Advanced Synthesis of Beta-Amino Acrylate Derivatives for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Synthesis of Beta-Amino Acrylate Derivatives for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The landscape of modern pharmaceutical synthesis is constantly evolving, driven by the urgent need for more efficient, cost-effective, and environmentally benign pathways to access complex bioactive scaffolds. A pivotal development in this domain is disclosed in patent CN113214112A, which introduces a novel class of beta-amino acrylate substituted acetonitrile compounds. These molecules are not merely academic curiosities; they represent critical building blocks with profound implications for drug discovery, particularly in the realm of antibacterial therapeutics. The structural significance of beta-amino acids cannot be overstated, as they serve as fundamental components in bioactive peptides and small molecule drugs, often acting as precursors to heterocyclic conjugated enamines that are indispensable in life science synthesis.
Furthermore, the integration of the cyano functional group adds a layer of metabolic stability and biocompatibility, enhancing the interaction between drug molecules and biological receptors through polarity and hydrogen bonding. This dual functionality is exemplified by established drugs such as Sitagliptin, Sparfloxacin, and Citalopram, which leverage similar structural motifs to achieve their therapeutic effects against diabetes, bacterial infections, and depression, respectively. The ability to efficiently construct these hybrid scaffolds opens new avenues for developing next-generation antibiotics, addressing the growing global challenge of antimicrobial resistance.
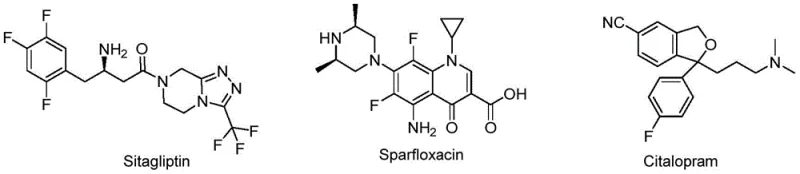
The strategic value of this technology lies in its ability to merge two potent pharmacophores—the beta-amino acid derivative and the acetonitrile group—into a single, versatile intermediate. As illustrated by natural products like Penicillin and Taxol, which contain beta-amino acid skeletons, and Ubenimex, which utilizes nitrile functionality, the convergence of these chemical features suggests a high probability of inheriting robust biological properties. Consequently, the development of an economic and efficient preparation method for these beta-amino acrylate substituted acetonitrile compounds is of paramount importance for the pharmaceutical industry, promising to accelerate the pipeline for novel antibacterial agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex beta-amino acid derivatives and their nitrile-substituted analogues has been fraught with significant challenges that hinder large-scale commercial adoption. Traditional methodologies often rely on multi-step sequences involving harsh reaction conditions, such as extreme temperatures or highly acidic environments, which can compromise the integrity of sensitive functional groups. Moreover, many conventional routes necessitate the use of expensive transition metal catalysts or stoichiometric amounts of hazardous reagents, leading to substantial increases in production costs and generating considerable amounts of toxic waste. The purification of intermediates in these legacy processes is frequently cumbersome, requiring extensive chromatographic separation to remove metal residues and by-products, thereby extending lead times and reducing overall process efficiency.
Another critical bottleneck in prior art is the limited substrate scope, where slight modifications to the starting materials can result in dramatic drops in yield or complete reaction failure. This lack of universality forces process chemists to develop bespoke synthetic routes for each new analogue, stifling innovation and slowing down the structure-activity relationship (SAR) studies essential for drug optimization. Furthermore, the instability of certain beta-amino acid precursors under standard conditions often leads to racemization or decomposition, resulting in poor stereochemical control and reduced purity of the final active pharmaceutical ingredient (API). These cumulative inefficiencies create a substantial barrier to entry for manufacturers seeking to produce high-quality intermediates at a competitive price point.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in patent CN113214112A presents a streamlined, one-pot synthetic strategy that fundamentally reshapes the production landscape for these valuable intermediates. By employing a base-catalyzed condensation between a cyano compound (Formula II) and a 3-(phenethylimino)-2-aryl-acrylate compound (Formula III), this novel approach achieves high yields under remarkably mild conditions. The reaction proceeds smoothly at temperatures ranging from 30°C to 120°C, eliminating the need for energy-intensive heating or cryogenic cooling systems that typically inflate operational expenditures. The simplicity of the operation, which often involves merely mixing the reagents and stirring under a nitrogen atmosphere, significantly reduces the technical expertise required for execution, making it highly amenable to automation and scale-up.
