Revolutionizing 1,2-Cyclohexanediamine Production: A Deep Dive into the One-Pot Epoxy Cyclohexane Method
Revolutionizing 1,2-Cyclohexanediamine Production: A Deep Dive into the One-Pot Epoxy Cyclohexane Method
The global demand for high-purity 1,2-cyclohexanediamine has historically been constrained by complex supply chains and limited production methodologies, but recent technological advancements documented in patent CN115160146B offer a transformative solution for the fine chemical industry. This groundbreaking intellectual property details a highly efficient one-pot preparation method that utilizes cyclohexene oxide and liquid ammonia as primary feedstocks, bypassing the traditional bottlenecks associated with adiponitrile hydrogenation byproducts. By integrating a specialized catalytic system involving Raney metals and alkali cocatalysts, this process achieves exceptional conversion rates while operating under manageable industrial conditions. For R&D directors and procurement strategists, this innovation represents a critical opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality without the volatility of legacy supply networks. The following analysis dissects the technical merits and commercial implications of this novel synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 1,2-cyclohexanediamine has been plagued by significant structural inefficiencies and raw material dependencies that hinder scalability and cost-effectiveness. The predominant commercial source has been as a byproduct of hexamethylenediamine production via adiponitrile hydrogenation, a route where supply is strictly controlled by a handful of international conglomerates and is inherently limited by the primary product's market dynamics. Alternative synthetic pathways, such as the hydrogenation of o-phenylenediamine described in older patents, rely on expensive noble metal catalysts like ruthenium and costly aromatic starting materials, rendering them economically unviable for large-scale commodity production. Furthermore, previous attempts to utilize epoxy cyclohexane involved multi-step processes requiring sulfuric acid for dehydration, followed by alkaline dissociation and repeated ammonia treatments, which generated substantial quantities of hazardous waste salts and wastewater. These conventional methods not only suffer from low atom economy but also impose heavy environmental compliance burdens and high energy consumption due to the extensive purification steps required to isolate the final diamine product from complex reaction mixtures.
The Novel Approach
In stark contrast to these legacy technologies, the one-pot method disclosed in CN115160146B introduces a streamlined, continuous workflow that dramatically simplifies the manufacturing landscape for this valuable diamine. By employing liquid ammonia in a single charging step mixed directly with the catalyst and cocatalyst, the process facilitates an immediate ring-opening reaction of cyclohexene oxide to form 2-aminocyclohexanol, which subsequently undergoes in-situ hydroamination to yield the target 1,2-cyclohexanediamine. This telescoped approach eliminates the need for isolating the unstable amino-alcohol intermediate, thereby reducing processing time, equipment footprint, and the risk of product degradation during handling. The utilization of cyclohexene oxide, which is abundantly available as a byproduct of the mature cyclohexane oxidation industry, ensures a robust and cost-stable raw material base that is independent of the adiponitrile market fluctuations. Consequently, this novel approach offers a compelling value proposition for cost reduction in fine chemical manufacturing, enabling producers to achieve high purity specifications with significantly reduced operational expenditure and environmental impact.
Mechanistic Insights into Catalytic Hydroamination and Ring Opening
The core of this technological breakthrough lies in the precise orchestration of nucleophilic attack and catalytic hydrogenation within a single reactor vessel, driven by a synergistic catalyst system. The initial phase involves the ring-opening of the strained epoxide group in cyclohexene oxide by liquid ammonia, a reaction that is thermodynamically favorable but kinetically controlled by the presence of basic cocatalysts such as sodium methoxide or potassium hydroxide. These alkali species enhance the nucleophilicity of the ammonia, facilitating the attack on the epoxide ring at moderate temperatures ranging from 60°C to 100°C under oxygen-free conditions to prevent oxidative degradation. The result of this first stage is the formation of 2-aminocyclohexanol, an intermediate that is notoriously prone to self-polymerization if not managed correctly, which would otherwise lead to tar formation and reduced yields.
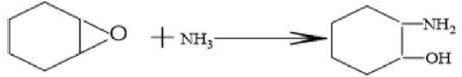
Following the formation of the amino-alcohol intermediate, the process transitions into a high-pressure hydroamination phase where hydrogen gas is introduced to the system at pressures between 2 MPa and 12 MPa. Under elevated temperatures of 180°C to 210°C, the catalyst—selected from robust options like Raney nickel, Raney cobalt, or supported palladium—activates the hydrogen to facilitate the substitution of the hydroxyl group with an amine group. This critical transformation converts the 2-aminocyclohexanol into the final 1,2-cyclohexanediamine product, effectively replacing the oxygen functionality with nitrogen without requiring harsh dehydrating agents. The catalyst plays a dual role here: it not only drives the hydrogenation but also suppresses the competitive auto-polymerization of the intermediate, ensuring that the reaction selectivity remains skewed heavily towards the desired diamine rather than oligomeric byproducts.
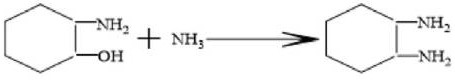
Furthermore, the absence of organic solvents in this reaction matrix is a deliberate mechanistic choice that enhances the concentration of reactants and minimizes side reactions. Comparative studies within the patent data reveal that introducing solvents like isopropanol can dilute the ammonia concentration and participate in competing aminolysis reactions, generating unwanted alkanolamines that complicate downstream purification. By maintaining a solvent-free environment where liquid ammonia serves as both reagent and medium, the process maximizes the collision frequency between the epoxide and the amine species, leading to the impressive yields of over 90% observed in the experimental examples. This mechanistic efficiency is paramount for achieving the high-purity 1,2-cyclohexanediamine required for sensitive applications in pharmaceutical synthesis and advanced polymer curing.
