Revolutionizing 3,4'-Diaminodiphenyl Ether Production: Safe Catalytic Reduction for Industrial Scale-Up
Revolutionizing 3,4'-Diaminodiphenyl Ether Production: Safe Catalytic Reduction for Industrial Scale-Up
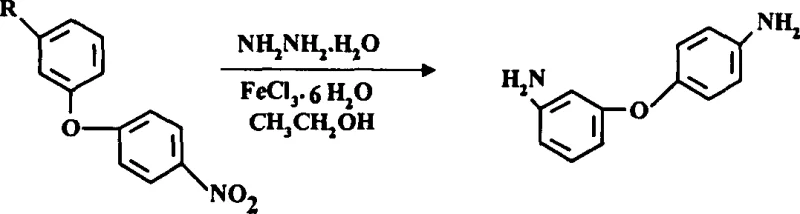
The global demand for high-performance polyimide materials continues to surge, driving the need for reliable and safe supply chains of critical monomers like 3,4'-diaminodiphenyl ether. Patent CN1263729C introduces a transformative manufacturing methodology that replaces hazardous high-pressure hydrogenation with a mild, iron-catalyzed hydrazine reduction system. This technological leap addresses long-standing safety concerns and operational inefficiencies inherent in legacy production routes, offering a robust pathway for the commercial scale-up of complex polyimide monomers. By shifting the reaction paradigm to atmospheric conditions, this process not only enhances worker safety but also significantly lowers the barrier to entry for manufacturers seeking to optimize their capital expenditure. The strategic implementation of this patent allows producers to deliver high-purity 3,4'-diaminodiphenyl ether with consistent quality metrics, ensuring downstream polymer performance remains uncompromised while mitigating industrial risk.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4'-diaminodiphenyl ether has relied heavily on catalytic hydrogenation using palladium on carbon (Pd/C) under elevated pressures, a method fraught with significant operational hazards and engineering challenges. The requirement for high-pressure reactors necessitates substantial capital investment in specialized equipment designed to withstand extreme conditions, thereby inflating the initial setup costs for any production facility. Furthermore, the post-reaction purification traditionally involves high-vacuum distillation, a thermally aggressive process that frequently results in the formation of tars and coke at the bottom of the distillation still. This coking phenomenon not only reduces heat transfer efficiency over time but also creates severe maintenance bottlenecks, requiring frequent and difficult cleaning cycles that disrupt production continuity. Additionally, the thermal sensitivity of the diamine product during high-temperature distillation can lead to degradation, potentially introducing impurities that compromise the quality of the final polyimide resin. These cumulative factors render the conventional high-pressure hydrogenation route increasingly unattractive for modern, safety-conscious chemical manufacturing environments.
The Novel Approach
In stark contrast, the innovative process disclosed in CN1263729C utilizes a hydrazine hydrate reduction system catalyzed by ferric chloride hexahydrate (FeCl3·6H2O) in an alcoholic solvent, operating effectively at atmospheric pressure. This method eliminates the need for dangerous high-pressure hydrogen gas and the associated heavy-duty reactor infrastructure, allowing the reaction to proceed safely at moderate temperatures ranging from 50°C to 80°C. The replacement of high-vacuum distillation with a simple crystallization step—achieved by adding water to the alcoholic filtrate—represents a major simplification of the downstream processing workflow. This gentle isolation technique prevents thermal degradation, ensuring that the structural integrity of the 3,4'-diaminodiphenyl ether is preserved throughout the purification stage. Consequently, this novel approach offers a streamlined, safer, and more economically viable alternative that aligns perfectly with the goals of cost reduction in fine chemical intermediates manufacturing while maintaining rigorous quality standards.
Mechanistic Insights into FeCl3-Catalyzed Hydrazine Reduction
The core of this technological advancement lies in the efficient electron transfer facilitated by the iron catalyst, which activates the hydrazine molecule for the reduction of the nitro group to the amine functionality. In this catalytic cycle, the FeCl3·6H2O acts as a Lewis acid and redox mediator, lowering the activation energy required for the cleavage of the nitrogen-oxygen bonds in the nitrodiphenyl ether substrate. The presence of activated carbon in the reaction mixture further aids in adsorbing potential by-products and stabilizing the reaction environment, ensuring a clean conversion profile. The reaction proceeds through a series of intermediate reduction steps, ultimately yielding the diamine with high selectivity, as evidenced by the minimal formation of azo or azoxy coupling by-products often seen in less controlled reductions. This mechanistic precision is critical for achieving the high purity levels required for electronic-grade polyimide applications, where trace impurities can detrimentally affect dielectric properties.
Furthermore, the impurity control mechanism is inherently superior due to the avoidance of thermal stress during workup. Unlike distillation, which subjects the molecule to prolonged heating that can induce rearrangement or decomposition, the crystallization method relies on solubility differences to isolate the product. By carefully controlling the addition of water to the ethanolic solution, the target 3,4'-diaminodiphenyl ether precipitates as white crystals, leaving soluble impurities in the mother liquor. This physical separation method is highly effective at removing residual catalyst, unreacted starting materials, and hydrazine by-products, resulting in a product with a purity of ≥99% as confirmed by HPLC analysis. The consistency of the melting point (74-76°C) across different batches further validates the robustness of this purification strategy, providing R&D teams with confidence in the material's reproducibility for sensitive polymerization reactions.
