Advanced Citral-Based Synthesis of Beta-Damascenone for Industrial Fragrance Manufacturing and Global Supply
The global fragrance and flavor industry continuously demands high-purity aroma chemicals that combine olfactory excellence with sustainable manufacturing practices. Patent CN109053407B introduces a transformative methodology for synthesizing beta-damascenone, a precious compound renowned for its intense rose and raspberry notes, directly from citral. This technical breakthrough addresses long-standing challenges in the fine chemical sector by replacing hazardous legacy reagents with safer, scalable alternatives while maintaining exceptional product quality. The disclosed process not only streamlines the production workflow but also enhances the economic viability of manufacturing this high-value spice, making it an ideal candidate for reliable fragrance intermediate supplier networks seeking to optimize their portfolios. By leveraging improved Pinnick oxidation and controlled acid catalysis, this route offers a robust solution for the commercial scale-up of complex fragrance intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of beta-damascenone has been plagued by severe safety hazards and environmental inefficiencies inherent in older chemical pathways. As illustrated in prior art such as the route reported in Helv. Chim. Acta (1970), traditional methods often necessitate the use of highly toxic aniline for cyclization steps, generating substantial volumes of amine nitrogen wastewater that require costly treatment. Furthermore, these legacy processes frequently rely on non-commercialized reagents like allyl bromide, which must be prepared in situ through troublesome procedures, and employ heavy metal oxidants such as manganese dioxide or chromium-containing reagents that pose significant disposal challenges. The reliance on N-bromosuccinimide (NBS) in final reaction stages further exacerbates the generation of three wastes, creating a complex operational landscape that is increasingly difficult to justify under modern environmental regulations and safety standards.
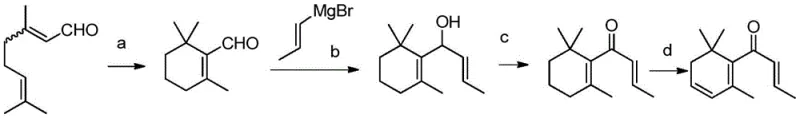
The Novel Approach
In stark contrast, the methodology disclosed in CN109053407B represents a paradigm shift towards greener and more efficient chemical manufacturing by utilizing citral as a readily available starting feedstock. This innovative route bypasses the need for toxic aniline and dangerous heavy metals, instead employing sodium chlorite for oxidation and concentrated phosphoric acid for cyclization under mild thermal conditions. The process is meticulously designed to be operationally simple, avoiding the extreme risks associated with potassium hydride or lithium amide found in other historical patents, thereby drastically simplifying the reactor requirements and safety protocols. Additionally, this approach uniquely enables the synchronous synthesis of alpha-damascone, another valuable spice, effectively doubling the utility of the production line and offering substantial cost savings in synthetic flavors manufacturing through improved atom economy and resource utilization.
Mechanistic Insights into Pinnick Oxidation and Acid-Catalyzed Cyclization
The mechanistic foundation of this synthesis begins with a highly selective Pinnick oxidation, where citral is converted into geranic acid using sodium chlorite in the presence of a scavenger like dipentene and a buffer system. This step is critical for preventing the over-oxidation or degradation of the sensitive aldehyde functionality, ensuring high yields of the carboxylic acid intermediate which serves as the precursor for ring closure. Following isolation, the geranic acid undergoes a sophisticated acid-catalyzed cyclization using concentrated phosphoric acid at elevated temperatures, a transformation that constructs the cyclohexene core with high regioselectivity. The subsequent activation of the acid via thionyl chloride and elimination with triethylamine generates the key enone intermediate, setting the stage for the crucial carbon-carbon bond formation that defines the molecular skeleton of the target damascones.
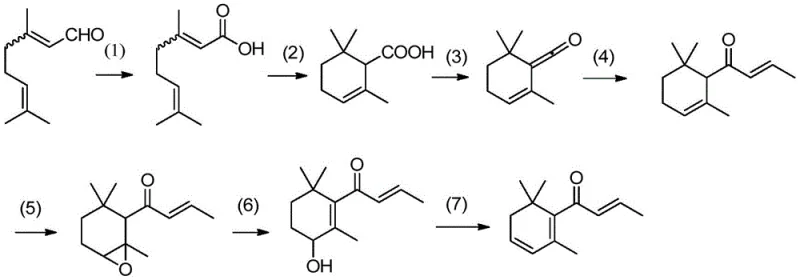
Further mechanistic refinement occurs during the Grignard addition phase, where allyl magnesium chloride reacts with the cyclic enone to introduce the necessary side chain, followed by an acid-catalyzed isomerization that establishes the conjugated ketone system of alpha-damascone. The final transformation into beta-damascenone involves a delicate sequence of regioselective epoxidation of the endocyclic double bond, followed by alkaline ring opening to form a hydroxy intermediate. The process concludes with a p-toluenesulfonic acid catalyzed dehydration, which eliminates water to restore the conjugated diene system characteristic of beta-damascenone. Throughout these steps, impurity control is rigorously managed through specific extraction protocols and recrystallization techniques, ensuring that the final high-purity beta-damascenone meets the stringent purity specifications required for premium fragrance applications.
