Advanced Manufacturing Strategies for High-Purity 4-Methoxy-3-Acetoxypicolinamide Derivatives
Advanced Manufacturing Strategies for High-Purity 4-Methoxy-3-Acetoxypicolinamide Derivatives
The global demand for high-efficacy fungicidal agents continues to drive innovation in the synthesis of complex heterocyclic aromatic amides. Patent CN109640657B presents a significant technological breakthrough in the preparation of 4-alkoxy-3-(acyl or aliphatic saturated hydrocarbyl)oxypicolinamides, specifically focusing on the scalable production of 4-methoxy-3-acetoxypicolinamide derivatives. These compounds serve as critical intermediates in the agrochemical sector, where purity and stereochemical integrity are paramount for biological activity. The disclosed methods offer a versatile platform for manufacturers, enabling the efficient coupling of picolinic acid derivatives with chiral 2-aminopropionate esters. By leveraging robust activation strategies and optimized workup procedures, this technology addresses the persistent challenges of regioselectivity and impurity control that have historically plagued the commercialization of such complex molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalized picolinamides often suffer from significant drawbacks that hinder their viability for industrial-scale manufacturing. Conventional approaches frequently rely on harsh reaction conditions that can compromise the stereochemical integrity of sensitive chiral centers, leading to racemization and reduced biological potency. Furthermore, achieving selective acylation at the 3-position of the pyridine ring without affecting the 4-methoxy group or the carboxylic acid moiety typically requires multiple protection and deprotection steps. These additional operations not only increase the overall processing time but also result in substantial yield losses and higher waste generation. The use of expensive transition metal catalysts in older methodologies introduces further complications regarding residual metal removal, which is strictly regulated in agrochemical and pharmaceutical applications. Consequently, there is a pressing need for a streamlined process that minimizes unit operations while maximizing atom economy and product purity.
The Novel Approach
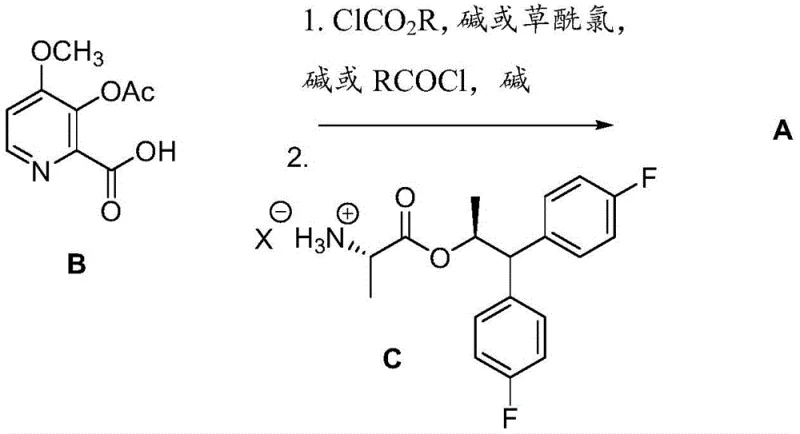
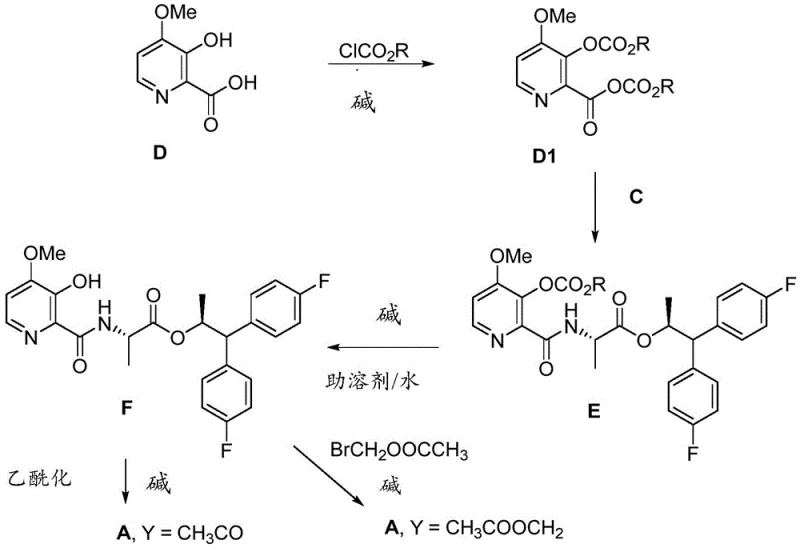
The methodology outlined in patent CN109640657B introduces a highly adaptable strategy that circumvents these traditional bottlenecks through two distinct yet complementary pathways. The first pathway involves the direct coupling of 4-methoxy-3-acetoxypicolinic acid (Formula B) with the chiral amine salt, utilizing mixed anhydride or acid chloride activation to ensure rapid amide bond formation under mild conditions. Alternatively, the second pathway allows for the coupling of 4-methoxy-3-hydroxypicolinic acid (Formula D) first, followed by a selective hydrolysis and subsequent acetylation or alkylation step. This modularity provides manufacturers with the flexibility to choose the most cost-effective route based on raw material availability and equipment constraints. By avoiding the need for transition metal catalysts and employing standard organic solvents like dichloromethane and tetrahydrofuran, this novel approach significantly simplifies the downstream purification process, thereby enhancing the overall economic feasibility of producing these high-value fungicidal intermediates.
