Advanced Long-Acting GLP-1 Analogues: Microwave-Assisted Synthesis for Commercial Scale
The global pharmaceutical landscape is currently witnessing a transformative shift in the management of type 2 diabetes, driven by the urgent need for therapies that offer sustained efficacy without the risks associated with traditional insulin administration. Patent CN103087175A introduces a groundbreaking class of novel long-acting glucagon-like peptide-1 (GLP-1) analogues that address the critical limitations of native GLP-1, specifically its rapid degradation by dipeptidyl peptidase IV (DPP-IV) and quick renal clearance. This proprietary technology leverages a sophisticated microwave-promoted solid-phase synthesis strategy to efficiently construct these complex polypeptide chains, ensuring high purity and structural integrity. By strategically modifying amino acid residues at positions 17, 26, 34, and 37, the invention achieves a plasma half-life extension from mere minutes to over 35 hours, representing a significant leap forward in therapeutic durability. For industry stakeholders, this patent data underscores a viable pathway for developing robust, long-acting antidiabetic agents that can be manufactured with enhanced process efficiency. The integration of microwave technology into the synthesis workflow not only accelerates production but also optimizes reagent usage, aligning perfectly with the demands of modern, cost-conscious pharmaceutical manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solid-phase peptide synthesis (SPPS) methods have long been plagued by inefficiencies that hinder large-scale commercial viability, particularly when dealing with long and complex peptide sequences like GLP-1. In conventional protocols, the coupling of a single amino acid to the growing resin-bound chain often requires extensive reaction times ranging from 2 hours to 20 hours, creating a significant bottleneck in production throughput. Furthermore, to drive these sluggish reactions to completion, conventional methods typically necessitate a substantial excess of protected amino acids, often ranging from 4 to 5-fold molar excess, which drastically inflates raw material costs and generates excessive chemical waste. The deprotection steps, particularly the removal of the Fmoc group, are similarly time-consuming, frequently taking between 30 minutes to 1 hour per cycle, further extending the overall synthesis timeline. These prolonged exposure times to reaction conditions can also lead to increased side reactions and racemization, resulting in lower crude purity that complicates downstream purification processes. Consequently, the economic and operational burdens of conventional SPPS make it less attractive for the high-volume manufacturing required to meet the growing global demand for peptide-based therapeutics.
The Novel Approach
The innovative approach detailed in the patent data revolutionizes this landscape by integrating microwave irradiation into the Fmoc/tBu orthogonal protection solid-phase synthesis strategy, fundamentally altering the kinetics of peptide bond formation. By applying controlled microwave energy, the coupling reaction time is drastically reduced to an average of merely 10 minutes per amino acid, representing a magnitude of improvement over traditional thermal methods. Similarly, the microwave-promoted removal of Fmoc protecting groups is accomplished in approximately 5 minutes, slashing the cycle time for each elongation step and enabling the rapid assembly of the full peptide sequence. This acceleration is not achieved at the expense of quality; on the contrary, the enhanced reaction kinetics lead to a crude product purity exceeding 85%, which significantly simplifies the subsequent purification workload. Moreover, the efficiency of the microwave-assisted coupling allows for a reduction in the required excess of protected amino acids to just 2-fold, delivering substantial savings in material costs and reducing the environmental footprint of the synthesis process. This method is inherently more amenable to automation and scale-up, providing a robust framework for the industrial production of high-value pharmaceutical intermediates.
Mechanistic Insights into Side-Chain Modified Cysteine Stabilization
The core scientific breakthrough of this technology lies in the strategic chemical modification of the GLP-1 peptide chain to enhance its metabolic stability and pharmacokinetic profile. Native GLP-1 is highly susceptible to enzymatic degradation by DPP-IV, which cleaves the N-terminal dipeptide, rendering the molecule biologically inactive within minutes of administration. To counteract this, the patent describes the introduction of small molecular groups via side-chain modified cysteine residues, which serve as anchor points for functional moieties that confer resistance to enzymatic hydrolysis. These modifications are designed to increase the binding affinity of the peptide to serum albumin, effectively creating a circulating reservoir that protects the molecule from rapid renal filtration and metabolic clearance. The structural integrity of these analogues is maintained through precise microwave-assisted coupling of the modified cysteine building blocks, ensuring that the delicate peptide backbone remains intact during the synthesis process. This dual mechanism of action, combining enzymatic resistance with albumin binding, results in a plasma half-life that exceeds 35 hours, a dramatic improvement over the 2-minute half-life of the native peptide.
