Advanced Catalytic Reduction for High-Purity 3,4'-Diaminodiphenyl Ether Production
Introduction to Advanced Monomer Synthesis
The chemical industry is constantly seeking safer and more efficient pathways for producing critical monomers used in high-performance polymers. Patent CN1485315A introduces a groundbreaking preparation method for 3,4'-diaminodiphenyl ether, a vital precursor in the synthesis of polyimides and other advanced materials. This innovation addresses significant safety and operational challenges found in legacy manufacturing processes by replacing hazardous high-pressure hydrogenation with a mild, catalytic reduction using hydrazine hydrate. By leveraging an iron-based catalyst system, specifically FeCl3·6H2O, this technology enables the production of high-purity intermediates under atmospheric pressure, drastically reducing capital expenditure on pressure vessels and vacuum systems. For R&D teams and procurement specialists alike, this represents a pivotal shift towards greener, more manageable chemistry that does not compromise on yield or quality. 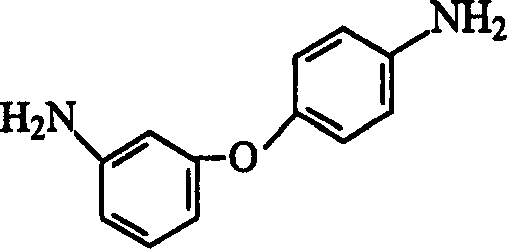
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4'-diaminodiphenyl ether has relied heavily on methods disclosed in older Japanese patents, such as JP-A-61-221159 and JP-A-54-88288. These conventional routes typically involve the condensation of aminophenols or nitrophenols with chloronitrobenzenes, followed by a critical reduction step using palladium on carbon (Pd/C) catalysts under high-pressure hydrogen conditions. This approach presents severe limitations for modern manufacturing, primarily due to the substantial equipment investment required for high-pressure reactors and the inherent safety risks associated with handling hydrogen gas at elevated pressures. Furthermore, the post-treatment phase in these legacy methods necessitates high-vacuum distillation to isolate the pure product, a process known for poor heat transfer efficiency and a high propensity for coking at the bottom of the still, leading to difficult cleaning procedures and potential product degradation.
The Novel Approach
In stark contrast, the novel methodology outlined in CN1485315A utilizes nitrodiphenyl ether as the starting material and employs hydrazine hydrate as the reducing agent in the presence of an iron chloride catalyst. This chemical transformation allows the reaction to proceed smoothly at atmospheric pressure, completely eliminating the need for dangerous high-pressure hydrogenation equipment. The reaction scheme is straightforward and robust, utilizing common alcoholic solvents like ethanol to facilitate the reduction at mild temperatures between 50-80°C. 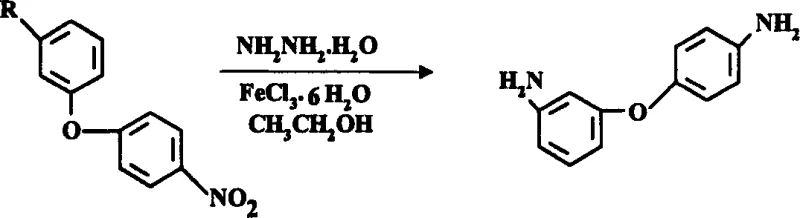 By avoiding high-vacuum distillation and instead using a simple precipitation method involving water addition, this approach not only enhances operational safety but also streamlines the downstream processing, making it an ideal solution for cost reduction in electronic chemical manufacturing.
By avoiding high-vacuum distillation and instead using a simple precipitation method involving water addition, this approach not only enhances operational safety but also streamlines the downstream processing, making it an ideal solution for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Hydrazine Reduction
The core of this technological advancement lies in the efficient activation of the nitro group reduction by the ferric chloride hexahydrate (FeCl3·6H2O) catalyst. In this mechanistic pathway, the iron species likely acts as a Lewis acid or an electron transfer mediator, facilitating the donation of electrons from the hydrazine hydrate to the nitro groups on the diphenyl ether backbone. This catalytic cycle is highly selective, ensuring that the nitro groups are reduced to amino groups without affecting the sensitive ether linkage or causing unwanted side reactions that often plague harsher reduction environments. The presence of activated carbon in the reaction mixture further aids in adsorbing impurities and potentially stabilizing the catalyst surface, contributing to the overall cleanliness of the reaction profile and the high purity of the final isolate.