Mechanistic Insights into Amide Coupling and Stereoselective Grignard Addition
The core of this synthetic technology lies in the precise activation of the carboxylic acid functionality and the preservation of chirality during the construction of the amine component. In the coupling phase, the carboxylic acid is activated using agents such as ethyl chloroformate, pivaloyl chloride, or oxalyl chloride in the presence of tertiary amines like triethylamine or DIPEA. This generates a reactive mixed anhydride or acid chloride intermediate in situ, which readily reacts with the nucleophilic amine to form the amide bond. The reaction kinetics are carefully controlled by maintaining low temperatures, typically between -15°C and 0°C, to suppress side reactions and prevent epimerization of the alpha-carbon in the amino acid ester. This temperature control is critical for maintaining the high enantiomeric excess required for the final fungicidal activity, as even minor deviations can lead to the formation of inactive diastereomers that are difficult to separate.
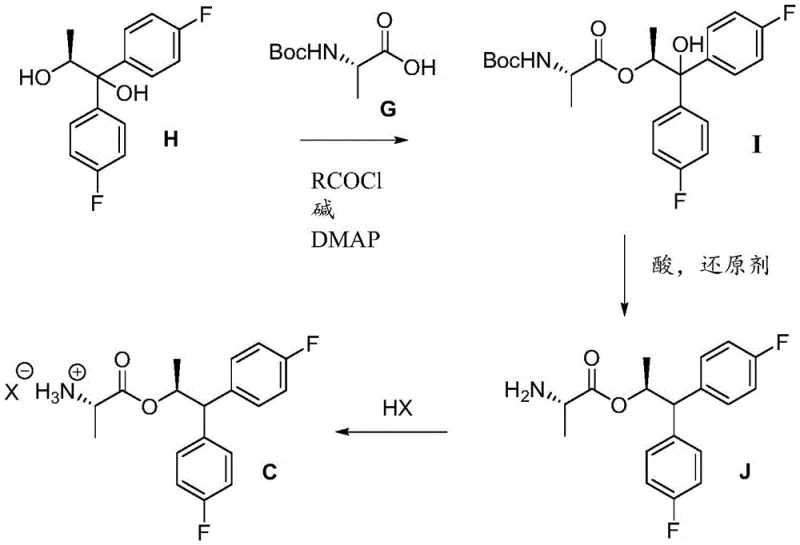
Equally important is the synthesis of the chiral 1,1-bis(4-fluorophenyl)propane-1,2-diol precursor (Formula H), which serves as the backbone for the amine component. This step involves the addition of excess 4-fluorophenyl magnesium bromide to (S)-ethyl lactate. The mechanism proceeds through a ketone intermediate which undergoes a second nucleophilic attack by the Grignard reagent. The stereochemistry is dictated by the initial chiral center of the lactate, which influences the facial selectivity of the incoming nucleophiles. Following the Grignard addition, the resulting diol is coupled with Boc-L-alanine using mixed anhydride activation, followed by a reductive deoxygenation step using silanes like triethylsilane or TMDS in the presence of strong acids like TFA. This reductive removal of the tertiary hydroxyl group is chemoselective, leaving the ester and carbamate functionalities intact, thereby ensuring the structural fidelity of the final amine salt used in the coupling reaction.
