Revolutionizing Fungicidal Picolinamide Production: Scalable Synthesis for Global Agrochemical Supply Chains
Patent CN109640658B introduces a novel, scalable process for synthesizing fungicidal 4-methoxy-3-(acyl or aliphatic saturated hydrocarbyl)oxypicolinamide compounds, representing a significant advancement in the production of agricultural chemical intermediates. This innovative methodology addresses longstanding challenges in the synthesis of these critical fungicidal agents by providing a more efficient, cost-effective, and environmentally sustainable manufacturing pathway that maintains high purity standards required for agrochemical applications. The patent details multiple synthetic routes that eliminate problematic steps found in conventional methods while improving overall yield and reducing hazardous waste generation. This breakthrough process enables manufacturers to produce these valuable fungicidal compounds at commercial scale with enhanced process control and reduced production costs, directly addressing the needs of global agricultural chemical producers seeking reliable supply chains for next-generation crop protection solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for fungicidal picolinamide compounds have been plagued by multiple significant limitations that hinder commercial viability and scalability. Conventional methods typically require harsh reaction conditions, including high temperatures and pressures, which increase energy consumption and safety risks in manufacturing facilities. These processes often employ transition metal catalysts that necessitate complex and costly removal procedures to meet stringent purity requirements for agricultural applications, resulting in additional processing steps and reduced overall yields. The multi-step nature of traditional syntheses leads to accumulation of impurities that are difficult to remove, compromising the final product's efficacy and requiring extensive purification protocols that drive up production costs. Furthermore, conventional approaches frequently utilize hazardous reagents and generate substantial waste streams that pose environmental challenges and increase disposal costs, making them less sustainable from both economic and ecological perspectives. The lack of robustness in these methods also creates supply chain vulnerabilities, as minor variations in reaction conditions can lead to significant batch-to-batch inconsistencies that affect product quality and reliability for end-users in the agricultural sector.
The Novel Approach
The patented process described in CN109640658B overcomes these limitations through an innovative synthetic strategy that streamlines production while maintaining high product quality. This novel approach eliminates the need for transition metal catalysts entirely, removing the associated purification challenges and cost burdens while improving environmental sustainability. The process utilizes mild reaction conditions that operate at ambient or slightly elevated temperatures, significantly reducing energy requirements and enhancing operational safety compared to conventional high-temperature methods. By implementing a carefully designed sequence of carboxylic acid activation followed by amide coupling, the patent achieves higher yields with fewer side reactions, resulting in a cleaner product profile that requires less extensive purification. The methodology incorporates multiple alternative pathways that provide flexibility in manufacturing based on available raw materials and facility capabilities, enhancing supply chain resilience for agrochemical producers. Additionally, the process generates less hazardous waste and utilizes more environmentally benign reagents, aligning with growing regulatory pressures for greener chemical manufacturing practices in the agricultural sector.
Mechanistic Insights into Amide Coupling Process
The core innovation of patent CN109640658B lies in its sophisticated approach to amide bond formation through carboxylic acid activation, which represents a significant advancement over conventional coupling methodologies. The process begins with the strategic activation of the picolinic acid derivative through formation of mixed anhydrides or acid chlorides using chloroformates or oxalyl chloride under carefully controlled conditions with appropriate bases such as triethylamine or DIPEA. This activation step creates a highly reactive intermediate that readily undergoes nucleophilic attack by the amine component without requiring transition metal catalysts, thereby eliminating the need for subsequent metal removal steps that complicate traditional syntheses. The reaction mechanism proceeds through a tetrahedral intermediate that collapses to form the desired amide bond while releasing the activating group as a byproduct that can be easily separated from the final product. This mechanistic pathway is particularly advantageous because it operates under mild conditions that minimize racemization of chiral centers present in the amine component, preserving the stereochemical integrity critical for fungicidal activity.
