Scalable Industrial Production of 2-Chloro-5-Fluoro-Nicotinic Acid via Catalytic Dechlorination
Scalable Industrial Production of 2-Chloro-5-Fluoro-Nicotinic Acid via Catalytic Dechlorination
The global demand for fluorinated heterocyclic compounds continues to surge, driven by their critical role in modern medicinal chemistry and agrochemical formulations. Among these, 2-chloro-5-fluoro-nicotinic acid stands out as a pivotal building block, yet its efficient synthesis has historically presented significant challenges regarding yield, safety, and scalability. A breakthrough detailed in patent CN100355733C offers a robust solution, outlining a streamlined three-step pathway that transforms readily available 2,6-dichloro-5-fluoro-nicotinic acid into the target molecule with exceptional efficiency. This technical insight report analyzes the mechanistic advantages of this novel route, specifically focusing on its ability to bypass hazardous reagents and complex purification steps, thereby offering a compelling value proposition for R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-5-fluoro-nicotinic acid has been plagued by inefficient multi-step sequences and the use of environmentally unfriendly reagents. One prominent prior art method, documented in literature such as J. Med. Chem. 1993, relies on a nucleophilic substitution strategy using thiomethyl alcohol (MeSH). As illustrated in the reaction scheme below, this approach requires replacing a chlorine atom with a methylthio group, followed by desulfurization using Raney Nickel.

This conventional pathway suffers from severe drawbacks that hinder industrial adoption. Firstly, the use of thiomethyl alcohol introduces intense, unpleasant odors that complicate facility management and require specialized scrubbing systems, drastically increasing operational overhead. Secondly, the selectivity of the hydrogenolysis step is notoriously poor; achieving the removal of the sulfydryl group without affecting other sensitive functionalities often results in yields as low as 30%. Furthermore, alternative routes involving diazotization of amino-nicotinic acids are equally problematic, characterized by excessively long reaction sequences and the handling of unstable diazonium salts, which pose significant safety risks during scale-up.
The Novel Approach
In stark contrast to these legacy methods, the process disclosed in CN100355733C adopts a strategic "deconstruct-reconstruct" philosophy. Instead of struggling to selectively remove one chlorine atom from a dichloro precursor—a task that often leads to mixtures and low yields—the new method opts for complete dechlorination followed by selective re-chlorination. By utilizing catalytic hydrogenation to strip both chlorine atoms from 2,6-dichloro-5-fluoro-nicotinic acid, the process generates a clean 5-fluoro-nicotinic acid intermediate. This intermediate is then activated via N-oxidation, which electronically directs the subsequent chlorination exclusively to the 2-position. This logical inversion of the synthetic problem not only simplifies the reaction control but also utilizes inexpensive, commodity-grade reagents like hydrogen gas and phosphorus oxychloride, fundamentally altering the cost structure of cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Catalytic Dechlorination and N-Oxide Activation
The core innovation of this synthesis lies in the precise manipulation of electronic effects within the pyridine ring. The initial step involves the catalytic hydrogenolysis of carbon-chlorine bonds. While previous attempts, such as those depicted in the applicant's earlier patent work shown below, focused on selective dechlorination which proved difficult to control for the free acid form, the current method leverages vigorous conditions to ensure total conversion.
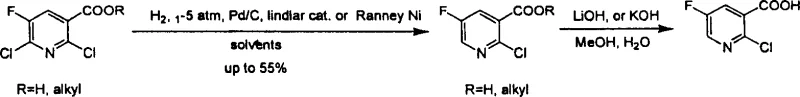
The transition from selective to total dechlorination is a masterstroke in process chemistry. By removing both chlorine atoms using catalysts like 5% Palladium on Carbon (Pd/C) or Raney Nickel at moderate pressures (1-5 atm), the reaction achieves near-quantitative yields (up to 98%) of 5-fluoro-nicotinic acid. This eliminates the formation of regio-isomeric impurities that would otherwise be inseparable without expensive chromatography. The subsequent N-oxidation step is equally critical; converting the pyridine nitrogen to an N-oxide dramatically changes the electron density of the ring. The N-oxide group acts as a powerful activating group for nucleophilic attack at the ortho (2-) position while deactivating the meta (4-) and para (6-) positions. When treated with chlorinating agents like POCl3 or PCl5, the oxygen of the N-oxide facilitates the displacement at the 2-position, effectively reinstalling the chlorine atom with high regioselectivity. This mechanism ensures that the final product profile is exceptionally clean, allowing for purification via simple recrystallization rather than column chromatography, a key factor for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Chloro-5-Fluoro-Nicotinic Acid Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize the benefits of the catalytic system. The process is divided into three distinct operational units: hydrogenation, oxidation, and chlorination. Each step has been optimized in the patent examples to balance reaction rate with safety and yield. For R&D teams looking to replicate or license this technology, understanding the interplay between catalyst loading, solvent choice, and temperature is essential for successful technology transfer.
