Advanced Nickel-Catalyzed Synthesis of Indanone Compounds for Commercial Scale-up
Advanced Nickel-Catalyzed Synthesis of Indanone Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing privileged scaffolds such as indanones. A significant breakthrough in this domain is detailed in Chinese Patent CN113214067B, which discloses a novel preparation method for indanone compounds via a nickel-catalyzed ring-opening and cyclization strategy. This technology addresses critical pain points in synthetic organic chemistry by eliminating the need for external coupling agents, thereby streamlining the production workflow. For R&D directors and procurement managers alike, this innovation represents a pivotal shift towards more sustainable and economically viable manufacturing processes. By leveraging the unique reactivity of o-bromophenyl cyclobutanones under mild nickel catalysis, this method offers a direct pathway to high-purity intermediates essential for drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzene-fused ring compounds like indanones from small-ring precursors has relied heavily on palladium catalysis or required the presence of specific coupling agents to drive the reaction thermodynamics. Previous literature, such as reports involving palladium catalysts, often necessitates the addition of alkynyl groups or alkyl bromides as coupling partners to facilitate the ring-opening and subsequent cyclization. This dependency introduces significant complexity to the reaction system, increasing the number of reagents required and complicating the downstream purification process. The presence of these additional coupling agents often leads to difficult-to-separate byproducts, reducing the overall isolated yield and increasing the cost of goods sold. Furthermore, palladium catalysts are notoriously expensive and subject to supply chain volatility, making them less attractive for large-scale commercial applications where cost reduction in API manufacturing is a primary objective.
The Novel Approach
In stark contrast, the methodology described in patent CN113214067B utilizes a nickel-catalyzed system that operates efficiently without any external coupling agents. The core transformation involves the reaction of an o-bromophenyl cyclobutanone, represented by Formula 1, which undergoes a seamless ring-opening and cyclization to yield the desired indanone structure, Formula I. This process is driven by the release of ring strain from the cyclobutanone moiety, providing the necessary thermodynamic driving force. The use of water as a proton source is particularly ingenious, as it replaces the need for complex hydride donors or organometallic coupling partners. This simplification not only reduces raw material costs but also minimizes waste generation. As illustrated in the general reaction scheme below, the transformation is direct and atom-economical, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their production portfolios.
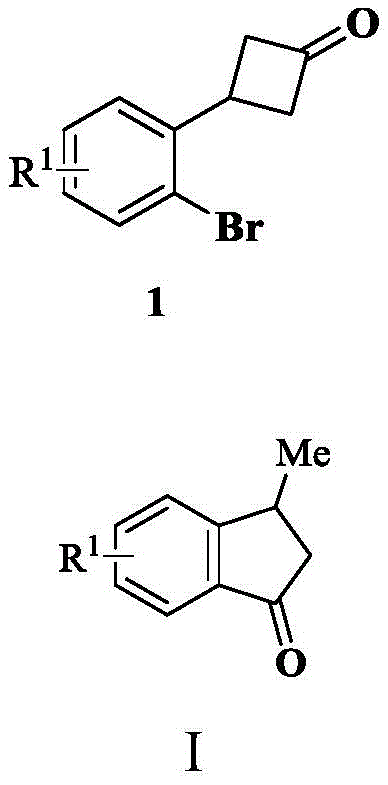
Mechanistic Insights into Nickel-Catalyzed C-C Bond Activation
The success of this transformation lies in the sophisticated catalytic cycle mediated by the nickel species. The reaction initiates with the reduction of the nickel(II) precatalyst to an active nickel(0) species, which subsequently undergoes oxidative addition with the aryl bromide bond of the substrate. This generates a nickel(II) aryl complex that is poised for the critical bond-breaking step. The bis(pinacolato)diboron reagent plays a dual role here; it acts as a reducing agent to convert the nickel(II) intermediate into a reactive nickel(I) species. This low-valent nickel(I) intermediate is capable of selectively inserting into one of the strained C(sp2)-C(sp3) sigma bonds of the cyclobutanone ring. This insertion event is the key to unlocking the ring strain and forming the new carbon-carbon bond required for the five-membered indanone scaffold. The resulting bicyclic nickel(III) complex then undergoes reductive elimination to forge the final C-C bond, releasing the indanone framework coordinated to an alkylnickel(I) species.
Following the reductive elimination, the catalytic cycle is completed by the protonation of the alkylnickel(I) species by water, which serves as the terminal proton source. This step releases the final indanone product and regenerates the active nickel catalyst, allowing the cycle to continue. This mechanism highlights the exquisite control exerted by the ligand environment, specifically when using ligands like Neocuproine, which stabilizes the various oxidation states of nickel throughout the cycle. Understanding this mechanistic pathway is crucial for R&D teams aiming to adapt this chemistry for diverse substrates. The ability to tolerate various substituents on the aromatic ring, such as methyl, methoxy, or trifluoromethyl groups, demonstrates the robustness of the catalytic system. For instance, the specific synthesis of 3,5-dimethyl-2,3-dihydro-1H-inden-1-one shown below exemplifies how the general mechanism translates into a concrete, high-yielding synthetic route for specific target molecules.
