Revolutionizing Phenacetin Manufacturing with Reactor-Coupled Simulated Moving Bed Technology
Revolutionizing Phenacetin Manufacturing with Reactor-Coupled Simulated Moving Bed Technology
The pharmaceutical and fine chemical industries are constantly seeking methodologies that reconcile high production efficiency with stringent environmental standards. A pivotal advancement in this domain is documented in patent CN101823979B, which outlines a clean production process for para-acetamino phenetole, commonly known as phenacetin. This technology represents a paradigm shift from traditional batch processing to a sophisticated integration of catalytic reaction and simulated moving bed (SMB) chromatography. By coupling the reactor directly with a chromatographic separation system, this innovation addresses critical pain points regarding purity, waste generation, and raw material utilization. For R&D directors and process engineers, understanding the nuances of this one-pot reduction and acetylation method is essential for developing next-generation synthetic routes that are both economically viable and environmentally sustainable.
The core breakthrough lies in the ability to perform reduction and acetylation reactions within a single reactor vessel, followed immediately by high-efficiency separation without the need for intermediate isolation or aqueous workups. This approach not only streamlines the operational workflow but also fundamentally alters the impurity profile of the final product. As global regulatory bodies impose tighter restrictions on solvent residues and heavy metal contaminants, the adoption of such clean technologies becomes a strategic imperative for any reliable phenacetin supplier aiming to serve top-tier pharmaceutical markets. The following analysis dissects the technical merits and commercial implications of this patented methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
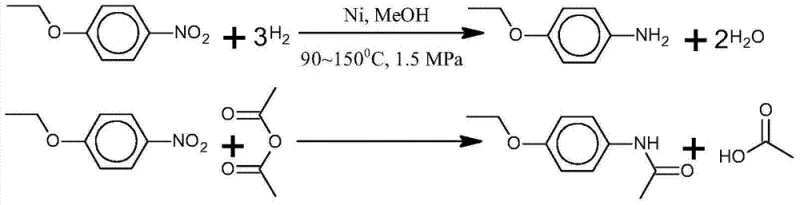
Traditional synthesis of phenacetin typically involves a multi-step sequence starting from p-nitrophenetole. Historically, this process requires the initial reduction of the nitro group to an amine (p-phenetidine), followed by isolation, neutralization, and washing to remove catalysts and byproducts before proceeding to the acetylation step. These conventional unit operations are fraught with inefficiencies. The necessity for aqueous washing generates substantial volumes of wastewater laden with organic residues and salts, creating a significant burden on waste treatment facilities. Furthermore, the isolation of the intermediate p-phenetidine often leads to yield losses due to mechanical transfer and oxidation sensitivity. The reliance on neutralization agents introduces additional inorganic impurities that are difficult to remove completely, potentially compromising the quality of the final API intermediate. From a supply chain perspective, these discontinuous steps extend the production cycle time and increase the consumption of auxiliary materials, thereby inflating the overall cost of goods sold.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes a reactor-coupled simulated moving bed system to achieve a seamless transition from raw materials to high-purity product. This method employs a one-pot strategy where p-nitrophenetole undergoes catalytic hydrogenation and subsequent acetylation in the presence of acetic anhydride. The brilliance of this design is the elimination of the neutralization and washing steps entirely. Instead of quenching the reaction with water, the mixture is subjected to solid-liquid separation, and the liquid phase is directly fed into the SMB chromatography unit. Here, acetic anhydride serves a dual function as both the reaction medium and the eluent for separation. This integration ensures that unreacted starting materials and intermediates are efficiently separated from the product and recycled back into the reactor. The result is a closed-loop system that drastically reduces raw material consumption and prevents the formation of wastewater, offering a compelling solution for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Reactor-Coupled Simulated Moving Bed Chromatography
The chemical foundation of this process rests on a tandem catalytic sequence. Initially, p-nitrophenetole is subjected to hydrogenation in the presence of a heterogeneous catalyst, such as nickel, cobalt, or platinum supported on activated carbon or alumina. Under conditions ranging from 30 to 160°C and pressures between 0.2 and 2.2 MPa, the nitro group is reduced to an amino group, forming p-phenetidine. Crucially, this reduction occurs in an anhydrous environment dominated by acetic anhydride. As soon as the p-phenetidine is formed, it reacts immediately with the surrounding acetic anhydride to form phenacetin and acetic acid. This in-situ acetylation prevents the accumulation of the free amine, which is prone to oxidation and side reactions, thereby enhancing the overall selectivity of the transformation. The use of acetic anhydride as the solvent ensures that the reaction medium is compatible with the subsequent chromatographic separation, avoiding the need for solvent exchange.
Following the reaction, the mixture enters the simulated moving bed (SMB) chromatography system, which is the heart of the purification strategy. Unlike traditional batch chromatography, SMB simulates a counter-current movement of the stationary phase against the mobile phase, allowing for continuous separation. In this specific application, the stationary phase may consist of activated carbon, silica gel, or alumina, while acetic anhydride acts as the mobile phase. The system exploits the differential adsorption affinities of phenacetin, unreacted p-nitrophenetole, and p-phenetidine. Phenacetin, having a distinct polarity and interaction profile, is eluted separately from the starting materials and intermediates. This high-resolution separation ensures that the product stream contains virtually no p-nitrophenetole or p-phenetidine, achieving a purity level exceeding 99%. Moreover, the fraction containing the unreacted materials is not discarded but is instead merged with the recovered solid catalyst and returned to the reactor, maximizing atom economy and process efficiency.
