Revolutionizing 2-Chloro-5-Fluoro-Nicotinic Acid Production via Selective Catalytic Hydrogenation
The pharmaceutical and agrochemical industries continuously demand high-purity fluorinated heterocycles, yet the synthesis of key intermediates like 2-chloro-5-fluoro-nicotinic acid has historically been plagued by inefficient multi-step processes and hazardous reagents. A significant technological breakthrough is detailed in Chinese Patent CN100355732C, which introduces a novel preparation method that fundamentally alters the economic and operational landscape for producing these vital building blocks. This patent discloses a streamlined approach utilizing direct selective catalytic hydrogenation to remove specific chlorine atoms from 2,6-dichloro-5-fluoro-nicotinate precursors, effectively bypassing the cumbersome and low-yielding steps associated with legacy synthetic routes. For R&D directors and procurement strategists, this innovation represents a critical opportunity to optimize supply chains for reliable pharmaceutical intermediate supplier networks, ensuring consistent quality while drastically reducing the environmental footprint associated with traditional halogenated pyridine synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-5-fluoro-nicotinic acid has relied on convoluted pathways that introduce significant operational risks and cost inefficiencies. One documented literature route involves the nitration of 2-hydroxy-nicotinic acid followed by chlorination, reduction, and a final diazotization step using fluoroboric acid to install the fluorine atom. As illustrated in the reaction scheme below, this multi-step sequence is not only lengthy but also suffers from extremely low yields during the critical heating of the nitrodiazonium fluoroborate intermediate, making it economically unviable for large-scale operations.
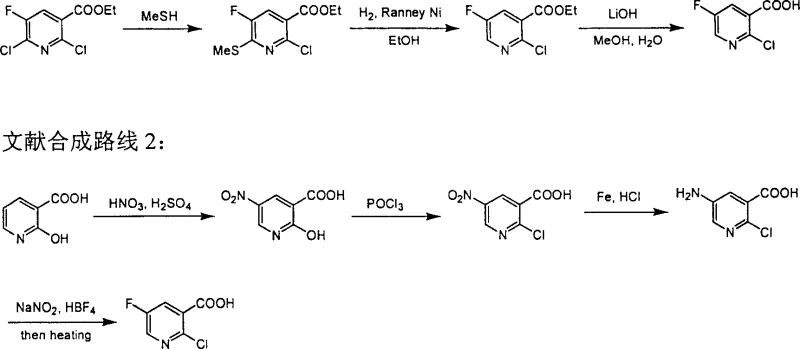
Furthermore, an alternative conventional method reported in medicinal chemistry literature relies on the reaction of 2,6-dichloro-5-fluoro-nicotinate with thiomethanol, followed by desulfurization using Raney Nickel. This approach is severely limited by the use of thiomethanol, a reagent known for its intensely unpleasant and hazardous odor, which complicates workplace safety and waste management. Additionally, the selectivity of the hydrogenation step in this older method is poor, often yielding only around 30% of the desired product while requiring highly specific grades of Raney Nickel that are difficult to source consistently, thereby creating bottlenecks in cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN100355732C offers a direct and elegant solution by leveraging selective catalytic hydrogenation. The core innovation lies in the ability to selectively remove the chlorine atom at the 6-position of the pyridine ring from 2,6-dichloro-5-fluoro-nicotinate or its corresponding acid, leaving the chlorine at the 2-position intact. This transformation is achieved under mild conditions using common hydrogenation catalysts such as Lindlar catalyst, Palladium on Carbon, or Raney Nickel in the presence of a base. The reaction scheme below highlights this efficient single-step conversion, which eliminates the need for malodorous thiols and dangerous diazonium salts, representing a paradigm shift towards greener and more efficient commercial scale-up of complex pharmaceutical intermediates.
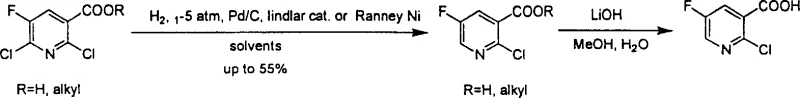
The versatility of this new approach is further demonstrated by its compatibility with various ester groups (where R can be H or alkyl), allowing manufacturers to produce either the free acid or the ester directly depending on downstream requirements. By operating at temperatures between 20°C and 50°C and pressures ranging from normal pressure up to 5 atm, the process ensures high safety standards and energy efficiency. This methodological simplicity not only improves the overall yield significantly compared to the 30% ceiling of previous methods but also simplifies the purification process, as the reaction mixture contains fewer byproducts, directly addressing the purity concerns of high-purity pharmaceutical intermediate buyers.
Mechanistic Insights into Selective Catalytic Hydrogenolysis
The success of this synthesis hinges on the precise mechanistic control of carbon-chlorine bond cleavage on the electron-deficient pyridine ring. In the 2,6-dichloro-5-fluoro-nicotinate substrate, the chlorine atoms at positions 2 and 6 exhibit different reactivities towards oxidative addition on the metal catalyst surface. The presence of the electron-withdrawing ester group at position 3 and the fluorine atom at position 5 creates a specific electronic environment that, when combined with the steric properties of the chosen catalyst (such as the lead-poisoned palladium in Lindlar catalyst), favors the activation and subsequent hydrogenolysis of the C-Cl bond at the 6-position. The addition of a base, such as triethylamine or trimethylamine, plays a crucial role in neutralizing the hydrogen chloride generated during the reaction, preventing catalyst poisoning and driving the equilibrium towards the formation of the desired 2-chloro-5-fluoro product.
