Advanced Synthesis of Paroxetine Intermediates Using CDI Coupling for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for critical antiviral intermediates, particularly those associated with next-generation treatments like Paxlovid. Patent CN114605492A introduces a significant technological advancement in the preparation of a key Paroxetine intermediate, specifically identified as (1R,2S,5S)-3-[N-(trifluoroacetyl)-L-tert-leucine]-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid. This compound serves as a vital building block in the synthesis of complex protease inhibitors, where stereochemical purity and structural integrity are paramount for biological efficacy. The disclosed methodology represents a departure from traditional multi-step syntheses, offering a streamlined approach that addresses long-standing challenges in yield optimization and environmental sustainability. By focusing on a direct condensation strategy, this innovation provides a compelling alternative for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier capable of meeting stringent quality standards. The structural complexity of the target molecule, featuring a fused bicyclic system and multiple chiral centers, demands precise control over reaction conditions to prevent racemization and ensure the final product meets the rigorous specifications required for active pharmaceutical ingredient (API) production.
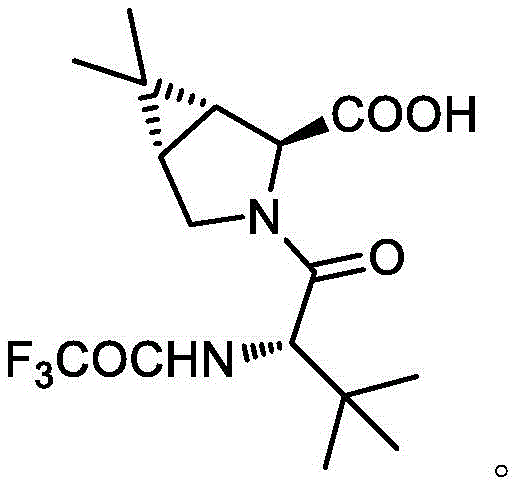
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, such as the process described in patent AU2021266232B1, relies on a cumbersome four-step sequence that significantly impacts both economic efficiency and environmental compliance. This legacy route typically involves the coupling of protected amino acids using expensive reagents like HATU (2-(7-azabenzotriazole)-N,N,N',N'-tetramethylurea hexafluorophosphate), followed by separate hydrolysis steps using strong bases like lithium hydroxide and acids like hydrochloric acid. These additional transformation stages not only extend the production timeline but also generate substantial volumes of acidic and basic waste streams, creating a heavy burden on wastewater treatment facilities. Furthermore, the use of HATU introduces cost volatility due to the high price of phosphonium salts, and the subsequent removal of urea byproducts often requires extensive purification efforts that erode overall yield. The cumulative effect of these inefficiencies results in a process that is difficult to scale economically, making it less attractive for high-volume commercial manufacturing where cost reduction in API manufacturing is a primary driver for procurement decisions.
The Novel Approach
In stark contrast, the methodology outlined in CN114605492A simplifies the synthesis into a direct condensation reaction between Compound I and Compound II, effectively bypassing the need for intermediate protection and deprotection cycles. As illustrated in the reaction scheme below, this novel approach utilizes N,N'-carbonyldiimidazole (CDI) as a highly efficient coupling agent, which activates the carboxylic acid moiety of the trifluoroacetylated tert-leucine derivative for immediate nucleophilic attack by the bicyclic amine. This strategic shift reduces the total number of unit operations, thereby minimizing material handling and potential points of failure in the production line. The elimination of harsh hydrolysis conditions not only enhances operator safety but also drastically reduces the generation of saline waste, aligning the process with modern green chemistry principles. By consolidating the synthesis into fewer steps with higher atom economy, this route offers a clear pathway for cost reduction in pharmaceutical intermediate manufacturing, providing supply chain leaders with a more resilient and predictable sourcing option for this critical molecular scaffold.
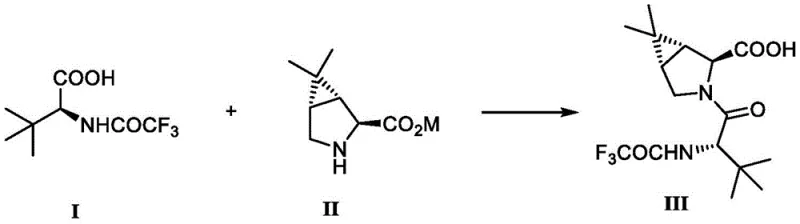
Mechanistic Insights into CDI-Mediated Amide Coupling
The core of this synthetic breakthrough lies in the mechanistic efficiency of CDI-mediated activation, which proceeds through the formation of a reactive acyl imidazole intermediate. When Compound I is treated with CDI in a solvent such as ethyl acetate, the carboxylic acid group attacks the electrophilic carbon of the carbonyldiimidazole, releasing imidazole and generating an activated ester species that is highly susceptible to nucleophilic substitution. This activation step is crucial because it avoids the formation of stable salts that can occur with other coupling reagents, ensuring that the reaction proceeds smoothly to the amide bond formation upon addition of Compound II. The choice of solvent plays a pivotal role in this mechanism; ethyl acetate is preferred not only for its solubility profile but also for its ability to stabilize the transition state without promoting side reactions. The reaction kinetics are carefully managed by controlling the temperature, typically maintaining conditions between -10°C and 30°C, which is essential to suppress the epimerization of the chiral alpha-carbon adjacent to the carbonyl group. This precise thermal control ensures that the stereochemical integrity of the L-tert-leucine moiety is preserved throughout the coupling process, resulting in a product with a favorable isomeric ratio.
