Scaling High-Purity Vitamin E Precursors: A Technical Breakdown of Continuous Ethynylation and Hydrogenation
Scaling High-Purity Vitamin E Precursors: A Technical Breakdown of Continuous Ethynylation and Hydrogenation
The global demand for high-purity pharmaceutical intermediates, particularly those serving as precursors for essential vitamins like Vitamin E, necessitates manufacturing processes that balance rigorous quality standards with economic efficiency. Patent CN100480221C introduces a sophisticated methodology for the preparation of alpha, beta-unsaturated higher alcohols, such as isophytol intermediates, through a sequence of continuous ethynylation, selective hydrogenation, and advanced distillation. This technical insight report analyzes the proprietary mechanisms within this patent to demonstrate how modern fine chemical manufacturing can achieve superior selectivity and space-time yields. By leveraging liquid ammonia solvent systems and divided-wall column technology, this process addresses the critical pain points of impurity control and energy consumption that traditionally plague the production of complex allyl alcohols. For R&D directors and supply chain leaders, understanding these mechanistic advantages is key to securing a reliable supply chain for high-value nutraceutical and pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional batch processes for synthesizing unsaturated alcohols often suffer from significant inefficiencies, primarily driven by the attempt to achieve quantitative conversion in the initial ethynylation step. When ketones are fully converted, secondary reactions inevitably occur, leading to the formation of diols and other high-boiling impurities that degrade the overall selectivity of the process. Furthermore, conventional neutralization methods using ammonium salts generate substantial wastewater streams laden with inorganic salts, creating environmental compliance burdens and increasing waste disposal costs. The reliance on suspended catalysts in hydrogenation steps further complicates the engineering landscape, as separating fine catalyst particles from the viscous product mixture requires extensive filtration and purification infrastructure. These factors collectively result in low space-time yields, necessitating larger reactor volumes and longer production cycles, which ultimately inflate the cost of goods sold and compromise supply chain agility for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast to legacy methodologies, the novel approach detailed in the patent utilizes a strategic partial conversion strategy during the ethynylation phase, limiting ketone conversion to between 50% and 95% to maximize selectivity above 97%. This counter-intuitive tactic prevents the formation of detrimental diol by-products, ensuring that the crude reaction mixture remains chemically clean before entering the hydrogenation stage. The integration of a divided-wall column for fractional distillation represents a significant engineering leap, allowing for the simultaneous separation of unreacted ketones and high-purity product within a single column shell. By recycling the unreacted ketones directly back to the ethynylation step, the process creates a closed-loop system that minimizes raw material waste and enhances overall atom economy. This streamlined continuous flow architecture not only reduces the physical footprint of the manufacturing plant but also drastically lowers energy consumption compared to traditional multi-column distillation sequences.

Mechanistic Insights into NH3/KOH Catalyzed Ethynylation and Selective Hydrogenation
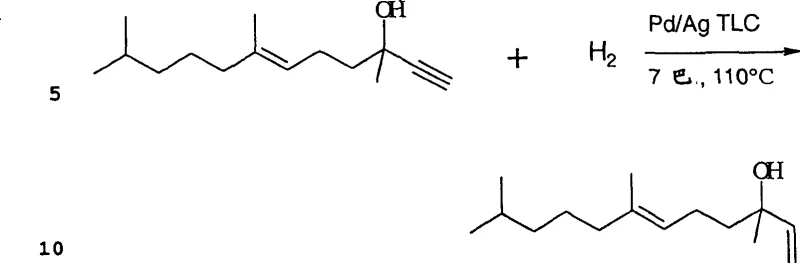
The core chemical transformation relies on the base-catalyzed monoethynylation of ketones in a liquid ammonia medium, typically utilizing potassium hydroxide or potassium methoxide as the catalytic base. Operating at moderate temperatures between 20°C and 50°C and pressures around 20 bar, this reaction environment favors the formation of the desired acetylenic alcohol while suppressing oligomerization. The use of liquid ammonia serves a dual purpose: it acts as a solvent to dissolve the reactants and facilitates the easy removal of unreacted acetylene and solvent via flash vaporization for recycling. This specific solvent system is crucial for maintaining the homogeneity of the reaction mixture and ensuring efficient heat transfer, which is vital for controlling the exothermic nature of the ethynylation reaction. The precise control of molar ratios, particularly maintaining an excess of acetylene relative to the ketone, drives the equilibrium towards the desired mono-adduct without promoting over-reaction.

Following ethynylation, the selective hydrogenation of the acetylenic bond to an allylic double bond is achieved using a palladium-containing thin-layer catalyst, often promoted with silver to enhance selectivity. This heterogeneous catalytic system is designed to hydrogenate the triple bond without reducing the existing double bonds or the carbonyl groups present in the molecule, a common challenge in complex terpene synthesis. The thin-layer configuration of the catalyst minimizes diffusion limitations, allowing for high reaction rates and excellent mass transfer characteristics within a continuous flow reactor. By carefully controlling the hydrogen partial pressure and introducing trace amounts of carbon monoxide as a selectivity modifier, the process effectively suppresses over-hydrogenation to the saturated alcohol. This level of catalytic precision ensures that the final product profile meets the stringent purity specifications required for downstream Vitamin E synthesis, minimizing the need for costly corrective purification steps.
