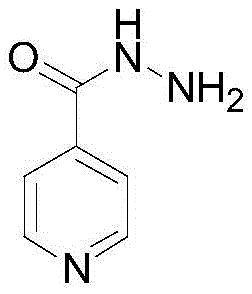
Advanced Isoniazid Manufacturing: Leveraging HCl Salt Intermediates for Ultra-High Purity
Advanced Isoniazid Manufacturing: Leveraging HCl Salt Intermediates for Ultra-High Purity
The global demand for high-quality anti-tuberculosis agents necessitates robust and efficient synthetic routes for key active pharmaceutical ingredients. Patent CN111138354A introduces a transformative preparation method for isoniazid that addresses longstanding challenges in purity and reaction efficiency. This technical insight report analyzes the novel pathway which incorporates a critical isonicotinic acid ester hydrochloride intermediate step. By integrating this specific purification stage prior to hydrazinolysis, the process effectively eliminates persistent impurities such as unreacted isonicotinic acid and 2-picolinic acid derivatives. The result is a finished product with exceptional purity levels reaching 99.99%, meeting stringent pharmacopoeial standards while simultaneously optimizing production timelines. For procurement and supply chain leaders, this represents a significant opportunity to enhance the reliability of pharmaceutical intermediates sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for isoniazid have historically struggled with balancing yield against purity. The direct condensation of isonicotinic acid with hydrazine hydrate often requires elevated temperatures and extended reaction times, leading to incomplete conversion and difficult purification scenarios. Alternatively, routes proceeding through 4-cyanopyridine involve hydrolysis steps that are notoriously difficult to control, frequently generating byproducts like isonicotinamide and complex triazole derivatives. Even standard esterification pathways followed by direct hydrazinolysis face significant hurdles; the reaction mixture often becomes highly viscous, impairing stirring efficiency and heat transfer. Furthermore, raw material impurities, specifically 2-picolinic acid, tend to carry through the synthesis, contaminating the final API and requiring costly downstream remediation efforts that erode profit margins.
The Novel Approach
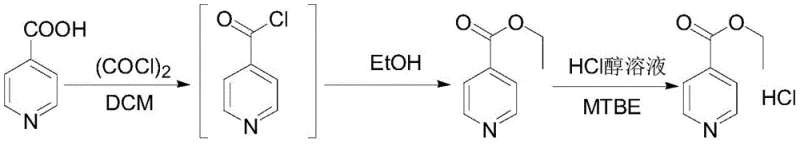
The methodology disclosed in CN111138354A fundamentally alters the process landscape by inserting a strategic hydrochloride salt formation step. Instead of proceeding directly from the crude ester to the hydrazide, the isonicotinic ester is reacted with an ether reagent in an alcoholic hydrogen chloride solution. This step converts the ester into its hydrochloride salt, which precipitates or crystallizes out, leaving behind soluble impurities like unreacted acids and isomers in the mother liquor. This in-process purification ensures that only high-purity intermediate enters the final condensation stage. Consequently, the subsequent hydrazinolysis reaction proceeds in a cleaner, less viscous environment, drastically reducing the required reaction time and simplifying the final refining process to achieve ultra-high purity specifications.
Mechanistic Insights into Ester Hydrochloride-Mediated Purification
The core innovation of this synthesis lies in the differential solubility and reactivity of the intermediate species. During the esterification phase, isonicotinic acid is converted to ethyl isonicotinate using an acylating agent such as oxalyl chloride. However, rather than isolating the free base ester, the process immediately treats the reaction mixture with hydrogen chloride in the presence of an ether solvent like methyl tert-butyl ether. The formation of the ethyl isonicotinate hydrochloride salt is thermodynamically favorable under these conditions. Crucially, potential impurities such as 2-picolinic acid ethyl ester or residual isonicotinic acid form salts with different solubility characteristics in the ether-alcohol matrix. This allows for a highly effective solid-liquid separation where the desired intermediate is retained as a high-purity solid, while contaminants remain dissolved in the filtrate.
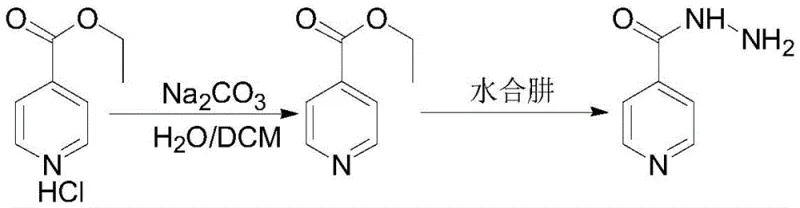
Following the isolation of the pure hydrochloride salt, the material is dissociated, typically using a mild base like sodium carbonate, to regenerate the free ester in situ or in a separate organic phase. This purified ester then undergoes condensation with hydrazine hydrate. Because the starting material for this step is virtually free of acidic impurities and isomers, the nucleophilic attack by hydrazine on the carbonyl carbon proceeds with high specificity and kinetics. The absence of competing side reactions means the system does not develop the high viscosity associated with traditional bulk reactions. This mechanistic clarity ensures that the final crude product requires minimal refinement, usually just a recrystallization from aqueous alcohol, to reach the target 99.99% purity threshold.