- Perform catalytic hydrogenation on 2,6-dichloro-5-fluoro-nicotinic acid using Pd/C or Raney Ni to remove both chlorine atoms, yielding 5-fluoro-nicotinic acid.
- Oxidize the resulting 5-fluoro-nicotinic acid using hydrogen peroxide or peracids in acetic acid to form the N-oxide intermediate.
- React the N-oxide intermediate with a chlorinating agent such as phosphorus oxychloride (POCl3) or phosphorus pentachloride (PCl5) to selectively reintroduce chlorine at the 2-position.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from thiol-based chemistry to this catalytic hydrogenation route represents a significant opportunity for risk mitigation and cost optimization. The elimination of malodorous sulfur reagents removes a major bottleneck in facility scheduling and waste management, allowing for more flexible production planning. Moreover, the reliance on standard hydrogenation equipment and common oxidants means that this process can be deployed in existing multipurpose plants without requiring specialized capital investment.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the drastic simplification of the purification workflow. By avoiding column chromatography and utilizing recrystallization, the consumption of silica gel and organic solvents is significantly reduced, leading to lower variable costs per kilogram. Additionally, the high yield of the hydrogenation step (approaching 98%) minimizes raw material waste, ensuring that the expensive fluorinated starting material is utilized with maximum efficiency. The replacement of exotic reagents with commodity chemicals like hydrogen peroxide and phosphorus oxychloride further stabilizes the bill of materials against market volatility.
- Enhanced Supply Chain Reliability: The starting material, 2,6-dichloro-5-fluoro-nicotinic acid, is a commercially available bulk chemical, ensuring a secure upstream supply base. Unlike routes dependent on custom-synthesized diazonium salts or unstable intermediates, every step in this new process generates stable, isolable intermediates. This stability allows for the creation of strategic inventory buffers at intermediate stages, thereby reducing lead time for high-purity pharmaceutical intermediates and protecting downstream customers from supply disruptions caused by single-batch failures.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is vastly superior. The absence of sulfur-containing waste streams simplifies effluent treatment, reducing the load on wastewater processing facilities. The use of catalytic hydrogenation is a well-understood unit operation in the fine chemical industry, making the commercial scale-up predictable and safe. The ability to produce the final acid in multi-hundred gram to kilogram batches with consistent quality (as demonstrated in the patent embodiments) confirms its readiness for ton-scale production, aligning with the rigorous compliance standards required by global regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-chloro-5-fluoro-nicotinic acid. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy for decision-makers evaluating this technology for their supply chains.
Q: Why is the catalytic hydrogenation step critical in this synthesis?
A: The initial hydrogenation step completely removes both chlorine atoms from the 2,6-dichloro starting material. This 'reset' strategy avoids the difficult selectivity issues of trying to remove only one chlorine atom, ensuring a cleaner intermediate for the subsequent selective chlorination.
Q: How does this method improve upon traditional thiol-based routes?
A: Traditional methods often rely on malodorous and toxic thiomethyl alcohol (MeSH) for nucleophilic substitution. This patented process eliminates the need for thiols entirely, significantly improving workplace safety and reducing environmental waste treatment costs associated with sulfur removal.
Q: What purification methods are suitable for the final product?
A: Unlike previous methods that required column chromatography, this industrial process allows for purification via simple recrystallization (e.g., using water). This makes the process highly scalable and cost-effective for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Fluoro-Nicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires more than just a patent; it demands engineering excellence and unwavering quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN100355733C process are fully realized in our manufacturing suites. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-chloro-5-fluoro-nicotinic acid meets the exacting standards of the global pharmaceutical industry.
We invite forward-thinking partners to collaborate with us on optimizing this valuable intermediate for your specific drug development programs. By leveraging our technical expertise, you can access a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, securing a competitive advantage in your supply chain through superior chemistry.
