Advanced Total Synthesis of SN38: A Scalable Route for Oncology API Manufacturing
Introduction to Patent CN102250103A
The global demand for effective oncology therapeutics continues to drive innovation in the synthesis of complex natural product derivatives. Among these, SN38 (7-ethyl-10-hydroxycamptothecin) stands out as the active metabolite of Irinotecan, a potent topoisomerase I inhibitor used widely in treating colorectal and lung cancers. However, the commercial availability of SN38 has historically been bottlenecked by its reliance on plant extraction or semi-synthesis from scarce camptothecin. Patent CN102250103A, filed in November 2011, presents a groundbreaking solution by detailing a robust total synthesis method for SN38 and its key intermediates. This intellectual property outlines a pathway starting from simple, commercially available raw materials, utilizing straightforward reagents and concise operational steps to efficiently construct the highly conjugated pentacyclic structure. By shifting the production paradigm from agriculture-dependent extraction to chemical synthesis, this technology offers a strategic advantage for securing the supply chain of critical anticancer agents.
The significance of this patent lies in its ability to bypass the ecological and logistical limitations associated with harvesting Camptotheca acuminata. Traditional methods not only threaten the plant species but also suffer from batch-to-batch variability and low overall yields. In contrast, the disclosed synthetic route provides a reproducible, scalable, and chemically defined process. For pharmaceutical manufacturers and procurement strategists, this represents a pivotal opportunity to stabilize the supply of high-purity SN38. The methodology described ensures that the production of this vital intermediate is no longer subject to the vagaries of crop yields or geopolitical restrictions on plant resources, thereby enhancing the resilience of the global oncology drug supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of camptothecin derivatives has been fraught with significant challenges that impede efficient drug manufacturing. The primary conventional method involves the extraction of camptothecin from the bark and seeds of the Happy Tree (Camptotheca acuminata). This approach is inherently unsustainable due to the slow growth rate of the tree and the low content of the alkaloid within the plant tissue, often leading to destructive harvesting practices that endanger the species. Furthermore, the extraction process yields a complex mixture of alkaloids, requiring extensive and costly purification steps to isolate the desired compound. Once extracted, semi-synthetic modification to produce SN38 or Irinotecan introduces additional complexity, as the starting material itself is expensive and supply-constrained. These factors collectively result in high production costs, long lead times, and a fragile supply chain that is vulnerable to agricultural disruptions and regulatory changes regarding endangered plant species.
The Novel Approach
The synthetic strategy disclosed in CN102250103A fundamentally disrupts this status quo by enabling the total synthesis of SN38 from basic chemical building blocks. Instead of relying on a complex natural product scaffold, the route begins with simple aniline derivatives and keto-esters, which are abundant and cost-effective industrial commodities. The process is characterized by high atom economy and the use of reliable, well-understood chemical transformations. For instance, the initial construction of the quinoline core is achieved through a highly efficient cyclization reaction catalyzed by iron(III) chloride, delivering yields as high as 93%. This shift from extraction to synthesis allows for precise control over impurities and stereochemistry, ensuring a superior quality profile for the final API. By decoupling production from biological sources, this novel approach facilitates a more predictable and scalable manufacturing model suitable for meeting the growing global demand for cancer therapies.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Chiral Control