Furthermore, the post-reaction workup strategy is designed to maximize purity while minimizing the retention of impurities such as unreacted starting materials or imidazole byproducts. The protocol specifies an acidic quench, often using dilute hydrochloric acid, which serves to protonate any unreacted amine and facilitate its removal into the aqueous phase during extraction. Following the separation of the organic layer, the crude product undergoes a specialized recrystallization process using a mixed solvent system of ethanol and water. This purification step is critical for achieving the high purity levels (>99%) required for pharmaceutical applications, as it effectively segregates the desired diastereomer from minor epimeric impurities that may have formed during the coupling. The ability to consistently produce material with such high optical purity demonstrates the robustness of the mechanism and provides R&D directors with confidence in the reproducibility of the process. The detailed understanding of these mechanistic nuances allows for fine-tuning of reaction parameters, ensuring that the commercial scale-up of complex pharmaceutical intermediates can be executed with minimal risk of batch-to-batch variability.
How to Synthesize Paroxetine Intermediate Efficiently
The execution of this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and stereochemical fidelity. The process begins with the activation of the acid component, followed by the controlled addition of the amine partner, and concludes with a rigorous purification sequence. Operators must monitor the reaction progress using analytical techniques such as HPLC or TLC to determine the precise endpoint, ensuring complete consumption of the limiting reagent before proceeding to workup. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for process engineers looking to implement this technology.
- Activate (S)-3,3-dimethyl-2-(2,2,2-trifluoroacetamido)butyric acid with CDI in ethyl acetate at controlled temperatures.
- React the activated intermediate with (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid lithium salt.
- Perform acidic workup followed by recrystallization to achieve high purity (>99%) and remove epimeric impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. The primary advantage lies in the significant simplification of the supply chain logistics, as the process relies on readily available starting materials and avoids the dependency on scarce or expensive coupling reagents like HATU. By eliminating the need for multiple intermediate isolation and purification steps, the overall lead time for production is drastically shortened, allowing for faster response to market demand fluctuations. This agility is crucial in the volatile landscape of antiviral drug manufacturing, where the ability to rapidly scale production can determine market share. Additionally, the reduction in solvent usage and the avoidance of hazardous hydrolysis reagents translate directly into lower operational expenditures related to waste disposal and environmental compliance, providing a tangible competitive edge in cost-sensitive markets.
- Cost Reduction in Manufacturing: The replacement of expensive phosphonium-based coupling agents with CDI represents a direct material cost saving, as CDI is generally more economical and generates benign byproducts like imidazole that are easier to remove. Furthermore, the consolidation of the synthesis into fewer steps reduces the consumption of utilities such as energy for heating and cooling, as well as labor hours required for monitoring and handling multiple batches. The elimination of the hydrolysis step removes the need for large quantities of lithium hydroxide and hydrochloric acid, further decreasing the raw material bill. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), enabling manufacturers to offer more competitive pricing to downstream API producers while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethyl acetate and standard inorganic acids enhances the resilience of the supply chain against disruptions. Unlike specialized reagents that may have limited suppliers or long lead times, the inputs for this process are widely available from multiple global sources, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and concentrations, also means that the process is less sensitive to minor variations in utility availability or equipment performance. This reliability ensures a consistent flow of high-purity intermediates, allowing pharmaceutical companies to maintain steady production schedules for their final drug products without the fear of unexpected delays.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns perfectly with the increasing regulatory pressure to reduce the ecological footprint of chemical manufacturing. The absence of heavy metal catalysts and the minimization of saline waste streams simplify the wastewater treatment process, making it easier for facilities to meet strict discharge limits. The use of ethyl acetate, a solvent with a favorable safety and environmental profile compared to chlorinated alternatives, further enhances the sustainability credentials of the process. These factors make the technology highly scalable, as the environmental permitting and waste management hurdles are significantly lower than those associated with traditional multi-step syntheses. Consequently, manufacturers can confidently invest in larger reactor capacities, knowing that the process is both economically viable and environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on critical process parameters and quality outcomes. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production lines.
Q: How does the CDI method improve yield compared to conventional HATU routes?
A: The CDI-mediated coupling eliminates the need for multiple hydrolysis and protection/deprotection steps found in older patents like AU2021266232B1, directly improving overall process yield and reducing material loss.
Q: What are the critical parameters for controlling epimer formation?
A: Maintaining reaction temperatures between -5°C and 25°C and utilizing ethyl acetate as the solvent are critical for minimizing epimerization, ensuring an isomer ratio favoring the desired product (approx. 90:10).
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process avoids expensive catalysts like HATU and hazardous hydrolysis reagents, utilizing standard solvents and simple workup procedures that are inherently safer and more cost-effective for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paroxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of advanced pharmaceutical intermediates for the global healthcare market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of major pharmaceutical contracts without compromising on quality. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our capability to implement the advanced CDI-coupling technology described in CN114605492A allows us to offer a superior product profile with improved cost-efficiency, positioning us as a strategic partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic advantages of switching to our supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your development timelines are met with precision and reliability. Let us collaborate to drive innovation and efficiency in the production of life-saving antiviral therapies.
