal catalytic anode and a gas diffusion cathode within an ionic liquid medium, which collectively facilitate a highly selective oxidation pathway. This system effectively suppresses the competing oxygen evolution reaction that typically plagues conventional electrochemical cells, allowing for a dramatic improvement in current efficiency from approximately 54% in aqueous systems to over 96% in the optimized ionic liquid setup. The absence of an ion exchange membrane simplifies the reactor design and reduces capital expenditure, while the mild reaction conditions of 20-40°C ensure operational safety and energy efficiency. This paradigm shift not only enhances product purity by avoiding halogen contamination but also streamlines the entire production workflow, making it an attractive solution for cost reduction in vitamin A intermediate manufacturing.
Mechanistic Insights into Cooperative Electrochemical Oxidation
The mechanistic elegance of this process stems from the precise engineering of the electrode surfaces and the unique properties of the ionic liquid electrolyte. At the anode, a modified titanium-ruthenium mesh electrode, treated through ultrasonic cleaning and calcination, provides a tailored surface that pre-oxidizes the FCA substrate into a radical intermediate without triggering excessive oxygen evolution. Simultaneously, at the cathode, a modified graphite felt gas diffusion electrode facilitates the reduction of dissolved oxygen to generate reactive oxygen species (ROS) such as superoxide anions. These in-situ generated ROS then attack the FCA radical intermediate, driving the conversion to FCAA with remarkable specificity. The use of ionic liquids like [Bmim]BF4 or [Emim]BF4 is critical, as they offer a wide electrochemical window that permits higher operating potentials without solvent decomposition, while their negligible vapor pressure ensures minimal loss and environmental impact.
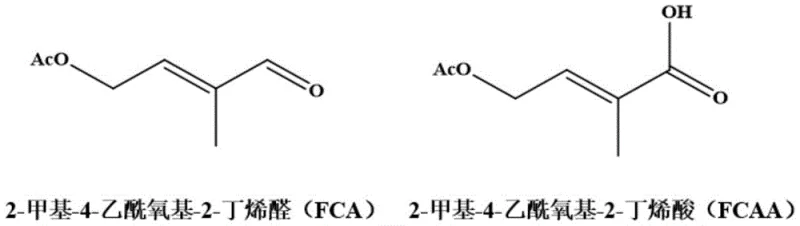
Furthermore, the impurity profile is tightly controlled through this mechanism, as the avoidance of harsh chemical oxidants prevents the formation of over-oxidized byproducts or chlorinated impurities that are common in traditional routes. The selectivity of the reaction exceeds 99%, ensuring that the resulting high-purity FCAA meets the stringent specifications required for downstream synthesis of carotenoids and Vitamin A derivatives. For technical teams, understanding this dual-electrode synergy is key to scaling the process, as it highlights the importance of maintaining optimal oxygen saturation and electrode potential to sustain the generation of active oxygen species. The stability of the ionic liquid solvent also plays a pivotal role in maintaining consistent reaction kinetics over prolonged operation, reducing the frequency of solvent replacement and minimizing waste generation.
How to Synthesize FCAA Efficiently
Implementing this advanced electrochemical protocol requires careful attention to electrode preparation and reaction parameter optimization to fully realize the benefits of the cooperative oxidation mechanism. The process begins with the meticulous modification of the graphite felt cathode and the titanium-ruthenium anode to ensure maximum surface area and catalytic activity. Once the electrode system is assembled in a three-electrode configuration, the reaction is conducted in an ionic liquid medium under a controlled oxygen atmosphere. Detailed standard operating procedures regarding the specific voltage ranges, stirring rates, and temperature controls are essential for reproducibility and safety. The following guide outlines the standardized synthesis steps derived from the patent examples to assist technical teams in replicating this high-efficiency route.
- Prepare the modified graphite felt cathode by acid boiling and cyclic voltammetry scanning to enhance oxygen reduction activity.
- Modify the titanium-ruthenium mesh anode through ultrasonic cleaning, acid pickling, and calcination to optimize oxidation potential.