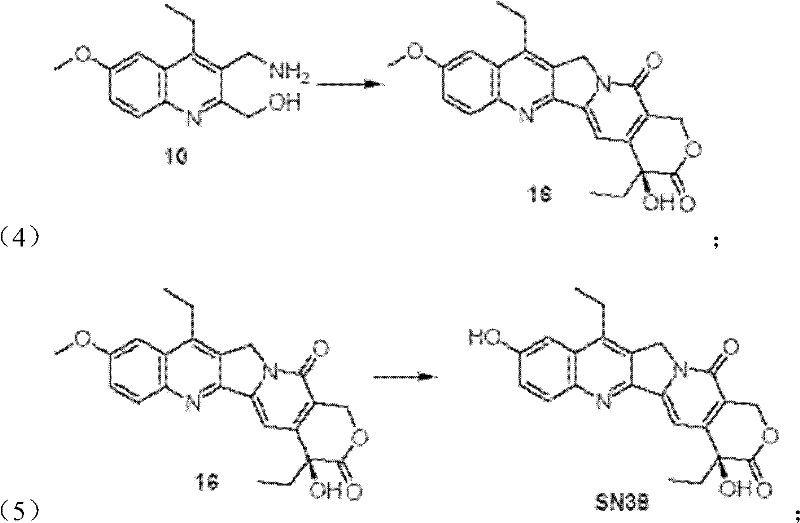
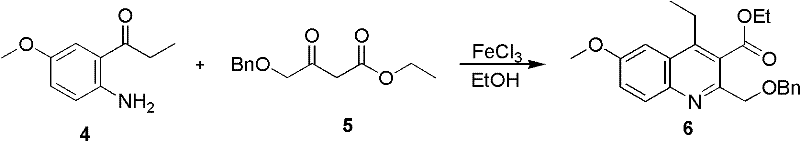
A critical aspect of this synthesis is the efficient construction of the ABC ring system, which forms the rigid planar core essential for DNA intercalation. The process initiates with a Friedel-Crafts-type cyclization where compound 4 and compound 5 react under the influence of FeCl3 in ethanol. This Lewis acid-catalyzed reaction promotes the condensation and subsequent ring closure to form the quinoline derivative (compound 6). The choice of FeCl3 is particularly advantageous from a process chemistry perspective; it is inexpensive, non-toxic compared to heavy metal alternatives, and easy to remove during workup. The reaction proceeds smoothly at room temperature over 15 hours, demonstrating mild conditions that are favorable for scale-up. The high yield of 93% reported in the examples indicates a clean reaction profile with minimal side products, which simplifies downstream purification and reduces solvent consumption. This step effectively sets the stage for the subsequent functionalization of the aromatic rings without requiring harsh conditions that could degrade sensitive functional groups.
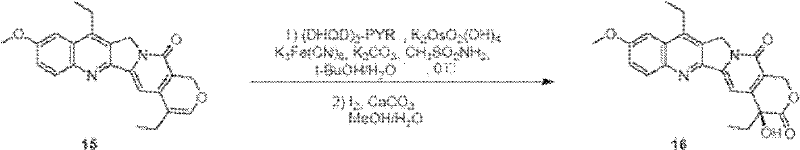
Beyond the core construction, the establishment of the chiral center at the C-20 position is paramount for the biological activity of SN38. The patent details a sophisticated sequence in Step 4 involving asymmetric dihydroxylation followed by oxidative lactonization. Specifically, compound 15 is subjected to Sharpless asymmetric dihydroxylation conditions using (DHQD)2-PYR as the chiral ligand and potassium osmate as the catalyst. This step introduces the necessary hydroxyl groups with high stereoselectivity. Subsequent treatment with iodine and calcium carbonate facilitates the oxidative lactonization, closing the E-ring and locking in the S-configuration at C-20. The process achieves an enantiomeric excess (ee) of 92%, which is critical for ensuring the potency of the final drug substance. This level of stereocontrol without the need for chiral resolution of the final product represents a significant technical achievement, streamlining the manufacturing process and maximizing the yield of the active enantiomer.
How to Synthesize SN38 Efficiently
The synthesis of SN38 described in this patent is a multi-step sequence that transforms simple precursors into a complex pentacyclic lactone. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions for the majority of the steps. Key transformations include the initial FeCl3-mediated cyclization, followed by a reduction-oxidation sequence to install the aldehyde functionality necessary for ring D construction. The subsequent steps involve oxime formation, hydrogenation, and a thermal cyclization to build the lactone ring. The final stages focus on establishing chirality and removing protecting groups to reveal the active phenol. For process chemists looking to implement this route, the detailed experimental conditions provided in the patent offer a clear roadmap for optimization. The standardized workup procedures, primarily involving extraction and column chromatography in the examples, can be adapted to crystallization and filtration for industrial scale. Detailed standardized synthesis steps are provided in the guide below.
- Condense compound 4 and 5 using FeCl3 catalyst in ethanol to form the quinoline core (compound 6).
- Reduce compound 6 with LiAlH4 to alcohol 7, then oxidize with Dess-Martin periodinane to aldehyde 8.
- Convert aldehyde 8 to oxime 9 using hydroxylamine hydrochloride, followed by hydrogenation to amine 10.
