Revolutionizing Ergothioneine Production: Advanced Enzymatic Catalysis for Commercial Scale
Revolutionizing Ergothioneine Production: Advanced Enzymatic Catalysis for Commercial Scale
The global demand for potent antioxidants in the nutraceutical and cosmetic sectors has intensified the search for efficient manufacturing routes for high-value compounds like ergothioneine. A significant technological breakthrough is detailed in patent CN115976129A, which discloses a sophisticated method for preparing ergothioneine utilizing specific biological enzyme catalysis. This innovation addresses the longstanding challenges of low yields and complex purification associated with traditional chemical synthesis. By leveraging a eukaryotic organism synthesis pathway, the technology enables the in vitro enzymatic preparation of ergothioneine under controlled aerobic conditions. The process is characterized by its ability to achieve high substrate concentrations and exceptional conversion rates within a remarkably short timeframe. For industry stakeholders, this represents a pivotal shift towards more sustainable and economically viable production of this critical nutritional ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of ergothioneine has been hindered by the inherent inefficiencies of chemical synthesis and early-stage microbial fermentation techniques. Chemical routes often rely on expensive starting materials and involve multi-step reactions that require harsh conditions, leading to insufficient overall yields and significant environmental burdens. Furthermore, the removal of toxic byproducts and residual solvents from chemically synthesized ergothioneine adds substantial complexity and cost to the downstream processing. While microbial fermentation using metabolically modified engineering bacteria has emerged as an alternative, it frequently suffers from long fermentation cycles, sometimes extending beyond 100 hours, which limits throughput. Additionally, achieving high titers in fermentation often requires complex media optimization and struggles with substrate inhibition, making consistent commercial scale-up difficult to maintain without compromising purity standards.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a streamlined two-step enzymatic cascade that bypasses the metabolic bottlenecks of whole-cell fermentation. This method employs specific Egt1 and Egt2 enzymes screened for high conversion efficiency, facilitating a direct transformation of histidine betaine and L-cysteine into the target molecule. The process operates under mild conditions, specifically between 25-40°C and a neutral to slightly alkaline pH range, which preserves enzyme stability and activity. By conducting the reaction in vitro, manufacturers gain precise control over reaction parameters, eliminating the variability associated with cell growth and metabolism. This results in a simplified process operation that is inherently more suitable for large-scale production, offering a robust solution for reliable ergothioneine supplier networks seeking to optimize their manufacturing footprint.
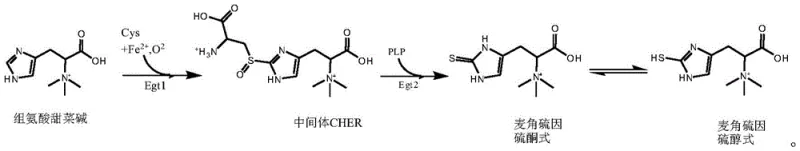
Mechanistic Insights into Egt1 and Egt2 Catalyzed Biosynthesis
The core of this technological advancement lies in the precise mechanistic action of the selected enzymes within a carefully balanced reaction system. The first stage involves the addition of Egt1 enzyme into a mixture of histidine betaine and L-cysteine, facilitated by ferrous ions and a reducing agent. This step catalyzes the formation of the intermediate CHER (hercynylcysteine sulfoxide) with remarkable efficiency. The patent specifies that maintaining an aerobic environment is crucial during this phase, as oxygen acts as a necessary co-substrate for the mononuclear non-heme iron enzyme activity. The reaction is optimized at a temperature of 30°C and a pH of 6.5-7.0, conditions under which the Egt1 enzyme derived from organisms like Chaetomium thermophilum demonstrates conversion rates approaching 99%. This high efficiency minimizes the accumulation of unreacted substrates, thereby simplifying the subsequent purification steps.
Following the formation of the intermediate, the second stage initiates the cleavage of CHER into ergothioneine through the action of the Egt2 enzyme. This step requires the presence of a coenzyme, specifically pyridoxal phosphate (PLP), along with a reducing agent to maintain the redox state of the system. The Egt2 enzyme, sourced from eukaryotes such as Rhodotorula mucilaginosa, operates optimally at a slightly higher pH range of 8.0-8.5 while maintaining the temperature at 30°C. The structural integrity of the final product is maintained through its stable thione form, although it exists in equilibrium with the thiol tautomer as shown in structural analyses. 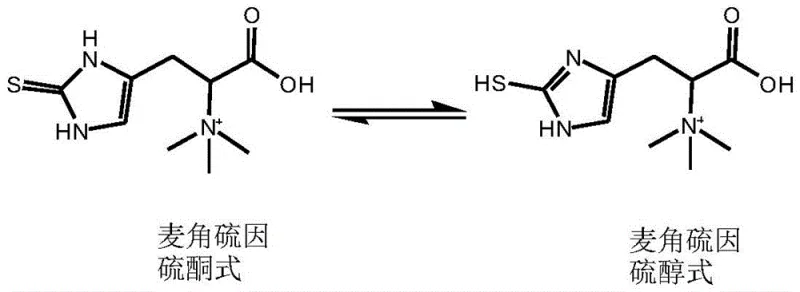 . This mechanistic understanding allows for rigorous impurity control, ensuring that the final product meets the stringent purity specifications required for high-purity nutritional ingredients in sensitive applications like infant formula or premium skincare.
