Advanced Synthesis and Commercial Scale-Up of Neuroprotective Piperidine Derivatives for Global Pharmaceutical Supply Chains
Advanced Synthesis and Commercial Scale-Up of Neuroprotective Piperidine Derivatives for Global Pharmaceutical Supply Chains
The global pharmaceutical landscape is increasingly focused on developing effective treatments for neurodegenerative disorders, particularly Alzheimer's disease, where current therapeutic options remain limited in efficacy and duration of action. Patent CN1024547C introduces a novel class of cyclic amine compounds, specifically piperidine derivatives linked to indanone moieties, which demonstrate potent and selective acetylcholinesterase inhibitory activity. These compounds represent a significant advancement over earlier generations of cholinergic drugs by offering improved safety profiles and prolonged pharmacological effects. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural versatility of these molecules is crucial for integrating them into modern drug development pipelines. The core innovation lies in the specific substitution patterns on the indanone ring and the linker chemistry connecting it to the piperidine nitrogen, which collectively enhance blood-brain barrier permeability and metabolic stability.
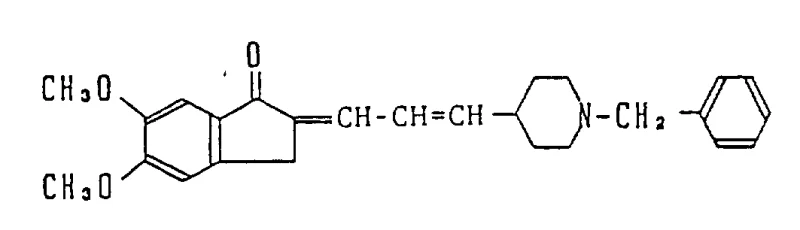
The structural complexity of these pharmaceutical intermediates requires a robust and reproducible synthetic strategy to ensure consistent quality at scale. The patent outlines multiple pathways, with the Wittig-Horner reaction emerging as a pivotal step for constructing the carbon-carbon double bond between the indanone core and the piperidine side chain. This methodology allows for precise control over the geometry of the intermediate olefin, which is subsequently reduced to the saturated alkyl linker found in the most biologically active analogs. From a supply chain perspective, the reliance on well-established organic transformations such as acylation, olefination, and catalytic hydrogenation minimizes the risk of process failures during technology transfer. Furthermore, the ability to introduce various substituents on the aromatic rings provides a flexible platform for generating diverse libraries of analogs, facilitating structure-activity relationship studies without necessitating entirely new process development efforts for each derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing acetylcholinesterase inhibitors often relied on naturally derived alkaloids or simpler synthetic scaffolds that suffered from significant pharmacokinetic drawbacks. Compounds like physostigmine and tacrine, while historically important, are associated with short half-lives, hepatotoxicity, and narrow therapeutic windows, which complicate dosing regimens and patient compliance. Chemically, older synthetic routes frequently involved harsh reaction conditions, toxic reagents, or multi-step sequences with poor overall yields, making them economically unviable for large-scale commercial production. The purification of these legacy compounds often required extensive chromatography, driving up manufacturing costs and generating substantial chemical waste. Additionally, the lack of structural modularity in conventional methods limited the ability of medicinal chemists to optimize the molecule for specific receptor subtypes or to improve metabolic stability against hepatic enzymes. These limitations created a bottleneck in the development of next-generation dementia therapies, necessitating a shift towards more efficient and tunable synthetic architectures.
The Novel Approach
The methodology described in the patent addresses these historical challenges by employing a convergent synthesis strategy that couples two distinct, readily available fragments: a functionalized indanone and a substituted piperidine. This modular approach not only simplifies the overall synthetic tree but also allows for independent optimization of each fragment before the final coupling step. By utilizing phosphonate esters derived from indanones, the process leverages the Horner-Wadsworth-Emmons modification of the Wittig reaction, which typically offers better E/Z selectivity and easier removal of byproducts compared to traditional phosphonium ylides. The subsequent catalytic reduction step is highly efficient, using standard palladium-on-carbon catalysts under mild hydrogen pressure to saturate the double bond without affecting other sensitive functional groups. This streamlined pathway results in higher overall yields and reduced solvent consumption, directly translating to lower cost of goods sold and a smaller environmental footprint. The flexibility of this route enables the rapid production of various salts and derivatives, ensuring that the final drug substance meets stringent regulatory specifications for purity and impurity profiles.