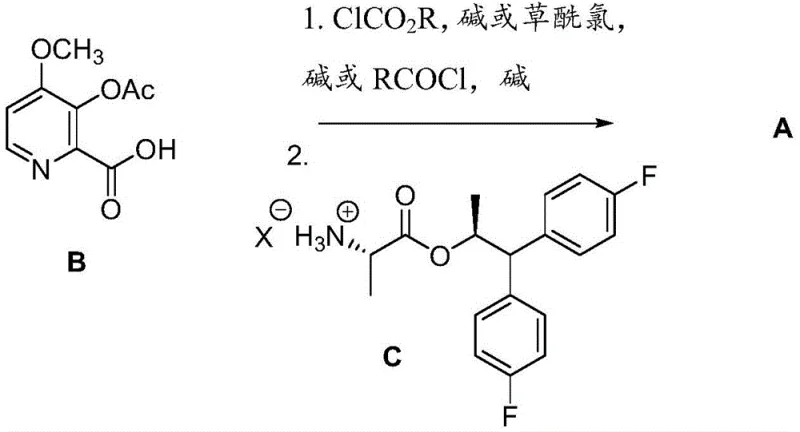
The patent further enhances this mechanism by introducing alternative activation strategies using sulfonated compounds that provide additional flexibility in manufacturing conditions while maintaining high reaction efficiency. These sulfonate-based activation methods offer improved selectivity and reduced side product formation compared to traditional approaches, contributing to higher overall yields and better process economics. The mechanistic design also incorporates careful control of reaction parameters such as temperature, solvent choice, and stoichiometry to optimize the coupling efficiency while minimizing unwanted side reactions that could lead to impurity formation.
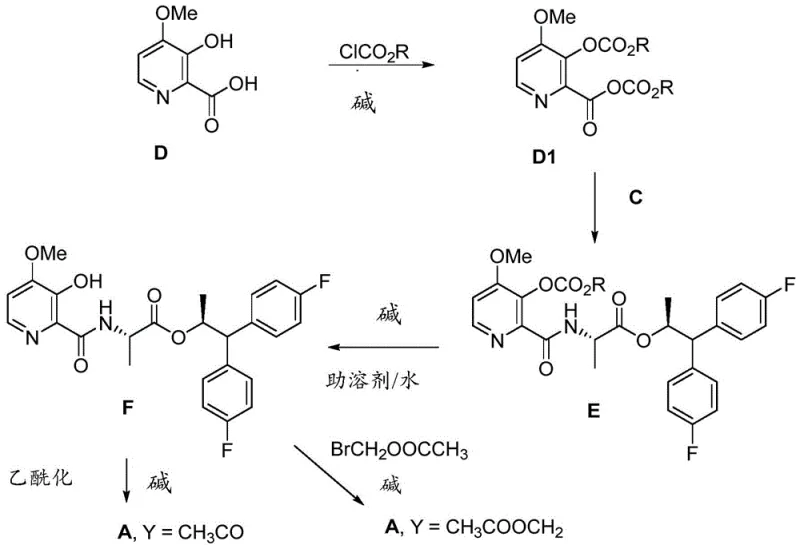
Impurity control is achieved through multiple built-in mechanisms within this synthetic approach. The selective activation chemistry minimizes the formation of common impurities such as dipeptides or racemized products that often plague traditional amide coupling methods. The process design includes specific purification steps at strategic points in the synthesis to remove potential impurities before they can accumulate or transform into more difficult-to-remove species later in the process. The use of mild reaction conditions prevents thermal degradation pathways that could generate unwanted byproducts, while the carefully selected solvent systems facilitate efficient separation of impurities during workup procedures. Additionally, the patent describes specific analytical methods for monitoring critical quality attributes throughout the process, enabling real-time adjustments to maintain product purity within the stringent specifications required for agrochemical applications.
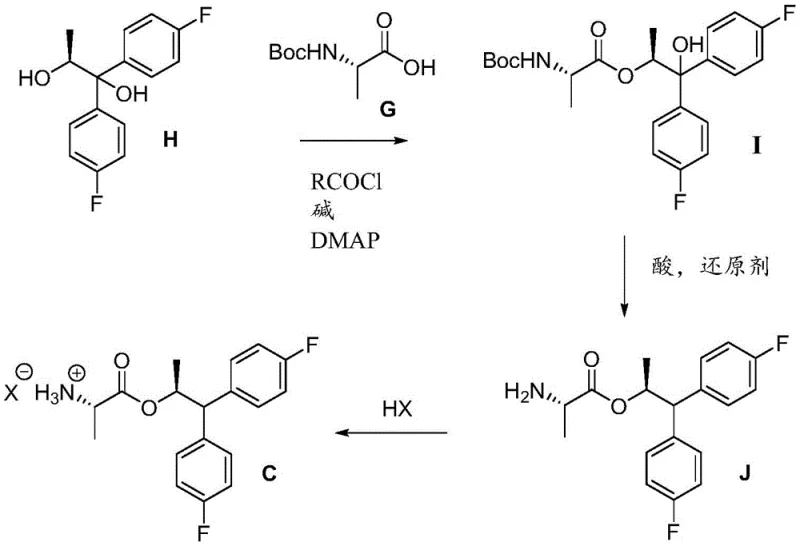
How to Synthesize Fungicidal Picolinamide Efficiently
The patented methodology provides a robust framework for synthesizing high-purity fungicidal picolinamide compounds through a series of well-defined chemical transformations that can be readily implemented in commercial manufacturing settings. This innovative process represents a significant improvement over previous synthetic approaches by eliminating problematic steps while maintaining excellent control over product quality and yield. The detailed standardized synthesis steps outlined below provide a comprehensive guide for process chemists and manufacturing teams looking to implement this technology in their production facilities, with specific attention given to critical process parameters that ensure consistent product quality at scale.