How to Synthesize Beta-Damascenone Efficiently
The synthesis of beta-damascenone via this patented route involves a sequential seven-step protocol that transforms citral into the final fragrance molecule through oxidation, cyclization, and functional group manipulation. The process is designed for industrial robustness, utilizing common solvents like toluene and dichloromethane to facilitate easy separation and purification at each stage. Operators must strictly adhere to temperature controls during the exothermic oxidation and Grignard addition steps to maintain safety and maximize yield. For a detailed breakdown of the specific reaction conditions, molar ratios, and workup procedures required to execute this synthesis successfully, please refer to the standardized technical guide provided below.
- Oxidize citral using sodium chlorite under buffered conditions to form geranic acid, followed by acid-catalyzed cyclization to alpha-cyclogeranic acid.
- Convert alpha-cyclogeranic acid to cyclogeranenone using thionyl chloride, then perform Grignard addition with allyl chloride to yield alpha-damascone.
- Subject alpha-damascone to selective epoxidation, alkaline ring opening, and final acid-catalyzed dehydration to obtain beta-damascenone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this citral-based synthesis route offers compelling strategic advantages that directly address the pain points of cost volatility and raw material scarcity. By shifting away from specialized, hard-to-source reagents like bromo-mesityl oxide or methyl crocetinate, manufacturers can leverage the global availability of citral, a commodity chemical with a stable and mature supply chain. This transition not only mitigates the risk of production stoppages due to raw material shortages but also simplifies the logistics of inventory management, allowing for more predictable planning and reduced lead time for high-purity fragrance intermediates. The elimination of complex multi-step preparations for reagents further streamlines the procurement process, enabling teams to focus on strategic sourcing rather than crisis management.
- Cost Reduction in Manufacturing: The economic architecture of this process is fundamentally optimized through the removal of expensive and hazardous catalysts, such as heavy metals and strong bases like potassium hydride, which traditionally drive up operational expenditures. By utilizing cost-effective reagents like sodium chlorite and phosphoric acid, the direct material costs are significantly lowered, while the simplified waste treatment requirements reduce the overhead associated with environmental compliance. Furthermore, the ability to co-produce alpha-damascone within the same workflow creates an additional revenue stream, effectively subsidizing the production cost of beta-damascenone and enhancing the overall profitability of the manufacturing unit without requiring additional capital investment.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the reliance on widely available industrial chemicals rather than niche intermediates that are subject to market fluctuations and limited vendor pools. The use of standard solvents and reagents ensures that production can be sustained even during periods of regional supply disruption, providing a buffer against geopolitical or logistical instabilities. This robustness allows for consistent delivery schedules to downstream perfume and flavor houses, fostering stronger long-term partnerships and reducing the administrative burden of managing multiple backup suppliers for critical process inputs.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the mild reaction conditions and absence of toxic byproducts make this route exceptionally suitable for large-scale commercial expansion. The avoidance of liquid ammonia and metallic sodium eliminates the need for specialized cryogenic or inert atmosphere equipment, lowering the barrier to entry for scale-up and reducing the energy footprint of the facility. Additionally, the generation of benign waste streams simplifies effluent treatment, ensuring that the manufacturing process remains compliant with increasingly strict global environmental regulations while supporting sustainable growth initiatives.
Frequently Asked Questions (FAQ)
The following questions and answers address common technical and commercial inquiries regarding the synthesis of beta-damascenone, derived directly from the detailed specifications and comparative data within the patent literature. These insights are intended to clarify the operational benefits and chemical rationale behind this advanced manufacturing route for stakeholders evaluating its implementation. Understanding these nuances is essential for making informed decisions about technology adoption and supply chain integration in the competitive fragrance market.
Q: How does this new synthesis route improve safety compared to traditional methods?
A: Unlike legacy processes that utilize highly toxic aniline, dangerous potassium hydride (KH), or explosive lithium amide conditions, this method employs mild reagents like sodium chlorite and phosphoric acid, significantly reducing industrial hazard risks.
Q: What are the primary raw materials required for this production process?
A: The process relies on commercially abundant and cost-effective feedstocks, primarily citral, sodium chlorite, allyl chloride, and common organic solvents like toluene and dichloromethane, ensuring stable supply chain continuity.
Q: Does this manufacturing method allow for the co-production of other valuable fragrance compounds?
A: Yes, the synthetic pathway is designed to simultaneously produce alpha-damascone as a valuable intermediate or co-product, thereby maximizing the economic efficiency and value output of the manufacturing line.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Damascenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial realities for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of beta-damascenone performs consistently in your final fragrance formulations. Our infrastructure is designed to support the complex chemistry required for high-value intermediates, providing a secure foundation for your product development pipelines.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your supply chain for premium fragrance ingredients.