. This mechanistic understanding allows for rigorous impurity control, ensuring that the final product meets the stringent purity specifications required for high-purity nutritional ingredients in sensitive applications like infant formula or premium skincare.
How to Synthesize Ergothioneine Efficiently
Implementing this synthesis route requires a systematic approach to enzyme preparation and reaction control to maximize yield and operational consistency. The process begins with the construction and expression of recombinant E. coli strains harboring the specific Egt1 and Egt2 genes, followed by the preparation of crude enzyme solutions or wet cell masses. These biocatalysts are then introduced sequentially into the reaction vessel containing the substrates and necessary cofactors. Precise monitoring of pH and temperature throughout the cascade is essential to maintain enzyme activity and prevent denaturation. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Prepare the reaction system by mixing histidine betaine and L-cysteine with Egt1 enzyme, ferrous ions, and a reducing agent at pH 6.0-8.0 and 25-40°C to generate the CHER intermediate.
- Introduce Egt2 enzyme along with pyridoxal phosphate coenzyme and reducing agent to the CHER mixture, maintaining pH 6.0-9.0 and 25-40°C.
- Allow the second reaction to proceed until complete conversion is achieved, followed by standard downstream purification to isolate high-purity ergothioneine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of complex chemical synthesis steps and the reduction in fermentation time directly contribute to a more streamlined manufacturing workflow. This efficiency gain allows for a significant reduction in operational expenditures related to energy consumption and facility usage time. Moreover, the high conversion rates mean that less raw material is wasted, optimizing the cost of goods sold without compromising on the quality of the final active ingredient. These factors collectively enhance the competitiveness of the supply chain in the volatile market for specialty fine chemicals.
- Cost Reduction in Manufacturing: The enzymatic process fundamentally alters the cost equation by removing the need for expensive transition metal catalysts and harsh reagents often required in chemical synthesis. By utilizing recombinant enzymes produced in standard E. coli systems, the cost of biocatalyst production is kept low and scalable. The high specificity of the enzymes reduces the formation of side products, which drastically simplifies the downstream purification process and lowers the consumption of chromatography resins and solvents. Consequently, this leads to substantial cost savings in nutritional ingredients manufacturing, allowing for more competitive pricing structures for bulk buyers.
- Enhanced Supply Chain Reliability: Dependence on agricultural extraction or unpredictable fermentation yields poses a risk to supply continuity, which this in vitro enzymatic method effectively mitigates. The ability to produce enzymes independently and store them as stable preparations ensures that production can be ramped up quickly in response to market demand fluctuations. The robust nature of the selected enzymes under the specified reaction conditions reduces the risk of batch failures due to contamination or metabolic shifts common in whole-cell fermentation. This reliability is crucial for reducing lead time for high-purity ergothioneine deliveries to global partners.
- Scalability and Environmental Compliance: The simplicity of the process operation facilitates easy commercial scale-up of complex antioxidants from laboratory benchtop to industrial reactors. The mild reaction conditions reduce the thermal load on cooling systems and minimize the generation of hazardous waste streams associated with traditional chemical methods. This aligns with increasingly strict environmental regulations and corporate sustainability goals, making the supply chain more resilient to regulatory changes. The process is inherently greener, supporting the production of eco-friendly materials that appeal to conscious consumers and brand owners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis technology. They are derived from the specific beneficial effects and technical disclosures found within the patent documentation. Understanding these details is vital for R&D teams evaluating the feasibility of integrating this route into their existing production lines. The answers provide clarity on enzyme performance, reaction conditions, and scalability potential.
Q: What are the key advantages of this enzymatic method over chemical synthesis?
A: This enzymatic approach eliminates the need for expensive starting materials and harsh chemical conditions typical of traditional synthesis. It offers significantly higher conversion rates and operates under mild aerobic conditions, reducing energy consumption and waste generation while simplifying the purification process for pharmaceutical-grade purity.
Q: Which enzymes are critical for the high conversion efficiency described in the patent?
A: The process relies on the synergistic action of Egt1 and Egt2 enzymes derived from specific eukaryotic organisms. The patent highlights enzymes from Chaetomium thermophilum and Rhodotorula mucilaginosa as particularly effective, achieving conversion rates exceeding 99% for the intermediate and final product respectively under optimized pH and temperature conditions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability. The in vitro enzyme catalysis allows for high substrate concentrations and short reaction times, which are critical factors for commercial viability. The use of recombinant E. coli for enzyme expression further ensures a stable and reproducible supply of biocatalysts for continuous manufacturing operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ergothioneine Supplier
The technical potential of this enzymatic route underscores the importance of partnering with a CDMO expert capable of translating complex biocatalytic pathways into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of ergothioneine meets the highest international standards for food and pharmaceutical applications. We understand the critical nature of consistency in the supply of active ingredients for global brands.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your current sourcing strategy. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this enzymatic grade material. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique product requirements. Let us collaborate to secure a sustainable and efficient supply chain for your next-generation formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