How to Synthesize Isoniazid Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and solvent selection to maximize the purification effect of the hydrochloride salt step. Operators must ensure that the molar ratio of the acylating agent to isonicotinic acid is maintained between 1:1.3 and 1:3.0 to drive complete conversion during the initial activation. Furthermore, the selection of the ether reagent is critical; methyl tert-butyl ether is preferred due to its ability to maximize the solubility difference between the target ester hydrochloride and impurity salts. The detailed standardized operating procedures, including specific temperature ramps and addition rates for the hydrogen chloride solution, are essential for reproducibility. For a comprehensive breakdown of the exact experimental parameters and safety protocols required for execution, please refer to the standardized guide below.
- Perform esterification of isonicotinic acid with alcohol and an acylating agent to obtain isonicotinic ester.
- React the isonicotinic ester with an ether reagent in an alcoholic hydrogen chloride solution to form isonicotinic ester hydrochloride, removing impurities.
- Dissociate the hydrochloride salt and conduct condensation hydrazinolysis with hydrazine hydrate to generate crude isoniazid.
- Refine the crude product using an aqueous alcohol solution to obtain finished isoniazid with purity up to 99.99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers distinct strategic advantages beyond mere chemical elegance. The primary value driver is the substantial reduction in processing time and resource consumption associated with purification. By removing impurities at the intermediate stage rather than the final API stage, manufacturers avoid the yield losses typically incurred during aggressive final recrystallizations or chromatographic separations. This efficiency translates directly into improved throughput and lower cost of goods sold. Additionally, the use of commodity chemicals such as ethanol, oxalyl chloride, and hydrazine hydrate ensures that the supply chain remains resilient and less susceptible to volatility compared to routes relying on exotic catalysts or specialized reagents.
- Cost Reduction in Manufacturing: The elimination of complex downstream purification steps significantly lowers operational expenditures. By preventing the carryover of 2-picolinic acid and other isomers, the need for extensive waste treatment and solvent recovery associated with failed batches or low-yield refinements is minimized. The process operates at moderate temperatures, reducing energy consumption for heating and cooling cycles compared to high-temperature direct condensation methods. Furthermore, the higher overall yield resulting from the protective salt formation step means less raw material is required per kilogram of finished isoniazid, driving down the unit cost of production substantially.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals mitigates the risk of supply disruptions. Isonicotinic acid is a mature commodity, and the reagents used for activation and salt formation are standard in fine chemical manufacturing. This universality allows for multi-sourcing strategies, ensuring that production schedules are not held hostage by single-supplier bottlenecks. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures due to minor process deviations, thereby guaranteeing consistent delivery timelines to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial reactors without requiring specialized high-pressure or cryogenic equipment. The use of aqueous workups and standard organic solvents facilitates straightforward waste stream management and solvent recycling, aligning with modern green chemistry principles. The reduction in reaction time for the hydrazinolysis step also increases reactor turnover rates, allowing existing infrastructure to produce greater volumes without capital expansion, thus optimizing asset utilization and environmental footprint per unit of output.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoniazid synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating technology transfer and for quality assurance teams establishing specification limits. The answers reflect the balance between theoretical yield and practical manufacturability inherent in this specific chemical pathway.
Q: How does the HCl salt formation step improve isoniazid purity?
A: The formation of isonicotinic ester hydrochloride allows for the removal of unreacted isonicotinic acid and 2-picolinic acid impurities, which have different solubility profiles in the ether-alcohol solvent system compared to the desired ester salt.
Q: What are the advantages of this method over direct condensation?
A: Unlike direct condensation which often suffers from high temperatures and incomplete reactions, this method utilizes a purified intermediate that significantly shortens the subsequent hydrazinolysis reaction time and avoids viscous reaction systems.
Q: Can this process be scaled for commercial API production?
A: Yes, the process uses common reagents like oxalyl chloride and ethanol, and operates at moderate temperatures (10-50°C for esterification, 60-100°C for hydrazinolysis), making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoniazid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the HCl salt intermediate method are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs capable of detecting trace impurities at the ppm level, guaranteeing that every batch of isoniazid meets or exceeds global pharmacopoeial requirements. Our commitment to quality ensures that your supply chain remains uninterrupted and compliant with the highest regulatory standards.
We invite you to collaborate with us to optimize your sourcing strategy for this critical anti-tuberculosis agent. By leveraging our technical capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven manufacturing competence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
