Advanced One-Pot Synthesis of Herbicidal Sulfonylureas for Commercial Agrochemical Production
Advanced One-Pot Synthesis of Herbicidal Sulfonylureas for Commercial Agrochemical Production
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing, particularly for sulfonylurea classes known for their potent biological activity. A pivotal advancement in this domain is detailed in Chinese Patent CN1071748C, which discloses a robust method for preparing sulphonyl ureas with herbicidal properties using N-pyrimidinyl or N-triazinyl carbamates as key intermediates. This technology represents a significant departure from legacy synthetic routes that heavily relied on hazardous reagents such as phosgene or alkali metal hydrides. By shifting to a safer, more efficient protocol involving diaryl carbonates and oxygen-containing bases, this invention addresses critical pain points in both laboratory research and industrial scale-up. For R&D directors and procurement strategists, understanding this mechanistic shift is essential for optimizing supply chains and ensuring regulatory compliance in the production of next-generation crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonylurea herbicides has been plagued by significant safety and efficiency challenges inherent to traditional chemical methodologies. Conventional processes frequently necessitate the use of extremely hazardous reagents, most notably phosgene gas, which poses severe toxicity risks and requires elaborate containment and scrubbing systems to manage emissions safely. Furthermore, many established protocols depend on alkali metal hydrides, such as sodium hydride, as strong bases to activate amine substrates. These hydrides are pyrophoric and react violently with moisture, demanding stringent anhydrous conditions and specialized handling equipment that drastically inflate operational expenditures. Additionally, when dealing with heterocyclic amines that exhibit poor solubility in common organic solvents, these older methods often suffer from incomplete reactions and sluggish kinetics, resulting in suboptimal yields and complex purification burdens that hinder commercial viability.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a transformative approach that replaces dangerous hydrides with safer, oxygen-containing bases such as alkali metal alkoxides, hydroxides, or carbonates. This novel route utilizes diaryl carbonates, specifically compounds like diphenyl carbonate, as the carbonyl source, which are stable, non-toxic solids that are far easier to handle and transport than gaseous phosgene. The process allows for the effective activation of heterocyclic amines, including those with challenging solubility profiles like 2-amino-4-methoxy-6-methyltriazine, converting them efficiently into the necessary carbamate intermediates. 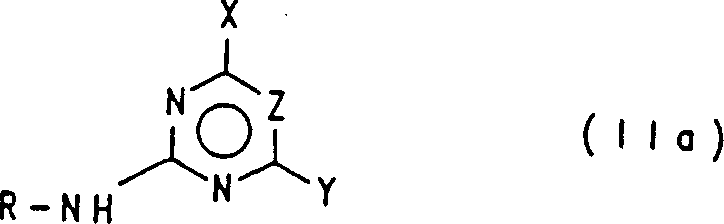 By enabling a one-pot synthesis where the carbamate intermediate reacts directly with the sulfonamide without isolation, this approach streamlines the workflow, minimizes solvent consumption, and eliminates the need for intermediate purification steps, thereby offering a compelling solution for cost reduction in herbicide manufacturing.
By enabling a one-pot synthesis where the carbamate intermediate reacts directly with the sulfonamide without isolation, this approach streamlines the workflow, minimizes solvent consumption, and eliminates the need for intermediate purification steps, thereby offering a compelling solution for cost reduction in herbicide manufacturing.
Mechanistic Insights into Oxygen-Base Catalyzed Carbamate Formation
The core of this technological breakthrough lies in the precise mechanistic pathway that facilitates the formation of the urea linkage under mild conditions. The reaction initiates with the deprotonation of the heterocyclic amine of formula (II) using a suitable oxygen-containing base, such as sodium tert-butoxide or potassium ethoxide, within a polar aprotic solvent like dimethylacetamide (DMA) or dimethylformamide (DMF). This step generates a reactive amine salt (II-S) in situ, which serves as a potent nucleophile. Subsequently, this anionic species attacks the carbonyl carbon of the diaryl carbonate (III), displacing a phenoxide leaving group to form the N-substituted carbamate (IV). This nucleophilic acyl substitution is highly favorable due to the stability of the phenoxide byproduct and the electrophilicity of the carbonate. The resulting carbamate then undergoes a second nucleophilic attack by the sulfonamide (V), ultimately yielding the target sulfonylurea (I) with the release of another equivalent of phenol or aryl alcohol.
From an impurity control perspective, this mechanism offers distinct advantages over hydride-based routes. The use of oxygen-containing bases avoids the generation of hydrogen gas, which can lead to pressure buildup and safety incidents in large reactors. Moreover, the reaction conditions, typically ranging from -20°C to 100°C depending on the solvent boiling point, allow for tight thermal control that suppresses side reactions such as hydrolysis of the carbonate or degradation of the sensitive heterocyclic ring. The selection of solvents like DMA or DMSO ensures that all ionic intermediates remain in solution, promoting homogeneous reaction kinetics and preventing the formation of insoluble byproducts that could trap impurities. 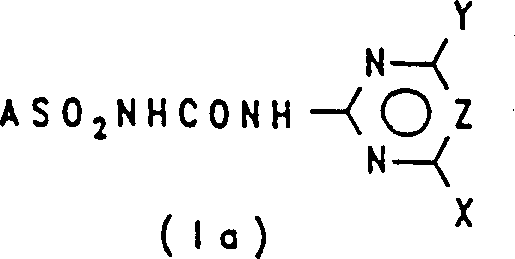 This controlled environment ensures that the final product achieves high purity levels, often exceeding 90% without the need for extensive chromatographic purification, which is critical for meeting the stringent specifications required by agrochemical registrars.