From an impurity control perspective, this mechanism offers distinct advantages over thermal or high-pressure catalytic methods. The mild reaction conditions prevent the thermal decomposition of the product, which is a common issue during high-vacuum distillation where localized overheating can occur. The subsequent workup procedure, which involves filtering the crude product and inducing crystallization by adding water to the ethanolic filtrate, effectively separates the target 3,4'-diaminodiphenyl ether from soluble byproducts and catalyst residues. This results in a white crystalline product with a purity of ≥99% as determined by HPLC, matching the melting point specifications of 74-76°C found in literature, thereby ensuring the material is suitable for demanding applications in polyimide synthesis.
How to Synthesize 3,4'-Diaminodiphenyl Ether Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins with the suspension of the nitro-precursor and catalyst in an alcoholic solvent, followed by the controlled addition of the reducing agent under an inert atmosphere to prevent oxidation. Maintaining the temperature within the 50-80°C range is critical for optimizing the reaction kinetics while ensuring safety. The detailed standardized synthetic steps below outline the precise stoichiometry and operational parameters required to achieve the reported yields of 89-93%.
- Charge nitrodiphenyl ether, activated carbon, and FeCl3·6H2O catalyst into an alcoholic solvent (ethanol) under inert atmosphere.
- Heat the mixture to 50-80°C and slowly add hydrazine hydrate (1.5-3.0 molar equivalents) over one hour.
- Reflux for 2-5 hours, filter hot, add water to the filtrate to precipitate the white crystal product, then dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this hydrazine-based reduction technology offers compelling economic and logistical benefits that directly impact the bottom line. By shifting away from high-pressure hydrogenation, manufacturers can significantly reduce capital expenditure (CAPEX) associated with specialized pressure-rated reactors and hydrogen storage infrastructure. Furthermore, the elimination of high-vacuum distillation simplifies the plant layout and reduces energy consumption, leading to substantial operational cost savings. The use of inexpensive and readily available reagents like iron chloride and ethanol, as opposed to precious metal catalysts like palladium, further drives down the variable costs of production, making this a highly attractive route for cost reduction in electronic chemical manufacturing.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with abundant iron salts drastically lowers raw material costs, while the atmospheric pressure operation removes the need for costly high-pressure maintenance and safety certifications. Additionally, the simplified purification process avoids the energy-intensive high-vacuum distillation step, resulting in lower utility bills and reduced waste generation from still bottoms. These factors combine to create a leaner manufacturing process that maximizes margin potential without sacrificing product quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethanol and hydrazine hydrate ensures a stable and resilient supply chain, minimizing the risk of disruptions often associated with specialty catalysts or high-purity hydrogen gas. The robustness of the reaction conditions allows for flexible production scheduling and easier scale-up, ensuring that delivery timelines for high-purity polyimide monomers can be met consistently even during periods of high market demand.
- Scalability and Environmental Compliance: Operating at atmospheric pressure and moderate temperatures inherently reduces the environmental footprint of the process by lowering energy requirements and minimizing the risk of catastrophic failures. The aqueous workup and filtration steps generate waste streams that are generally easier to treat compared to the complex residues from high-temperature distillation, facilitating compliance with increasingly stringent environmental regulations and supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,4'-diaminodiphenyl ether using this novel catalytic reduction method. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on process safety, purity standards, and scalability for potential partners and clients.
Q: What are the safety advantages of this hydrazine reduction method over traditional hydrogenation?
A: Unlike traditional methods requiring high-pressure hydrogen gas and expensive Pd/C catalysts, this process operates at atmospheric pressure using hydrazine hydrate. This eliminates the explosion risks associated with high-pressure hydrogenation and removes the need for complex high-vacuum distillation equipment, significantly enhancing operational safety.
Q: How is product purity controlled in this synthesis route?
A: The process utilizes a specific crystallization technique where water is added to the ethanolic filtrate to precipitate the product. This simple yet effective purification step yields 3,4'-diaminodiphenyl ether with a purity of ≥99% (HPLC) and a melting point consistent with literature standards (74-76°C), avoiding the thermal degradation often seen in high-vacuum distillation.
Q: Can this process be scaled for industrial production of polyimide monomers?
A: Yes, the patent explicitly states the method is suitable for large-scale industrial production. The use of common solvents like ethanol, inexpensive iron catalysts, and mild reaction conditions (50-80°C) makes the process highly scalable and cost-effective for manufacturing electronic chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4'-Diaminodiphenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of next-generation polyimides and electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3,4'-diaminodiphenyl ether performs consistently in your downstream polymerization processes.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain stability and reduce your overall production costs.