How to Synthesize 4-Methoxy-3-Acetoxypicolinamide Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to ensure consistent quality and yield. The process begins with the activation of the picolinic acid derivative, followed by the addition of the chiral amine salt under controlled thermal conditions. Detailed operational guidelines regarding solvent volumes, stoichiometric ratios, and quenching procedures are essential for successful scale-up. The patent provides extensive experimental data demonstrating the robustness of these conditions across different scales, from gram-level optimization to multi-kilogram production runs. For a comprehensive breakdown of the specific operational steps, including exact reagent quantities and workup protocols, please refer to the standardized synthesis guide below.
- Activate 4-methoxy-3-acetoxypicolinic acid or 4-methoxy-3-hydroxypicolinic acid using chloroformates or acid chlorides in the presence of a base.
- Couple the activated acid intermediate with 2-aminopropionic acid (S,S)-1,1-bis(4-fluorophenyl)propan-2-yl ester salt to form the amide bond.
- If starting from the hydroxy acid, perform hydrolysis followed by acetylation or alkylation to install the final Y group.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the processes described in CN109640657B offer substantial strategic advantages over legacy manufacturing methods. The reliance on commodity chemicals such as 4-fluorophenyl magnesium bromide, ethyl lactate, and common acylating agents ensures a stable and resilient supply chain that is less susceptible to market volatility. By eliminating the need for specialized transition metal catalysts, manufacturers can avoid the significant costs associated with catalyst procurement, recovery, and the rigorous testing required to certify low residual metal levels in the final product. This simplification of the bill of materials directly translates to improved margin potential and reduced working capital requirements for inventory management. Furthermore, the use of standard solvents and ambient pressure reactions facilitates the utilization of existing multipurpose reactor infrastructure, negating the need for costly capital expenditure on specialized equipment.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction of protection-deprotection sequences significantly lower the raw material and processing costs. The ability to isolate intermediates via simple crystallization or extraction rather than complex chromatography reduces solvent consumption and waste disposal fees. Additionally, the high yields reported in the patent examples minimize the loss of valuable chiral starting materials, ensuring that the cost per kilogram of the active intermediate remains competitive in the global agrochemical market.
- Enhanced Supply Chain Reliability: The starting materials identified in this process, such as 4-bromofluorobenzene and lactic acid derivatives, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate slight variations in temperature and addition rates without compromising product quality, ensures consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream formulators and agrochemical companies.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from laboratory benchtops to stainless steel reactors handling tens of kilograms of material, indicating strong potential for ton-scale commercial production. The aqueous workup procedures and the absence of heavy metals simplify wastewater treatment and align with increasingly stringent environmental regulations. The use of recyclable solvents like dichloromethane and ethyl acetate further supports sustainability goals, making this manufacturing route attractive for companies aiming to reduce their carbon footprint and enhance their corporate social responsibility profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on critical process parameters and quality control measures. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs. For more detailed technical specifications or custom feasibility studies, please consult with our engineering team.
Q: What are the key starting materials for the chiral amine component?
A: The chiral amine component is derived from (S)-ethyl lactate and 4-fluorophenyl magnesium bromide, ensuring high stereochemical purity.
Q: How is the regioselectivity of the acylation controlled?
A: Regioselectivity is managed by choosing between direct coupling of the pre-acetylated acid (Formula B) or post-coupling acetylation of the hydroxy intermediate (Formula F).
Q: Is this process suitable for large-scale production?
A: Yes, the patent describes examples scaled to over 10 kg using standard reactors and workup procedures like crystallization and extraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methoxy-3-Acetoxypicolinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation fungicides. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay. Our facility is equipped to handle the specific solvent systems and thermal requirements outlined in patent CN109640657B, guaranteeing a seamless transition from process development to full-scale manufacturing.
We invite you to collaborate with us to optimize your supply chain for these valuable agrochemical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's efficiency and profitability. Let us be your partner in bringing high-performance fungicidal solutions to the global market.