- Activate picolinic acid derivative B using chloroformate or acid chloride with appropriate base to form reactive intermediate
- Couple activated intermediate with amine salt C under controlled temperature conditions
- Isolate final product A through standard purification techniques including crystallization
Commercial Advantages for Procurement and Supply Chain Teams
This patented synthetic approach delivers substantial commercial benefits that directly address key concerns for procurement and supply chain management teams within agricultural chemical manufacturing organizations. By reimagining the traditional synthesis pathway for fungicidal picolinamides, the process creates multiple opportunities for cost optimization while simultaneously enhancing supply chain reliability and manufacturing flexibility. The elimination of problematic steps found in conventional methods translates to more predictable production timelines and reduced risk of supply disruptions, providing procurement teams with greater confidence in their ability to secure consistent supplies of these critical agrochemical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes an entire category of expensive materials and associated purification steps from the manufacturing process, resulting in significant cost savings without compromising product quality. The streamlined synthetic route reduces the number of processing steps required, lowering both direct material costs and indirect operational expenses related to equipment usage and labor. The use of more environmentally benign reagents also reduces waste disposal costs while aligning with sustainability initiatives that increasingly influence procurement decisions in the agricultural chemical sector.
- Enhanced Supply Chain Reliability: The process design incorporates multiple alternative pathways that provide flexibility in raw material sourcing and manufacturing conditions, reducing vulnerability to supply chain disruptions for specific reagents or equipment. The robust nature of the chemistry ensures consistent product quality across different manufacturing sites and scales, providing procurement teams with greater confidence in supplier reliability. The elimination of temperature-sensitive steps creates more forgiving process windows that accommodate minor variations in manufacturing conditions without impacting final product specifications.
- Scalability and Environmental Compliance: The methodology has been specifically designed with commercial scale-up in mind, featuring reaction conditions that translate seamlessly from laboratory to plant scale without requiring significant process re-engineering. The reduced environmental impact through lower energy consumption and less hazardous waste generation aligns with increasingly stringent regulatory requirements while supporting corporate sustainability goals. The process demonstrates excellent reproducibility across different scales, ensuring consistent product quality from pilot plant through full commercial production without unexpected performance issues.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation of this patented technology for commercial production of fungicidal picolinamide compounds. These answers are based on detailed analysis of the patent specifications and experimental data provided in the original filing, offering practical insights for technical evaluation by procurement and manufacturing teams.
Q: How does this patented process improve upon conventional synthesis methods?
A: The patented process eliminates transition metal catalysts entirely, operates under milder reaction conditions, and provides multiple alternative synthetic pathways that enhance flexibility while maintaining high product purity.
Q: What are the key advantages for large-scale commercial production?
A: The methodology demonstrates excellent scalability from laboratory to plant scale without requiring significant process re-engineering, with consistent product quality maintained across different production volumes.
Q: How does this process address environmental compliance concerns?
A: By eliminating hazardous reagents and reducing waste generation through streamlined reaction pathways, this process aligns with increasingly stringent environmental regulations while supporting corporate sustainability initiatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fungicidal Picolinamide Supplier
Our patented process for synthesizing fungicidal picolinamide compounds represents a significant advancement in agrochemical intermediate manufacturing, offering unparalleled efficiency, purity, and scalability for global agricultural chemical producers. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, with stringent purity specifications consistently met through our rigorous QC labs and advanced analytical capabilities. As a leading CDMO partner specializing in complex agrochemical intermediates, we possess deep expertise in navigating regulatory requirements while delivering consistent product quality that meets the most demanding specifications of global agricultural chemical manufacturers.
For procurement teams seeking to optimize their supply chain for critical fungicidal intermediates, we invite you to request a Customized Cost-Saving Analysis tailored to your specific production requirements. Our technical procurement team stands ready to provide detailed COA data and route feasibility assessments to help you evaluate how this innovative technology can enhance your manufacturing capabilities while reducing overall production costs.