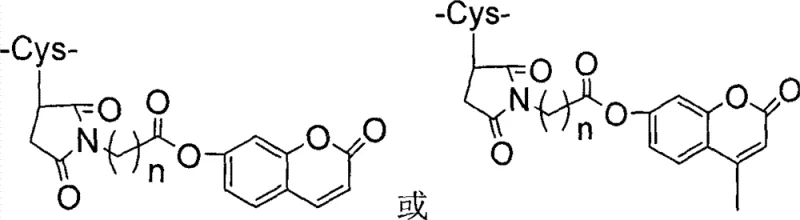
Beyond merely extending half-life, the specific arrangement of amino acid substitutions at positions 17, 26, 34, and 37 is critical for maintaining high receptor binding affinity while preventing degradation. The modified cysteine residues, linked to groups such as maleimide-coumarin derivatives as shown in the structural data, provide a stable covalent attachment that withstands physiological conditions. This structural robustness ensures that the analogue retains its glucose-dependent insulinotropic activity over an extended period, allowing for less frequent dosing regimens which improve patient compliance. The synthesis of these complex structures requires high-fidelity coupling conditions to avoid epimerization or deletion sequences, which is where the microwave-promoted method proves superior by ensuring rapid and complete reactions. The resulting impurity profile is significantly cleaner, with the crude product containing fewer deletion peptides and side products compared to conventional synthesis. This high level of control over the molecular architecture is essential for meeting the stringent regulatory requirements for pharmaceutical active ingredients, ensuring that the final drug product is both safe and efficacious for long-term diabetes management.
How to Synthesize Long-Acting GLP-1 Analogues Efficiently
The synthesis of these advanced GLP-1 analogues follows a rigorous, standardized protocol that leverages microwave technology to achieve high yields and purity. The process begins with the swelling of the Fmoc-Rink amide-MBHA resin, followed by the microwave-assisted removal of the initial Fmoc protecting group to expose the reactive amine. Subsequent cycles involve the coupling of Fmoc-protected amino acids and the specific small molecule modified cysteine building blocks using activating agents like HBTU and HOBT in NMP solvent. Each coupling and deprotection step is optimized with specific microwave power and temperature settings to maximize efficiency while minimizing side reactions. The detailed standardized synthesis steps see the guide below for the complete operational parameters.
- Swelling of Fmoc-Rink amide-MBHA Resin and microwave-promoted removal of the initial Fmoc protecting group using piperidine/NMP solution.
- Microwave-assisted coupling of Fmoc-protected amino acids and small molecule modified cysteine using HBTU/HOBT/DIPEA in NMP.
- Cleavage of the polypeptide from the resin using Reagent K, followed by precipitation in ether and purification via preparative HPLC.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microwave-promoted synthesis technology offers compelling advantages that directly impact the bottom line and operational resilience. The primary benefit stems from the drastic reduction in reaction times and reagent consumption, which translates into a more streamlined and cost-effective manufacturing process. By reducing the excess of protected amino acids from the conventional 4-5 fold to just 2 fold, the material costs associated with raw materials are significantly lowered, providing a clear pathway for cost reduction in API manufacturing. Furthermore, the shortened synthesis cycle allows for higher throughput within the same facility footprint, enabling suppliers to respond more rapidly to market demand fluctuations without the need for massive capital expansion. This efficiency also reduces the energy consumption per batch, aligning with sustainability goals and potentially lowering utility costs. The high crude purity achieved minimizes the burden on purification resources, reducing solvent usage and waste disposal costs, which are often significant hidden expenses in peptide production.
