Advanced Microwave-Assisted Synthesis of Long-Acting GLP-1 Analogues for Commercial Diabetes Therapeutics
Advanced Microwave-Assisted Synthesis of Long-Acting GLP-1 Analogues for Commercial Diabetes Therapeutics
The pharmaceutical landscape for Type 2 diabetes treatment has been revolutionized by the discovery of Glucagon-like Peptide-1 (GLP-1), yet its clinical utility has historically been constrained by rapid enzymatic degradation and renal clearance. Patent CN103087177A introduces a groundbreaking class of long-acting GLP-1 analogues that overcome these pharmacokinetic limitations through strategic structural modification at the 17th residue. By integrating a specialized small molecule moiety that promotes serum albumin binding, these analogues achieve a plasma half-life exceeding 40 hours, a massive improvement over the mere 5-minute half-life of native GLP-1. Furthermore, the patent details a highly efficient microwave-promoted solid-phase synthesis strategy that not only accelerates production but also enhances the purity profile of the final polypeptide, addressing critical bottlenecks in the manufacturing of complex peptide APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solid-phase peptide synthesis (SPPS) for long sequences like GLP-1 (30 amino acids) is notoriously inefficient and resource-intensive. In conventional protocols, the coupling of each amino acid residue to the growing peptide chain on the resin often requires extensive reaction times, typically ranging from 2 to 20 hours per cycle to ensure complete conversion. Similarly, the removal of the Fmoc protecting group usually necessitates incubation periods of 30 minutes to 1 hour. This sluggish kinetics leads to prolonged synthesis cycles, often spanning several days for a single batch, which drastically increases labor costs and energy consumption. Moreover, to drive these slow reactions to completion, conventional methods rely on a large excess of expensive protected amino acids, typically using 4 to 5 equivalents per coupling step. This excessive reagent usage not only inflates the raw material costs significantly but also generates substantial chemical waste, complicating downstream purification and environmental compliance.
The Novel Approach
The methodology outlined in the patent represents a paradigm shift by leveraging microwave irradiation to energize the peptide bond formation and deprotection steps. This novel approach utilizes a microwave-promoted Fmoc/tBu orthogonal protection strategy that dramatically accelerates reaction kinetics. Specifically, the average coupling time is reduced to approximately 10 minutes, and Fmoc deprotection is completed in merely 5 minutes under controlled microwave conditions at 50°C. This acceleration allows for the rapid assembly of the 30-residue peptide chain within a fraction of the traditional time. Crucially, the enhanced efficiency of microwave heating enables the use of only 2 equivalents of protected amino acids, halving the reagent consumption compared to standard practices. The result is a crude product with a purity greater than 80%, which significantly simplifies the subsequent purification workflow and improves overall yield.
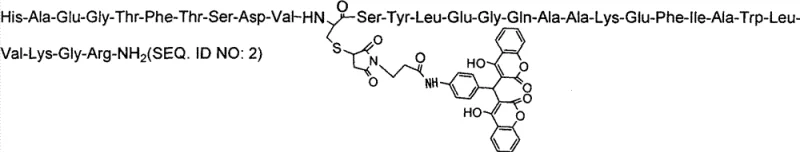
Mechanistic Insights into Albumin-Binding Mediated Stability
The core innovation of these analogues lies in the chemical modification at the 17th position of the GLP-1 sequence, where a cysteine derivative bearing a hydrophobic linker and a coumarin-based group is introduced. This structural alteration is designed to exploit the physiological abundance of serum albumin in the bloodstream. Once administered, the hydrophobic moiety on the peptide reversibly binds to serum albumin, effectively increasing the molecular size of the complex. This increased hydrodynamic radius prevents the peptide from passing through the glomerular filtration barrier in the kidneys, thereby inhibiting rapid renal clearance which is a primary elimination pathway for small peptides. Additionally, the specific conformational changes induced by this modification sterically hinder the access of Dipeptidyl Peptidase-IV (DPP-IV) enzymes to the N-terminal cleavage site, rendering the analogue resistant to enzymatic degradation.
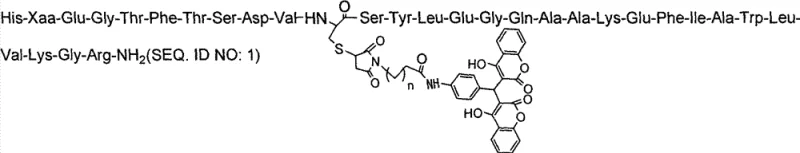
Biological evaluation data confirms that these mechanistic design principles translate into profound pharmacological benefits. While native GLP-1 is rapidly hydrolyzed by DPP-IV into inactive fragments within minutes, the modified analogues retain over 95% of their intact peptide chain even after 4 hours of incubation with the enzyme. In vivo studies demonstrate that the plasma half-life is extended from approximately 15 minutes for the prototype to over 40 hours, with some variants reaching up to 62 hours. This sustained presence in circulation ensures a continuous therapeutic effect, eliminating the need for frequent multiple daily injections required by short-acting formulations. The ability to maintain hypoglycemic activity for more than 24 hours post-administration validates the efficacy of the albumin-binding strategy in creating a truly long-acting therapeutic agent.