- Conduct the electrolysis in an ionic liquid solvent at 20-40°C with oxygen saturation, maintaining a cathode potential of -1.5 to -1.8V.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology offers substantial strategic advantages that extend beyond mere technical performance. The elimination of stoichiometric chemical oxidants fundamentally alters the cost structure of the synthesis, removing the expense associated with purchasing, storing, and disposing of hazardous reagents like sodium chlorite. This transition to an electrically driven process significantly reduces the variable costs per kilogram of product, providing a more predictable and stable pricing model for long-term supply agreements. Additionally, the simplified downstream processing, which no longer requires extensive washing to remove inorganic salts or quenching agents, translates into reduced utility consumption and shorter batch cycle times. These operational efficiencies collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands without the bottlenecks associated with complex waste management.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of expensive chemical oxidants with electricity and oxygen, which are universally available and cost-effective resources. By removing the need for supporting electrolytes and ion exchange membranes, the process further lowers the material input costs and reduces the capital intensity of the reactor setup. The high current efficiency of over 96% ensures that electrical energy is utilized effectively, minimizing power waste and keeping operational expenditures low. Moreover, the ability to recover and reuse the ionic liquid solvent due to its high stability and low volatility creates a closed-loop system that drastically cuts down on raw material consumption over the lifecycle of the plant.
- Enhanced Supply Chain Reliability: Relying on electricity and oxygen as primary reagents mitigates the supply risks associated with specialized chemical oxidants, which can be subject to market volatility and regulatory restrictions. The robustness of the modified electrodes and the stability of the ionic liquid system ensure consistent production output, reducing the likelihood of batch failures or delays caused by reagent quality issues. This reliability is crucial for maintaining continuous supply to downstream customers in the pharmaceutical and nutraceutical sectors, where interruption can have significant commercial consequences. The simplified equipment requirements also mean that maintenance schedules are less frequent and less complex, further enhancing overall equipment effectiveness and uptime.
- Scalability and Environmental Compliance: The membrane-free design of the electrochemical cell simplifies scale-up efforts, as it removes the engineering challenges associated with sealing and maintaining large ion exchange membranes. The green nature of the process, characterized by the absence of halogen waste and volatile organic emissions, ensures compliance with increasingly stringent environmental regulations globally. This environmental stewardship not only avoids potential fines and remediation costs but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions. The mild reaction conditions further reduce the energy load for heating or cooling, making the process adaptable to various geographic locations with different energy infrastructures.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical oxidation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational feasibility and advantages of the method. Understanding these aspects is vital for stakeholders evaluating the integration of this process into existing manufacturing lines or new facility designs.
Q: Why is an ion exchange membrane not required in this electrochemical process?
A: The process utilizes a cooperative mechanism where the anode pre-oxidizes the substrate and the cathode generates reactive oxygen species (ROS) to complete the oxidation. This specific pathway avoids the competitive oxygen evolution reaction at the anode, eliminating the need for physical separation via a membrane.
Q: How does this method improve environmental compliance compared to chemical oxidation?
A: Unlike traditional chemical oxidation which relies on hazardous oxidants like sodium chlorite and generates significant halogen-containing wastewater, this electrochemical method uses electricity and oxygen as reagents. The use of stable ionic liquids further minimizes volatile organic compound (VOC) emissions and waste generation.
Q: What represents the primary cost advantage of using ionic liquids in this synthesis?
A: Ionic liquids serve as both the solvent and the electrolyte, removing the need for additional supporting salts. Their high stability and negligible volatility allow for efficient recovery and reuse, drastically reducing raw material consumption and waste disposal costs over time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable FCAA Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the electrochemical oxidation of FCA can be successfully translated into robust industrial processes. We are committed to delivering high-purity FCAA that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex electrochemical syntheses safely and efficiently, guaranteeing a consistent supply of high-quality intermediates for your Vitamin A and carotenoid projects.
We invite you to collaborate with us to explore how this advanced technology can optimize your supply chain and reduce overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals. Let us be your partner in driving efficiency and sustainability in the production of essential pharmaceutical intermediates.