Mechanistic Insights into Indanone-Piperidine Coupling and Reduction
The chemical mechanism underpinning the formation of the key carbon-carbon bond involves the deprotonation of a phosphonate ester to generate a nucleophilic carbanion, which then attacks the carbonyl carbon of a piperidine aldehyde. This addition-elimination sequence proceeds through a betaine intermediate that collapses to release a phosphate byproduct and form the desired olefin. The choice of base, such as sodium hydride or sodium methoxide, and the solvent system, typically tetrahydrofuran or dimethylformamide, plays a critical role in driving the equilibrium towards product formation while minimizing side reactions like self-condensation of the aldehyde. Following olefination, the catalytic hydrogenation step requires careful monitoring to ensure complete reduction of the alkene without over-reduction of the ketone moiety on the indanone ring, which could lead to inactive alcohol impurities. The use of selective catalysts and controlled reaction temperatures ensures that the stereochemical integrity of the molecule is maintained, particularly when chiral centers are present in the starting materials or generated during the reduction. Understanding these mechanistic nuances is essential for process chemists to establish robust control strategies that guarantee batch-to-batch consistency and compliance with international pharmacopoeia standards.
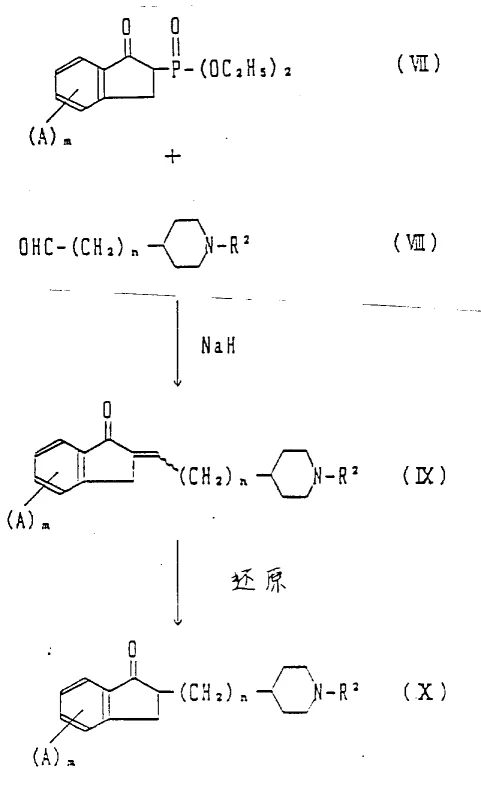
Impurity control is another critical aspect of the mechanistic profile, as the presence of residual phosphorus species or unreduced olefins can impact the safety and efficacy of the final pharmaceutical product. The synthetic route is designed to facilitate the removal of these impurities through standard workup procedures, such as aqueous washes to remove phosphate salts and crystallization to isolate the pure hydrochloride salt of the target amine. The stability of the indanone ketone under the reaction conditions is paramount, as any degradation or side reaction at this position would compromise the biological activity of the compound. Detailed kinetic studies and in-process controls are therefore implemented to monitor the progress of both the olefination and reduction steps, ensuring that reaction endpoints are reached precisely to maximize yield and minimize the formation of difficult-to-remove byproducts. This rigorous approach to mechanism-based process design underscores the commitment to delivering high-quality intermediates that support the development of safe and effective medicines for patients suffering from cognitive decline.
How to Synthesize 1-Benzyl-4-[(5,6-dimethoxy-1-indanone)-2-yl]methylpiperidine Efficiently
The synthesis of this specific high-value intermediate serves as a representative example of the broader platform technology described in the patent, demonstrating the practical application of the Wittig-reduction sequence. The process begins with the preparation of the phosphonate precursor from 5,6-dimethoxy-1-indanone, followed by coupling with 1-benzyl-4-piperidinecarbaldehyde under anhydrous conditions to prevent hydrolysis of the reactive intermediates. Strict control of moisture and oxygen levels is maintained throughout the reaction to ensure optimal performance of the strong bases used in the olefination step. After the formation of the enone intermediate, the mixture is subjected to catalytic hydrogenation, where parameters such as hydrogen pressure, catalyst loading, and agitation speed are optimized to achieve complete conversion within a reasonable timeframe. The final isolation involves salt formation with hydrochloric acid, which not only stabilizes the amine but also facilitates purification through recrystallization from suitable solvent systems like methanol or isopropanol. Detailed standardized synthesis steps see the guide below.
- Preparation of phosphonate intermediates from substituted indanones using standard phosphorylation techniques.
- Execution of Wittig-Horner olefination with piperidine aldehydes under controlled basic conditions to form the enone linkage.