How to Synthesize Long-Acting GLP-1 Analogues Efficiently
The synthesis protocol described in the patent provides a robust framework for producing these high-value peptide intermediates with exceptional speed and purity. The process begins with the preparation of a specialized side-chain modified cysteine building block, followed by its incorporation into the peptide sequence on a Rink amide-MBHA resin using microwave-assisted cycles. This method is particularly advantageous for contract development and manufacturing organizations (CDMOs) looking to optimize their peptide production lines for speed and cost-efficiency. The detailed standardized synthetic steps for replicating this high-yield process are provided in the guide below.
- Synthesize the side-chain modified cysteine derivative by reacting Fmoc-Cys-OH with a coumarin-based linker in DCM using DIEA as a catalyst.
- Perform microwave-promoted Fmoc deprotection on Rink amide-MBHA resin using piperidine/HOBT in NMP at 50°C for significantly reduced cycles.
- Execute sequential amino acid coupling using microwave irradiation (25W, 50°C) for 7 minutes per residue, incorporating the modified cysteine at position 17.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microwave-promoted synthesis technology offers tangible strategic benefits beyond mere technical superiority. The transition from conventional thermal heating to microwave irradiation fundamentally alters the cost structure of peptide manufacturing by optimizing reagent utilization and throughput capacity. By reducing the requirement for protected amino acids from 4-5 equivalents down to just 2 equivalents, the direct material costs associated with the most expensive components of the synthesis are drastically lowered. This reduction in reagent excess also minimizes the volume of solvent and waste generated per kilogram of product, leading to significant savings in waste disposal and environmental compliance costs, which are increasingly critical in modern pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The implementation of microwave-assisted synthesis eliminates the need for prolonged reaction times, which translates directly into reduced energy consumption and lower labor overheads per batch. Since the coupling efficiency is significantly higher, the loss of valuable resin-bound intermediates due to incomplete reactions is minimized, thereby improving the overall yield of the final active pharmaceutical ingredient. Furthermore, the higher crude purity (>80%) achieved through this method reduces the burden on preparative HPLC purification, lowering the consumption of chromatography columns and solvents, which are major cost drivers in downstream processing.
- Enhanced Supply Chain Reliability: The accelerated synthesis cycle time means that production lead times can be significantly shortened, allowing for more responsive inventory management and faster time-to-market for new drug candidates. The robustness of the microwave method ensures consistent batch-to-batch quality, reducing the risk of production failures that can disrupt supply continuity. Additionally, the use of standard Fmoc-protected amino acids, which are widely available commodities, ensures that the supply chain is not dependent on obscure or hard-to-source specialty reagents, mitigating the risk of raw material shortages.
- Scalability and Environmental Compliance: The patent explicitly highlights that this method is easily automated and suitable for large-scale industrial production, making it an ideal candidate for commercial scale-up of complex peptide therapeutics. The reduction in chemical waste and solvent usage aligns with green chemistry principles, helping manufacturers meet stringent environmental regulations and sustainability goals. The ability to produce high-purity intermediates efficiently supports the reliable supply of high-purity API intermediates required for the formulation of stable, long-acting diabetes medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these long-acting GLP-1 analogues. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals evaluating this technology for potential licensing or manufacturing partnerships.
Q: How does the microwave-assisted method improve upon conventional solid-phase peptide synthesis?
A: The microwave-promoted method drastically reduces coupling time from approximately 20 hours to just 10 minutes and deprotection time from 1 hour to 5 minutes. Furthermore, it lowers the required excess of protected amino acids from 4-5 equivalents to only 2 equivalents, significantly reducing raw material costs and waste generation while improving crude product purity to over 80%.
Q: What is the biological advantage of modifying the 17th residue of GLP-1?
A: Modifying the 17th residue with a small molecule group facilitates binding to serum albumin. This modification prevents rapid kidney filtration and protects the peptide from degradation by DPP-IV enzymes, extending the plasma half-life from roughly 15 minutes for native GLP-1 to over 40 hours for the analogues described in the patent.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the microwave-promoted solid-phase synthesis method is easily automated and scalable. The high coupling efficiency, reduced reaction cycles, and simplified purification process due to higher crude purity make it highly suitable for industrial-scale manufacturing of complex peptide therapeutics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GLP-1 Analogues Supplier
The technological advancements detailed in patent CN103087177A represent a significant opportunity for the development of next-generation antidiabetic therapies with improved patient compliance profiles. NINGBO INNO PHARMCHEM stands at the forefront of peptide manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the rigorous demands of microwave-assisted peptide synthesis, ensuring that we can deliver these complex molecules with stringent purity specifications and rigorous QC labs to guarantee batch consistency and safety for clinical and commercial applications.
We invite global pharmaceutical partners to collaborate with us to leverage this innovative synthesis platform for your pipeline projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall cost of goods sold. Please contact our technical procurement team today to request specific COA data for our GLP-1 analogue portfolio and to discuss route feasibility assessments for your custom peptide synthesis needs.