How to Synthesize Para-acetamino phenetole Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters and chromatographic conditions to ensure reproducibility and safety. The process begins with the preparation of a reaction mixture comprising p-nitrophenetole, hydrogen gas, a selected solid catalyst, and acetic anhydride. The reaction is conducted under elevated temperature and pressure to drive the hydrogenation to completion while simultaneously facilitating the acetylation. Once the reaction pressure stabilizes, indicating the consumption of hydrogen, the mixture undergoes solid-liquid separation to recover the catalyst. The liquid phase is then treated to remove the generated acetic acid, typically via distillation or flash evaporation, before entering the SMB unit. The detailed standardized synthesis steps, including specific flow rates, column configurations, and recycling protocols, are outlined in the guide below.
- Prepare a mixture of p-Nitrophenetole, hydrogen, a solid catalyst (Ni, Co, or Pt), and acetic anhydride in a reactor.
- Conduct reduction and acetylation reactions simultaneously at 30-160°C and 0.2-2.2 MPa to generate phenacetin and acetic acid.
- Separate the solid catalyst, remove acetic acid, and purify the phenacetin using simulated moving bed chromatography with acetic anhydride as the eluent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this reactor-coupled SMB technology offers transformative benefits that extend beyond mere technical elegance. The primary advantage lies in the drastic simplification of the downstream processing train. By eliminating the need for neutralization, washing, and multiple crystallization steps, the process significantly reduces the consumption of water, acids, bases, and energy. This reduction in utility usage translates directly into lower operational expenditures. Furthermore, the ability to recycle unreacted raw materials and the catalyst in a closed loop minimizes the purchase volume of expensive starting materials like p-nitrophenetole and acetic anhydride. This inherent efficiency provides a robust buffer against raw material price volatility, ensuring more stable pricing for the final product. The streamlined nature of the process also shortens the manufacturing cycle, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The elimination of aqueous workup steps removes the associated costs of wastewater treatment and salt disposal, which are significant hidden expenses in traditional chemical manufacturing. Additionally, the dual use of acetic anhydride as both reagent and eluent simplifies solvent inventory management and reduces the need for complex solvent recovery systems. The continuous nature of the SMB separation allows for higher throughput with smaller equipment footprints compared to batch crystallization, leading to better capital efficiency. These factors combine to create a leaner production model that inherently drives down the cost per kilogram of the high-purity phenacetin produced.
- Enhanced Supply Chain Reliability: The robustness of this one-pot process reduces the number of potential failure points in the manufacturing line. Traditional multi-step syntheses are vulnerable to delays at each isolation and purification stage; by consolidating these steps, the risk of batch failure is minimized. The recycling of catalysts and raw materials ensures that the process is less dependent on the immediate availability of fresh inputs for every batch, providing a buffer against supply disruptions. This reliability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical clients who require just-in-time inventory management for their own production lines.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent globally, the zero-emission capability of this process is a major strategic asset. The thorough prevention of wastewater and slag generation ensures compliance with even the toughest environmental standards without the need for expensive end-of-pipe treatment facilities. This makes the technology highly scalable, as expanding production capacity does not proportionally increase the environmental burden. The ability to scale from pilot quantities to commercial tons while maintaining a green footprint positions manufacturers as preferred partners for multinational corporations committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clean production technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these details is vital for assessing the feasibility of integrating this process into existing manufacturing frameworks or for evaluating new supply sources.
Q: How does the simulated moving bed technique improve phenacetin purity?
A: The simulated moving bed (SMB) chromatography system allows for the continuous separation of phenacetin from unreacted p-Nitrophenetole and p-phenetidine without requiring neutralization or water washing steps. By using acetic anhydride as the eluent, the system achieves a chromatographic purity of over 99%, effectively removing impurities that traditional crystallization might miss.
Q: What are the environmental benefits of this clean production process?
A: This process eliminates the generation of wastewater and slag entirely. Traditional methods often require neutralization and washing steps that produce significant saline wastewater. By coupling the reactor directly with the separation system and recycling unreacted materials and solvents, this method realizes zero emission of waste water and dregs.
Q: Can the catalyst and raw materials be recycled in this process?
A: Yes, the process is designed for high efficiency recycling. After solid-liquid separation, the solid catalyst is recovered. Furthermore, the fraction containing unreacted p-Nitrophenetole and p-phenetidine from the SMB separation is merged with the catalyst and returned directly to the reactor for the next cycle, minimizing raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Para-acetamino phenetole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical industry. While the technology described in CN101823979B represents a significant leap forward, our team possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to life. Our state-of-the-art facilities are equipped to handle complex catalytic hydrogenations and continuous chromatographic separations, ensuring that we can deliver phenacetin and related intermediates with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify that every batch meets the highest standards of quality, free from the impurities often associated with traditional synthesis routes.
We invite you to collaborate with us to explore how this clean production technology can optimize your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable para-acetamino phenetole supplier dedicated to delivering value through innovation, quality, and sustainability.