Moreover, the versatility of this method is evidenced by its wide substrate universality, accommodating a diverse array of substituents on both the aryl formyl and alkyl groups without compromising reaction efficiency. This flexibility allows medicinal chemists to rapidly generate libraries of analogues for biological screening, accelerating the identification of lead candidates. The use of readily available and inexpensive catalysts, such as DBU, potassium carbonate, or triethylamine, further underscores the economic viability of this process, removing the dependency on scarce precious metals. Ultimately, this novel approach delivers a robust, scalable, and cost-effective solution that directly addresses the pain points of traditional synthesis, positioning it as a superior choice for industrial manufacturing.
Mechanistic Insights into Base-Catalyzed Condensation
The core of this technological breakthrough lies in the elegant mechanistic pathway facilitated by the chosen base catalysts. The reaction initiates with the deprotonation of the active methylene group in the cyano compound (Formula II) by the base, generating a stabilized carbanion nucleophile. This nucleophilic species then undergoes a Michael-type addition to the electron-deficient alkene moiety of the imino-acrylate compound (Formula III). The presence of the electron-withdrawing ester and imine groups on the acrylate significantly enhances the electrophilicity of the beta-carbon, driving the addition forward with high regioselectivity. Following the initial addition, a cascade of intramolecular rearrangements or proton transfers occurs, ultimately leading to the formation of the stable beta-amino acrylate substituted acetonitrile skeleton (Formula I).
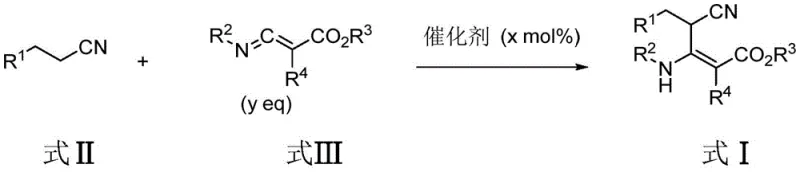
This mechanism is particularly advantageous because it avoids the formation of unstable intermediates that often plague other synthetic routes. The mild basic conditions ensure that sensitive functional groups, such as esters and nitriles, remain intact throughout the transformation, preserving the chemical integrity of the molecule. Furthermore, the choice of solvent plays a crucial role in stabilizing the transition states and solubilizing the ionic intermediates, with polar aprotic solvents like acetonitrile or DMF often yielding optimal results. The reaction's tolerance to various substituents suggests that the electronic effects of the aryl groups are well-managed by the catalytic system, preventing side reactions such as polymerization or hydrolysis. This precise control over the reaction pathway is key to achieving the high purity levels required for pharmaceutical applications.
From an impurity control perspective, the mechanism inherently minimizes the generation of difficult-to-remove by-products. Since the reaction is highly atom-economical and proceeds through a concerted pathway, the formation of oligomeric impurities is suppressed. The use of nitrogen protection further prevents oxidative degradation of the reactants or products, ensuring a clean reaction profile. In cases where stereocenters are generated, the specific choice of chiral catalysts or auxiliaries could potentially be integrated into this framework to induce asymmetry, although the current patent focuses on the racemic or diastereomeric mixtures suitable for initial screening. The robustness of this mechanistic design ensures that the process remains consistent and reproducible, a critical factor for maintaining quality standards in GMP manufacturing environments.
How to Synthesize Beta-Amino Acrylate Substituted Acetonitrile Compounds Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to a standardized protocol that maximizes yield while ensuring safety and reproducibility. The process begins with the careful selection of reagents, ensuring that the cyano compound and the imino-acrylate precursor meet specified purity criteria to prevent catalyst poisoning. The reaction is typically conducted under an inert atmosphere to exclude moisture and oxygen, which could interfere with the base catalyst or degrade the sensitive intermediates. While the general procedure is straightforward, attention to detail regarding stoichiometry and temperature control is essential to replicate the high efficiencies reported in the patent examples.