- Cost Reduction in Manufacturing: The implementation of microwave-assisted coupling drastically reduces the consumption of expensive protected amino acids by lowering the required molar excess from 4-5 fold to approximately 2 fold. This optimization directly lowers the raw material cost per gram of the final product, offering substantial cost savings without compromising quality. Additionally, the reduced reaction times lead to lower energy consumption and increased equipment utilization rates, further driving down the overall cost of goods sold. The elimination of prolonged reaction steps also minimizes the risk of batch failures due to equipment downtime or operator error, ensuring a more predictable and economical production environment.
- Enhanced Supply Chain Reliability: The accelerated synthesis cycle significantly reduces the lead time for high-purity pharmaceutical intermediates, allowing for faster turnaround from order to delivery. This agility is crucial for maintaining supply continuity in the face of fluctuating market demands or unexpected disruptions in the raw material supply chain. The robustness of the microwave method ensures consistent batch-to-batch quality, reducing the risk of supply shortages caused by failed quality control tests. By enabling faster production speeds, suppliers can maintain lower inventory levels while still meeting customer deadlines, optimizing working capital and reducing storage costs. This reliability makes the technology an attractive option for long-term supply agreements with major pharmaceutical companies.
- Scalability and Environmental Compliance: The microwave-promoted solid-phase synthesis method is inherently designed for automation and large-scale implementation, making it highly suitable for commercial scale-up of complex polymer additives and peptides. The reduced use of solvents and reagents aligns with green chemistry principles, minimizing the generation of hazardous waste and simplifying compliance with environmental regulations. The high efficiency of the process reduces the overall environmental footprint of the manufacturing operation, which is increasingly important for companies aiming to meet sustainability targets. The ability to scale this technology without losing efficiency or purity ensures that supply can grow in tandem with market demand, supporting the long-term commercial viability of the GLP-1 analogues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these long-acting GLP-1 analogues. The answers are derived directly from the patent data and technical specifications to provide accurate and reliable information for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing supply chain or R&D pipeline. The responses cover aspects of stability, synthesis efficiency, and scalability to ensure a comprehensive understanding of the value proposition.
Q: How does the chemical modification improve the stability of GLP-1 analogues?
A: The analogues are modified with small molecular groups at specific cysteine residues, which increases binding to serum albumin. This modification prevents rapid renal filtration and protects the N-terminus from DPP-IV enzymatic degradation, extending the plasma half-life to over 35 hours.
Q: What are the technical advantages of using microwave promotion in peptide synthesis?
A: Microwave promotion drastically reduces coupling times from hours to minutes and lowers the required excess of protected amino acids from 4-5 fold to approximately 2 fold. This results in higher crude purity (>85%) and significantly shorter synthesis cycles.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the microwave-assisted solid-phase synthesis method is designed for automation and large-scale implementation. The high efficiency and reduced reagent consumption make it economically viable for commercial manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Long-Acting GLP-1 Analogues Supplier
The technical potential of microwave-promoted synthesis for long-acting GLP-1 analogues represents a significant opportunity for pharmaceutical companies looking to optimize their diabetes treatment portfolios. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such complex molecules to market. Our facility is equipped with state-of-the-art rigorous QC labs and adheres to stringent purity specifications to ensure that every batch meets the highest international standards. We understand the critical nature of peptide synthesis and have invested heavily in the infrastructure necessary to handle sensitive modifications and high-purity requirements. Our team is dedicated to providing a seamless transition from process development to commercial manufacturing, ensuring that your supply chain remains robust and reliable.
We invite you to initiate a dialogue with our technical procurement team to explore how we can support your specific needs through a Customized Cost-Saving Analysis. By partnering with us, you can gain access to specific COA data and route feasibility assessments that will help you make informed decisions about your supply strategy. Our goal is to become your trusted partner in the production of high-quality pharmaceutical intermediates, delivering value through efficiency, quality, and reliability. Contact us today to discuss how we can help you optimize your supply chain and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