- Catalytic hydrogenation of the olefinic bond using palladium-carbon to yield the saturated target pharmaceutical intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and sourcing reliability. The starting materials, including substituted indanones and piperidine derivatives, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency and price volatility. The elimination of exotic reagents or specialized catalysts further simplifies the procurement process, allowing for competitive bidding and long-term supply agreements that lock in favorable pricing structures. Moreover, the high atom economy of the Wittig-Horner reaction compared to alternative coupling methods means less waste generation and lower disposal costs, contributing to a more sustainable and cost-effective manufacturing operation. The scalability of the process has been demonstrated through successful execution from gram to kilogram scales, indicating a clear path to multi-ton commercial production without the need for significant process re-engineering. This readiness for scale-up ensures that pharmaceutical partners can secure a continuous supply of critical intermediates to meet market demand for finished dosage forms.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of expensive transition metal catalysts often required in cross-coupling reactions, relying instead on more economical base-mediated olefination and standard hydrogenation techniques. This shift significantly lowers the direct material costs associated with catalyst procurement and recovery. Additionally, the high yields achieved in each step reduce the amount of raw material required per unit of product, effectively amplifying the purchasing power of the procurement budget. The simplified purification protocol minimizes the consumption of chromatography media and solvents, which are often major cost drivers in fine chemical manufacturing. By streamlining the process flow, manufacturers can achieve substantial operational savings that can be passed down the supply chain or reinvested into further process optimization initiatives.
- Enhanced Supply Chain Reliability: Sourcing strategies are bolstered by the use of widely available building blocks that are not subject to geopolitical restrictions or complex regulatory controls. The robustness of the chemical transformations ensures that production schedules are less likely to be disrupted by batch failures or quality deviations, providing a predictable output rate for downstream planning. The ability to store stable intermediates allows for the creation of strategic inventory buffers, mitigating the impact of potential logistics delays or raw material shortages. Furthermore, the compatibility of the process with standard stainless steel reactor equipment means that production can be easily transferred between different manufacturing sites if necessary, enhancing the overall resilience of the supply network against unforeseen disruptions.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with reaction conditions that can be safely managed in large vessels without exothermic runaways or hazardous pressure buildups. Waste streams are primarily composed of aqueous salts and organic solvents that can be treated using conventional effluent management systems, ensuring compliance with strict environmental regulations. The reduction in hazardous waste generation aligns with corporate sustainability goals and reduces the liability associated with chemical disposal. As production volumes increase, the efficiency of the process improves due to economies of scale, further lowering the environmental footprint per kilogram of product. This alignment of economic and environmental objectives makes the technology attractive for long-term investment and partnership in the evolving landscape of green chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these cyclic amine compounds, based on the detailed specifications and experimental data provided in the patent documentation. These insights are intended to clarify the feasibility of implementation and the strategic value of integrating this technology into existing pharmaceutical portfolios. Understanding these aspects helps stakeholders make informed decisions about resource allocation and partnership opportunities. The answers reflect a commitment to transparency and technical excellence, ensuring that all parties have a clear understanding of the capabilities and limitations of the manufacturing process.
Q: What are the primary purity challenges in synthesizing indanone-piperidine derivatives?
A: The main challenges involve controlling stereoisomers during the reduction phase and ensuring complete removal of phosphorus byproducts from the Wittig reaction step.
Q: How does this synthetic route impact overall production costs compared to traditional methods?
A: By utilizing commercially available starting materials like substituted indanones and avoiding complex chiral resolution steps early in the sequence, the process significantly reduces raw material expenses and processing time.
Q: Is the manufacturing process scalable for commercial API production?
A: Yes, the reaction conditions utilize standard solvents and catalysts suitable for large-scale reactor operations, ensuring robust supply continuity for downstream drug manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Benzyl-4-[(5,6-dimethoxy-1-indanone)-2-yl]methylpiperidine Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure required to bring complex synthetic routes like this from the laboratory bench to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to quality assurance extends beyond mere compliance, as we proactively identify and mitigate potential risks in the supply chain to guarantee uninterrupted delivery. By leveraging our deep understanding of organic synthesis and process engineering, we help our clients accelerate their drug development timelines and reduce time-to-market for critical therapies.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project requirements. Request a Customized Cost-Saving Analysis to understand how optimizing this synthetic route can improve your margin structure. We are ready to provide specific COA data and route feasibility assessments to support your due diligence process. Partnering with us means gaining access to a reliable source of high-quality pharmaceutical intermediates backed by a track record of success in the global market. Let us help you navigate the complexities of chemical manufacturing so you can focus on delivering life-changing medicines to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