From an impurity control perspective, this mechanism offers distinct advantages over nucleophilic substitution routes. Traditional methods often struggle with over-reaction or the formation of defluorinated byproducts due to the harsh conditions required for diazotization or nucleophilic displacement. In this catalytic hydrogenation system, the mild conditions and the specific selectivity of the catalyst minimize the risk of reducing the fluorine atom or the ester functionality. The result is a cleaner reaction profile where the primary impurity is typically the starting material or the fully dechlorinated species, both of which are easily separated. This high level of chemoselectivity ensures that the final product meets the stringent specifications required for API synthesis, reducing the burden on downstream purification teams and enhancing the overall reliability of the supply chain for reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize 2-Chloro-5-Fluoro-Nicotinate Efficiently
The implementation of this synthesis route is straightforward and relies on standard equipment found in most fine chemical manufacturing facilities, making it an ideal candidate for rapid technology transfer. The process begins with the dissolution of the 2,6-dichloro-5-fluoro precursor in a solvent such as ethyl acetate, ethanol, or tetrahydrofuran, followed by the addition of a stoichiometric amount of base and a catalytic loading of the metal catalyst. The reaction is then pressurized with hydrogen gas and stirred at ambient or slightly elevated temperatures until conversion is complete, typically within 12 hours. For a detailed breakdown of the specific molar ratios, workup procedures, and purification techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Dissolve 2,6-dichloro-5-fluoro-nicotinate in a suitable solvent such as ethyl acetate and add a base like triethylamine along with a catalyst such as Lindlar catalyst or Pd/C.
- Conduct hydrogenation under mild pressure (1-5 atm) at room temperature for approximately 12 hours to achieve selective dechlorination.
- Filter off the catalyst, concentrate the reaction mixture, and purify the resulting 2-chloro-5-fluoro-nicotinate via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic hydrogenation technology translates into tangible strategic benefits that extend beyond simple yield improvements. By eliminating the reliance on specialized and hazardous reagents like thiomethanol and fluoroboric acid, manufacturers can significantly reduce the costs associated with raw material sourcing, storage, and hazardous waste disposal. The simplified workflow reduces the number of unit operations required, which in turn lowers labor costs and increases plant throughput, allowing suppliers to offer more competitive pricing structures for long-term contracts without compromising on margin. This operational efficiency is critical for maintaining stability in the volatile market for fluorinated building blocks.
- Cost Reduction in Manufacturing: The elimination of the multi-step nitration and diazotization sequence removes several expensive and low-yielding stages from the production line, leading to substantial cost savings. Furthermore, the avoidance of malodorous thiol reagents negates the need for specialized scrubbing systems and extensive personal protective equipment, reducing overhead capital expenditure. The use of common hydrogenation catalysts like Pd/C or Raney Nickel, which are widely available and recyclable, ensures that catalyst costs remain a minor fraction of the total production cost, facilitating significant economic optimization.
- Enhanced Supply Chain Reliability: The starting material, 2,6-dichloro-5-fluoro-nicotinate, is derived from commercially available precursors and can be produced on a large scale, ensuring a robust and continuous supply of feedstock. Unlike the legacy routes which depend on reagents that may face regulatory restrictions or supply shortages due to their hazardous nature, the reagents used in this new process (hydrogen, base, common solvents) are commodity chemicals with stable global supply chains. This resilience minimizes the risk of production stoppages and ensures that delivery schedules for critical pharmaceutical intermediates can be met consistently, even during periods of market disruption.
- Scalability and Environmental Compliance: The reaction conditions are inherently safe, operating at low pressures and near-room temperatures, which simplifies the engineering requirements for scaling up from pilot plant to commercial tonnage. The absence of heavy metal waste streams associated with iron powder reductions or toxic diazonium byproducts makes the process easier to permit and operate under increasingly strict environmental regulations. This green chemistry profile not only future-proofs the manufacturing site against regulatory changes but also aligns with the sustainability goals of major multinational pharmaceutical clients who prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-chloro-5-fluoro-nicotinic acid using this patented methodology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on yield expectations, catalyst selection, and process safety. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own API synthesis workflows.
Q: What are the primary advantages of the new catalytic hydrogenation method over traditional nitration routes?
A: The new method eliminates the need for hazardous nitration and diazotization steps, significantly shortening the synthetic route and avoiding the use of malodorous thiol reagents found in older protocols.
Q: Which catalysts are most effective for the selective dechlorination of 2,6-dichloro-5-fluoro-nicotinate?
A: Patent data indicates that Lindlar catalyst, Palladium on Carbon (Pd/C), and Raney Nickel are all effective, with Lindlar catalyst demonstrating yields up to 55% under optimized conditions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available starting materials and operates under mild conditions (room temperature, low pressure), making it highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Fluoro-Nicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in patent literature can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-chloro-5-fluoro-nicotinic acid meets the exacting standards required for modern drug development, providing our partners with the confidence they need to advance their pipelines.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this superior synthesis method. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your premier partner for high-quality fluorinated pyridine intermediates.