How to Synthesize 3,4'-Diaminodiphenyl Ether Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing 3,4'-diaminodiphenyl ether with exceptional safety and yield profiles. The process begins with the preparation of a reaction slurry containing the nitro-precursor, activated carbon, and the iron catalyst in an alcohol solvent such as ethanol, all under an inert nitrogen atmosphere to prevent oxidation. Following the addition of hydrazine hydrate and a controlled reflux period, the workup involves a straightforward filtration and anti-solvent crystallization, bypassing the complexities of distillation entirely. For detailed operational parameters, stoichiometry, and specific temperature controls required to replicate this high-efficiency synthesis, please refer to the standardized guide below.
- Prepare the reaction mixture by adding nitrodiphenyl ether, activated carbon, and FeCl3·6H2O catalyst into an alcohol solvent (preferably ethanol) under an inert nitrogen atmosphere.
- Heat the mixture to 50-80°C and slowly add hydrazine hydrate (1.5-3.0 molar equivalents) over one hour, maintaining reflux for 2-5 hours to ensure complete reduction.
- Filter the hot reaction mixture to remove catalyst and carbon, then add water to the filtrate to precipitate the white crystalline product, which is then washed and dried.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this FeCl3-catalyzed hydrazine reduction process translates into tangible strategic benefits that extend far beyond simple chemical synthesis. By fundamentally altering the reaction conditions from high-pressure to atmospheric, the technology removes the dependency on scarce and expensive high-pressure hydrogenation infrastructure, thereby diversifying the potential supplier base. This shift also mitigates the risk of production stoppages caused by safety inspections or equipment failures associated with high-pressure systems, ensuring a more resilient and continuous supply of this critical polyimide monomer. The simplification of the purification train further reduces energy consumption and maintenance downtime, contributing to a leaner and more responsive manufacturing operation capable of meeting tight delivery schedules.
- Cost Reduction in Manufacturing: The elimination of high-pressure reactors and high-vacuum distillation columns results in a drastic reduction in both capital expenditure (CAPEX) and operational expenditure (OPEX). Without the need for expensive hydrogen gas handling systems and the energy-intensive vacuum pumps required for distillation, the overall production cost per kilogram is significantly lowered. Additionally, the removal of the coking issue associated with distillation reduces the frequency of reactor cleaning and maintenance, further decreasing labor and downtime costs. This economic efficiency allows suppliers to offer more competitive pricing structures while maintaining healthy margins, directly benefiting the bottom line of downstream polymer manufacturers.
- Enhanced Supply Chain Reliability: Operating at atmospheric pressure and moderate temperatures significantly reduces the safety risks associated with chemical production, leading to fewer regulatory hurdles and insurance premiums. The use of readily available reagents like hydrazine hydrate and iron salts, rather than precious metal catalysts like palladium, insulates the supply chain from volatility in the precious metals market. This stability ensures that production can continue uninterrupted even during periods of raw material scarcity, providing a dependable source of reliable polyimide monomer supplier capabilities for long-term contracts. The robust nature of the process also facilitates easier technology transfer between manufacturing sites, enhancing global supply security.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving filtration and crystallization rather than complex distillation, makes this process exceptionally easy to scale from pilot plant to multi-ton commercial production. The absence of high-temperature thermal degradation minimizes the generation of hazardous tarry waste, simplifying wastewater treatment and solid waste disposal protocols. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the corporate sustainability profile of the manufacturer. The ability to scale rapidly without proportional increases in complexity or risk makes this technology ideal for meeting surging market demand for high-performance electronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,4'-diaminodiphenyl ether using this advanced catalytic reduction method. These insights are derived directly from the experimental data and process descriptions found in patent CN1263729C, providing a factual basis for evaluating the technology's viability. Understanding these details is essential for technical teams assessing the feasibility of integrating this monomer into their specific polymer formulations or supply chains.
Q: How does the FeCl3-catalyzed hydrazine method improve safety compared to traditional hydrogenation?
A: Traditional methods require high-pressure hydrogenation which poses significant explosion risks and requires expensive specialized reactors. The patented FeCl3-catalyzed hydrazine reduction operates at atmospheric pressure and moderate temperatures (50-80°C), drastically reducing operational hazards and equipment investment costs.
Q: Why is the purification process superior in this new method?
A: Conventional processes rely on high-vacuum distillation which often leads to thermal decomposition, coking at the still bottom, and difficult cleaning. This new method utilizes a simple crystallization technique by adding water to the alcoholic filtrate, yielding high-purity product (≥99%) without thermal stress or complex distillation equipment.
Q: What are the typical yield and purity specifications achievable with this process?
A: According to the patent data, this process consistently achieves yields between 89% and 93% with a purity of ≥99% (HPLC). The melting point of the product is 74-76°C, consistent with high-quality literature standards, making it suitable for demanding polyimide applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4'-Diaminodiphenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of next-generation polyimide materials, and we are committed to delivering excellence in every batch. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that our 3,4'-diaminodiphenyl ether meets the exacting standards required for electronic and aerospace applications. By leveraging advanced catalytic technologies like the one described in CN1263729C, we provide a supply solution that balances cost-effectiveness with uncompromising quality and safety.
We invite you to collaborate with us to optimize your material sourcing strategy and achieve significant efficiencies in your manufacturing operations. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced production capabilities can support your long-term growth and innovation goals.