- Under nitrogen protection, uniformly mix a selected base catalyst (such as DBU, K2CO3, or Et3N), a cyano compound (Formula II), a 3-(phenethylimino)-2-aryl-acrylate compound (Formula III), and an appropriate organic solvent.
- Stir the reaction mixture at a controlled temperature ranging from -60°C to 150°C, preferably between 30°C and 120°C, monitoring progress via TLC until the starting materials are substantially consumed.
- Upon completion, separate the product (Formula I) through standard workup procedures such as direct column chromatography using petroleum ether and ethyl acetate eluents to obtain the high-purity white solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates into tangible strategic benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike processes that rely on exotic ligands or proprietary metal catalysts, this method utilizes commodity chemicals such as potassium carbonate, triethylamine, and DBU, which are available in bulk quantities from multiple global suppliers. This abundance mitigates the risk of supply disruptions and price volatility, ensuring a steady flow of materials necessary for continuous production schedules. Additionally, the elimination of heavy metal catalysts removes the need for expensive and time-consuming metal scavenging steps, significantly reducing the cost of goods sold (COGS) and simplifying the waste management workflow.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the replacement of costly reagents with inexpensive alternatives. By utilizing simple organic or inorganic bases instead of precious metal complexes, manufacturers can achieve substantial cost savings on catalyst procurement. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to lower utility bills. The high yields and selectivity observed in this process minimize the loss of valuable starting materials, enhancing the overall material efficiency. Consequently, the total cost of producing high-purity pharmaceutical intermediates is significantly lowered, improving profit margins and competitiveness in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route directly enhances supply chain resilience. Because the reaction tolerates a wide range of substrates and conditions, it is less susceptible to batch-to-batch variations caused by minor fluctuations in raw material quality. This consistency reduces the frequency of failed batches and the need for re-processing, ensuring reliable delivery timelines to downstream customers. The simplicity of the workup procedure, often involving direct column chromatography or crystallization, shortens the production cycle time, allowing for faster turnaround from order to shipment. This agility is crucial for meeting the dynamic demands of the pharmaceutical industry, where speed to market can be a decisive competitive advantage.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by its inherent safety and simplicity. The absence of pyrophoric reagents or high-pressure requirements makes it easier to adapt to large-scale reactors without significant engineering modifications. From an environmental perspective, the use of greener solvents and the reduction of hazardous waste align with increasingly stringent regulatory standards for chemical manufacturing. The lower E-factor (mass of waste per mass of product) associated with this high-yielding process supports corporate sustainability goals and reduces the burden of waste disposal. This alignment with green chemistry principles not only ensures compliance but also enhances the brand reputation of the manufacturer as a responsible and forward-thinking partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and potential of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: The method described in patent CN113214112A utilizes cheap and easily obtainable raw materials and catalysts. It operates under mild reaction conditions with simple operation, offering wide substrate universality compared to harsher traditional methods.
Q: What is the potential pharmaceutical application of these compounds?
A: These beta-amino acrylate substituted acetonitrile compounds exhibit better pharmaceutical activity, specifically showing promise in the preparation of antibacterial drugs, including anti-mycobacteria agents similar to Sparfloxacin.
Q: Which catalysts are compatible with this reaction system?
A: The process supports a wide range of basic catalysts including organic bases like DBU, TBD, and DABCO, as well as inorganic bases like potassium carbonate and potassium tert-butoxide, allowing for flexible process optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Acrylate Supplier
The development of beta-amino acrylate substituted acetonitrile compounds represents a significant leap forward in the synthesis of antibacterial intermediates, offering a blend of efficiency, versatility, and economic viability that is rare in modern organic synthesis. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this technology and are fully equipped to bring it from the patent literature to commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from R&D to full-scale manufacturing. Our state-of-the-art facilities are designed to handle complex chemistries with precision, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee the highest quality standards for every batch produced.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route for your drug development programs. Our team of expert process chemists is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain and a wealth of technical expertise dedicated to accelerating your path to market with cost-effective, high-quality pharmaceutical intermediates.