How to Synthesize 1,2-Cyclohexanediamine Efficiently
Implementing this one-pot synthesis strategy requires strict adherence to safety protocols regarding high-pressure hydrogen and anhydrous ammonia handling, alongside precise control of thermal profiles to manage the exothermic nature of the ring-opening step. The patent outlines a robust procedure where the catalyst is first immobilized in the reactor, followed by the introduction of liquid ammonia and the cocatalyst under an inert nitrogen atmosphere to eliminate oxygen risks. The cyclohexene oxide is then added dropwise to control the reaction rate and heat generation, ensuring that the initial formation of 2-aminocyclohexanol proceeds smoothly before the system is pressurized with hydrogen for the final conversion step. Detailed standardized synthesis steps, including specific molar ratios and purification parameters, are provided in the technical guide below to assist process engineers in replicating these high-yield results.
- Mix catalyst (e.g., Raney Nickel), liquid ammonia, and cocatalyst (e.g., sodium methoxide) in a reactor under oxygen-free conditions.
- Dropwise add cyclohexene oxide at 60-100°C and react until pressure stabilizes to form 2-aminocyclohexanol intermediate.
- Charge hydrogen (2-12 MPa) and heat to 180-210°C to convert the intermediate into 1,2-cyclohexanediamine, followed by dehydration and rectification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot epoxy cyclohexane methodology presents a strategic avenue for mitigating risk and optimizing the total cost of ownership for diamine intermediates. The shift away from adiponitrile-dependent routes decouples the supply of 1,2-cyclohexanediamine from the volatile hexamethylenediamine market, providing a more predictable and stable sourcing channel that is less susceptible to capacity constraints imposed by major nylon producers. Additionally, the elimination of solvent usage and the reduction of purification steps from multiple stages to a simple dehydration and rectification sequence drastically lower the utility consumption and waste disposal costs associated with production. These operational efficiencies translate directly into margin improvements and allow for more competitive pricing structures in the global marketplace without compromising on the stringent quality standards required by downstream users.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of low-cost, abundant raw materials and the removal of expensive auxiliary chemicals from the production bill of materials. By avoiding the use of sulfuric acid and the subsequent generation of inorganic salt waste, manufacturers can significantly reduce their expenditure on effluent treatment and regulatory compliance, which are often hidden but substantial costs in traditional chemical processing. Moreover, the high selectivity of the catalyst system minimizes the loss of valuable raw materials to byproducts, ensuring that a greater proportion of the input mass is converted into saleable product, thereby enhancing the overall material efficiency of the plant.
- Enhanced Supply Chain Reliability: Sourcing cyclohexene oxide is inherently more stable than relying on niche byproduct streams, as it is derived from the massive scale oxidation of cyclohexane, a cornerstone process in the nylon and phenol industries. This broad industrial base ensures that feedstock availability remains high even during periods of market fluctuation, reducing the risk of production stoppages due to raw material shortages. For supply chain heads, this means reducing lead time for high-purity 1,2-cyclohexanediamine deliveries and establishing a more resilient inventory strategy that can withstand external shocks in the petrochemical sector.
- Scalability and Environmental Compliance: The simplicity of the one-pot design facilitates easier scale-up from pilot plants to commercial production units, as it removes the complexity of transferring reactive intermediates between different vessels. The process generates minimal hazardous waste, aligning with increasingly strict global environmental regulations and sustainability goals, which is a critical factor for multinational corporations seeking green chemistry partners. The ability to operate with high atom economy and reduced energy intensity positions this technology as a future-proof solution for the commercial scale-up of complex pharmaceutical intermediates and specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis route, based on the detailed disclosures within the patent literature. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains or R&D pipelines. The answers provided reflect the specific advantages of the one-pot method over conventional alternatives, focusing on yield, purity, and operational safety.
Q: What are the primary advantages of the one-pot epoxy cyclohexane method over traditional adiponitrile byproduct routes?
A: The one-pot method utilizes cyclohexene oxide, a widely available byproduct of cyclohexanone oxidation, ensuring a stable and independent raw material source unlike the supply-constrained adiponitrile route. Furthermore, it eliminates complex multi-step purification processes involving sulfuric acid and salt formation, significantly reducing waste generation and operational costs.
Q: How does the catalyst system prevent the formation of polymeric byproducts?
A: The specific combination of Raney metals (Ni, Co, Cu) or supported noble metals with alkali metal alkoxide cocatalysts effectively inhibits the auto-polymerization of the 2-aminocyclohexanol intermediate. This selectivity ensures that the reaction pathway favors hydroamination to the diamine rather than oligomerization, resulting in yields exceeding 90%.
Q: Is solvent required for this synthesis process?
A: No, the process is designed to be solvent-free, utilizing liquid ammonia as both the reactant and the reaction medium. Comparative data indicates that adding solvents like isopropanol can dilute ammonia concentration and lead to side reactions, thereby lowering yield and increasing energy consumption during downstream separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Cyclohexanediamine Supplier
As the chemical industry evolves towards more sustainable and efficient manufacturing paradigms, NINGBO INNO PHARMCHEM stands at the forefront as a trusted partner capable of translating innovative patent technologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial volume is seamless and compliant with international quality standards. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,2-cyclohexanediamine meets the exacting requirements of pharmaceutical and electronic grade applications, thereby securing your production continuity.
We invite forward-thinking organizations to collaborate with us to leverage these advanced synthetic methods for their specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements, and ask for specific COA data and route feasibility assessments to validate the potential of this technology for your portfolio. Together, we can drive down costs and enhance the reliability of your supply chain through the adoption of next-generation chemical processes.