- Construct the E-ring via acylation, cyclization, and asymmetric dihydroxylation/lactonization to yield compound 16.
- Perform final demethylation on compound 16 to obtain the target molecule SN38.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend beyond mere technical feasibility. The primary advantage is the drastic reduction in supply risk. By transitioning from a plant-dependent supply chain to a petrochemical-based synthesis, manufacturers can secure a continuous and reliable flow of SN38 intermediates regardless of agricultural seasons or environmental regulations. This stability is crucial for maintaining the production schedules of life-saving oncology drugs. Furthermore, the use of commodity chemicals as starting materials means that raw material costs are significantly lower and less volatile than botanical extracts. The high yields observed in key steps, such as the 93% yield in the first cyclization and 94% in the oxime formation, contribute to substantial cost savings by minimizing raw material waste and maximizing throughput per batch. This efficiency directly translates to a more competitive cost structure for the final API.
- Cost Reduction in Manufacturing: The elimination of expensive botanical extraction processes and the reliance on cheap, bulk-available chemical reagents leads to a significant decrease in the cost of goods sold (COGS). The process avoids the use of precious metal catalysts in the early stages, utilizing iron salts instead, which further optimizes the cost profile. Additionally, the high convergence of the synthesis minimizes the number of isolation steps, reducing solvent usage and energy consumption associated with drying and purification. These factors collectively drive down the manufacturing cost, allowing for better margin management in a competitive generic drug market.
- Enhanced Supply Chain Reliability: Dependence on a single geographic source for plant extraction creates a single point of failure in the supply chain. This synthetic route diversifies the source of supply, allowing for production in multiple geographic locations with access to basic chemical infrastructure. The robustness of the reaction conditions, many of which proceed at room temperature or moderate heating, ensures that the process is less susceptible to equipment failures or utility fluctuations. This reliability enables procurement teams to negotiate longer-term contracts with greater confidence, knowing that supply interruptions due to raw material scarcity are virtually eliminated.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, utilizing reactions that are well-suited for large-scale reactor vessels. The avoidance of hazardous reagents and the use of standard solvents like ethanol and ethyl acetate simplify waste treatment and regulatory compliance. The high atom economy of the key cyclization steps reduces the generation of chemical waste, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing site. This ease of scale-up ensures that production can be rapidly ramped from pilot scale to commercial tonnage to meet surging market demand without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this SN38 synthesis technology. These answers are derived directly from the experimental data and claims within patent CN102250103A, providing clarity on the feasibility and advantages of the route. Understanding these details is essential for R&D teams evaluating the technology for technology transfer and for quality assurance teams assessing the control strategy for critical quality attributes like chirality and purity.
Q: What are the primary advantages of this synthetic route over plant extraction?
A: This total synthesis route eliminates dependence on the scarce Camptotheca acuminata plant, ensuring consistent supply不受 seasonal or ecological constraints. It utilizes simple, commercially available starting materials and achieves high yields (e.g., 93% in the first step), significantly reducing raw material costs and environmental impact compared to extraction methods.
Q: How is the critical C-20 chiral center controlled in this process?
A: The stereochemistry at the C-20 position is established during the conversion of compound 15 to 16. The process employs an asymmetric dihydroxylation using (DHQD)2-PYR and K2OsO2(OH)4 catalysts, followed by oxidative lactonization. This method achieves a high enantiomeric excess of 92% ee, ensuring the biological activity required for topoisomerase I inhibition.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly designs the route for industrial feasibility. It avoids exotic reagents, relying on standard catalysts like FeCl3 and Pd/C, and uses common solvents such as ethanol and dichloromethane. The operations involve standard unit processes like heating, extraction, and crystallization, which are readily adaptable from kilogram to multi-ton scales in a GMP environment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SN38 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-value oncology intermediates like SN38. Our technical team has thoroughly analyzed the synthetic route disclosed in CN102250103A and possesses the expertise to execute this complex chemistry with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and quality. Our facilities are equipped with rigorous QC labs capable of monitoring stringent purity specifications, including the critical enantiomeric excess at the C-20 position. We are committed to delivering high-purity SN38 and its intermediates that meet the demanding requirements of global regulatory agencies, supporting your drug development and commercial manufacturing goals.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your procurement budget. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us be your partner in advancing cancer therapy through reliable and innovative chemical manufacturing.