This controlled environment ensures that the final product achieves high purity levels, often exceeding 90% without the need for extensive chromatographic purification, which is critical for meeting the stringent specifications required by agrochemical registrars.
How to Synthesize N-(pyrimidinyl or triazinyl)-carbamate Efficiently
The practical implementation of this synthesis route is designed for scalability and operational simplicity, making it ideal for contract development and manufacturing organizations (CDMOs). The process begins by suspending the heterocyclic amine in a dry polar aprotic solvent and treating it with a stoichiometric amount of an alkali metal alkoxide at temperatures between -10°C and 100°C. Once the salt formation is complete, the diaryl carbonate is added, and the mixture is stirred to form the carbamate intermediate. Without isolating this intermediate, the sulfonamide component is introduced to the same reaction vessel, allowing the urea bond to form seamlessly. Detailed standardized synthetic steps see the guide below.
- Deprotonate the heterocyclic amine (formula II) using an oxygen-containing base like sodium tert-butoxide in a polar aprotic solvent to form the corresponding salt.
- React the amine salt directly with a diaryl carbonate (formula III) without isolation to generate the N-pyrimidinyl or N-triazinyl carbamate intermediate.
- Add the sulfonamide (formula V) to the reaction mixture to complete the urea linkage, followed by acidification and purification to isolate the final sulfonylurea.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits that extend beyond mere chemical yield. By eliminating the reliance on alkali metal hydrides and phosgene, manufacturers can drastically reduce the capital expenditure associated with safety infrastructure, such as explosion-proof facilities and toxic gas monitoring systems. This shift not only lowers the barrier to entry for production but also mitigates the risk of supply disruptions caused by the strict regulatory controls often placed on hazardous precursor chemicals. Furthermore, the ability to perform the reaction in a one-pot fashion significantly reduces solvent usage and waste generation, aligning with modern green chemistry principles and reducing the environmental compliance burden.
- Cost Reduction in Manufacturing: The replacement of expensive and dangerous reagents like sodium hydride with readily available alkoxides and diaryl carbonates leads to substantial raw material cost savings. Additionally, the elimination of intermediate isolation steps reduces labor costs, energy consumption for drying and solvent recovery, and equipment occupancy time. The process avoids the need for specialized containment required for pyrophoric materials, further lowering the overall cost of goods sold (COGS) for the final herbicide active ingredient.
- Enhanced Supply Chain Reliability: Diaryl carbonates and alkali metal alkoxides are commodity chemicals with robust global supply chains, unlike specialized hazardous reagents that may face logistical bottlenecks or shipping restrictions. This availability ensures consistent production schedules and reduces the lead time for high-purity agrochemical intermediates. The stability of the reagents also allows for longer shelf-life and simpler storage requirements, minimizing inventory losses and ensuring continuity of supply even during market fluctuations.
- Scalability and Environmental Compliance: The one-pot nature of the reaction simplifies scale-up from pilot plant to commercial production, as there are fewer unit operations and transfer steps that could introduce variability or contamination. The reduced generation of hazardous waste, particularly the absence of hydride quenching byproducts and phosgene scrubbing waste, simplifies effluent treatment and lowers disposal costs. This environmental profile makes the process highly attractive for manufacturing in regions with strict environmental regulations, facilitating smoother regulatory approvals and long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonylurea synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: Why is the diaryl carbonate method superior to traditional phosgene methods for sulfonylurea synthesis?
A: Traditional methods often rely on highly toxic phosgene or dangerous alkali metal hydrides like sodium hydride. The patented diaryl carbonate route eliminates these hazards, using stable solid reagents that significantly reduce safety infrastructure costs and simplify waste treatment protocols.
Q: How does this process handle heterocyclic amines with poor solubility?
A: The process utilizes strong oxygen-containing bases in polar aprotic solvents to effectively deprotonate even poorly soluble heterocyclic amines. This ensures high conversion rates and excellent yields without requiring complex solubilizing agents or extreme conditions.
Q: Can the carbamate intermediate be isolated before reacting with the sulfonamide?
A: Yes, the carbamate intermediate can be isolated using conventional methods like filtration or crystallization. However, the patent highlights a preferred one-pot approach where the intermediate reacts directly with the sulfonamide, saving time and solvent usage while maintaining high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonylurea Intermediate Supplier
The technological potential of this carbamate-mediated synthesis route is immense, offering a pathway to produce high-quality herbicide intermediates with superior safety and efficiency profiles. As a leading CDMO expert, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemical transformations like this are executed with precision and reliability. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of sulfonylurea intermediate meets the exacting standards required for global agrochemical registration and formulation.
We invite you to collaborate with us to leverage this advanced chemistry for your product pipeline. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and enhance your competitive position in the herbicide market.