How to Synthesize High-Purity Allyl Alcohols Efficiently
Implementing this synthesis route requires a seamless integration of reaction and separation units to maintain the delicate balance of partial conversion and continuous recycling. The process begins with the continuous feeding of ketone, liquid ammonia, and catalyst solution into a tubular reactor designed for plug flow characteristics, ensuring uniform residence time and reaction conditions. Following the reaction, the mixture undergoes a multi-stage degassing and neutralization process where unreacted gases are separated and the catalyst is quenched using carbon dioxide and water, avoiding the introduction of inorganic salts. The resulting organic phase is then fed directly into the hydrogenation unit, where the acetylenic intermediates are converted to the target allyl alcohols over the fixed-bed catalyst system. Detailed standardized synthesis steps and specific operating parameters for scaling this pathway are outlined in the technical guide below.
- Perform base-catalyzed monoethynylation of ketones in liquid ammonia at 20-50°C, maintaining partial conversion (50-95%) to ensure high selectivity.
- Execute selective hydrogenation of the acetylenic alcohol intermediate using a Pd-containing thin-layer catalyst in the presence of hydrogen.
- Purify the crude hydrogenation product via fractional distillation in a divided-wall column, separating unreacted ketones for recycling and isolating the high-purity allyl alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this continuous manufacturing technology offers substantial strategic advantages over traditional batch processing methods. The ability to recycle unreacted starting materials directly back into the process significantly reduces the consumption of raw ketones, which are often expensive and subject to market volatility. This closed-loop material flow enhances supply chain resilience by decoupling production output from fluctuations in raw material availability, ensuring a more consistent and reliable supply of critical intermediates. Furthermore, the continuous nature of the operation allows for a smaller physical footprint and reduced utility consumption per unit of product, translating into lower operational expenditures and a reduced carbon footprint for the manufacturing facility. These efficiencies collectively contribute to a more competitive cost structure without compromising the high-quality standards demanded by the pharmaceutical industry.
- Cost Reduction in Manufacturing: The implementation of partial conversion strategies combined with efficient recycling loops eliminates the need for expensive purification steps to remove diol by-products, thereby reducing solvent usage and waste treatment costs. By avoiding the generation of inorganic salt waste through CO2 neutralization instead of acid neutralization, the process significantly lowers environmental compliance costs and wastewater treatment burdens. The high selectivity of the catalyst system extends catalyst life and reduces the frequency of catalyst replacement, further driving down long-term operational expenses. These cumulative efficiencies result in a leaner manufacturing cost profile that provides significant value to procurement teams managing tight budgets.
- Enhanced Supply Chain Reliability: Continuous processing equipment typically offers higher availability and shorter changeover times compared to batch reactors, ensuring a steady and predictable output of intermediates. The robustness of the thin-layer catalyst system against poisoning and deactivation means fewer unplanned shutdowns for maintenance, guaranteeing consistent delivery schedules for downstream customers. Additionally, the modular nature of the continuous flow units allows for easier capacity expansion or replication at different sites, mitigating the risk of supply disruptions due to single-site failures. This reliability is crucial for pharmaceutical supply chains where continuity of supply is often as important as the price of the material.
- Scalability and Environmental Compliance: The process is inherently scalable, as increasing production capacity often involves running the continuous units for longer durations or numbering up reactor modules rather than building massive new batch vessels. The reduced energy consumption of the divided-wall distillation column and the elimination of salt-laden wastewater align with increasingly stringent global environmental regulations and corporate sustainability goals. By minimizing the generation of hazardous waste and optimizing energy efficiency, the manufacturing process supports a greener supply chain that appeals to environmentally conscious stakeholders. This forward-looking approach ensures long-term operational viability in a regulatory landscape that is constantly tightening around chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These answers are derived directly from the patent specifications and practical engineering considerations for scaling fine chemical processes. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The responses highlight the specific advantages in terms of purity, efficiency, and operational stability that distinguish this method from conventional alternatives.
Q: How does partial conversion in ethynylation improve product purity?
A: By limiting ketone conversion to 50-95% rather than 100%, the process minimizes secondary reactions that form diols. This results in selectivity greater than 97%, significantly reducing downstream purification burdens and impurity profiles.
Q: What are the advantages of using a divided-wall column for this separation?
A: Divided-wall columns allow for the separation of multicomponent mixtures in a single shell, reducing energy consumption and capital investment compared to traditional multi-column sequences while achieving high-purity side draws.
Q: Is this process suitable for large-scale Vitamin E precursor production?
A: Yes, the continuous nature of the ethynylation and hydrogenation steps, combined with efficient catalyst life and recycling loops, makes it highly scalable for commercial production of isophytol intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isophytol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes for producing high-value pharmaceutical intermediates like isophytol and its precursors. Our CDMO capabilities are specifically engineered to translate complex patent technologies into commercial reality, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of allyl alcohol or ketone intermediate meets the exacting standards required for Vitamin E synthesis. Our technical team is adept at optimizing reaction parameters such as temperature, pressure, and catalyst loading to maximize yield and minimize impurities, ensuring a consistent and high-quality product supply for our global partners.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for these critical intermediates. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our continuous manufacturing capabilities can reduce your total cost of ownership. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your project requirements. Let us partner with you to secure a sustainable and efficient supply of high-purity chemical intermediates for your next generation of pharmaceutical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