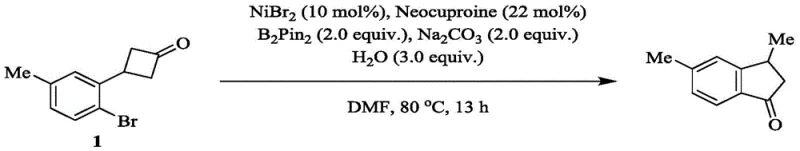
How to Synthesize 3,5-Dimethyl-2,3-dihydro-1H-inden-1-one Efficiently
Implementing this nickel-catalyzed protocol in a laboratory or pilot plant setting requires strict adherence to the optimized conditions identified in the patent data to ensure reproducibility and high purity. The process begins with the careful weighing and loading of the solid reagents, including the nickel catalyst, ligand, base, and boron reagent, into a reaction vessel under an inert atmosphere to prevent catalyst deactivation by oxygen. The substrate and water are then dissolved in the polar aprotic solvent, typically DMF, and introduced to the reaction mixture. Maintaining the correct stoichiometry is vital; for example, using 2.0 equivalents of the boron reagent and 3.0 equivalents of water has been shown to maximize yield while maintaining economic efficiency. The reaction is then heated to a moderate temperature range of 70°C to 90°C, with 80°C being the optimal setpoint, and stirred for a duration of 12 to 24 hours. Detailed standardized synthesis steps for this specific transformation are provided in the guide below.
- Combine 3-(2-bromo-5-methylphenyl)cyclobutan-1-one, NiBr2 catalyst, Neocuproine ligand, B2Pin2, and Na2CO3 in a sealed tube under argon protection.
- Dissolve the substrate and water in DMF solvent, add to the reaction vessel, and seal tightly to maintain an inert atmosphere.
- Heat the mixture to 80°C for 13 hours with stirring, then perform aqueous workup with dilute HCl and ethyl acetate extraction followed by column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed methodology offers tangible strategic advantages that extend beyond mere chemical novelty. The primary benefit is the substantial cost savings achieved by replacing expensive palladium catalysts with abundant and affordable nickel salts. Additionally, the elimination of external coupling agents reduces the bill of materials and simplifies the inventory management of hazardous or sensitive reagents. From a supply chain reliability perspective, the use of commodity chemicals like sodium carbonate and DMF ensures that production is not bottlenecked by the availability of specialized reagents. The simplified workup procedure, which avoids complex separations of coupling byproducts, translates directly into reduced processing time and lower energy consumption during the purification phase. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition from palladium to nickel catalysis inherently lowers the raw material costs associated with the precious metal catalyst. Furthermore, by removing the requirement for additional coupling agents such as alkynes or alkyl halides, the process reduces the total mass of reagents purchased per kilogram of product. This reduction in material intensity, combined with the simplified purification workflow that avoids difficult chromatographic separations of side products, leads to significant operational expenditure savings. The overall process efficiency is enhanced, allowing for better resource utilization and a lower cost of goods sold, which is critical for maintaining competitiveness in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable reagents such as NiBr2, Neocuproine, and sodium carbonate mitigates the risk of supply disruptions often associated with exotic or custom-synthesized coupling agents. Water, serving as the proton source, is universally accessible and eliminates the need for sourcing specialized hydride donors. This robustness ensures that production schedules can be maintained consistently without delays caused by reagent shortages. For supply chain planners, this means greater predictability in lead times and the ability to scale production volumes confidently to meet market demand without compromising on the continuity of supply for critical drug intermediates.
- Scalability and Environmental Compliance: The reaction conditions are mild, operating at temperatures between 70°C and 90°C, which are easily achievable in standard stainless steel reactors without requiring extreme heating or cooling capabilities. The use of DMF as a solvent is well-established in industrial settings, and the waste stream is simplified due to the absence of coupling agent byproducts. This facilitates easier waste treatment and compliance with environmental regulations regarding hazardous waste disposal. The process is inherently safer and more amenable to commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to increase batch sizes from kilograms to tons while maintaining strict control over product quality and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential licensees and manufacturing partners. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines. The responses cover aspects ranging from substrate tolerance to purification strategies, ensuring that stakeholders have a comprehensive understanding of the process capabilities and limitations before committing to scale-up efforts.
Q: What is the primary advantage of this nickel-catalyzed method over traditional palladium routes?
A: Unlike traditional palladium-catalyzed methods that require additional coupling agents like alkynes or alkyl bromides, this nickel-catalyzed protocol utilizes water as the proton source. This eliminates the need for extra coupling reagents, significantly simplifying the reaction mixture and making product isolation and purification much easier and more cost-effective.
Q: What represents the optimal reaction conditions for maximizing yield?
A: According to the patent data, the optimal conditions involve using NiBr2 as the catalyst and Neocuproine as the ligand in DMF solvent. The reaction should be conducted at a temperature of 80°C for approximately 13 hours. Sodium carbonate serves as the preferred base, and using 2.0 equivalents of bis(pinacolato)diboron provides the best balance of efficiency and economy.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is highly suitable for scale-up. It operates at moderate temperatures (70-90°C) and uses commercially available, stable reagents. The absence of sensitive coupling agents and the use of standard solvents like DMF facilitate safe handling in large reactors, ensuring consistent quality for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the nickel-mediated ring-opening cyclization described in CN113214067B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle nickel catalysis with rigorous safety protocols, and our stringent purity specifications guarantee that every batch of indanone intermediates meets the highest quality standards required by global regulatory bodies. Our rigorous QC labs employ advanced analytical techniques to verify the structural integrity and purity profile of the final product, ensuring consistency and reliability for your drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this nickel-catalyzed route can optimize your manufacturing budget. We encourage you to contact our technical procurement team today to request specific COA data for our catalog intermediates or to discuss route feasibility assessments for your proprietary compounds. Together, we can accelerate your path to market with efficient, scalable, and cost-effective chemical solutions.
