Industrial Scale-Up of Caspofungin: A Safer, Non-Toxic Synthetic Route for Global Supply Chains
The pharmaceutical landscape for antifungal treatments has long relied on the efficacy of echinocandins, with Caspofungin standing as a cornerstone therapeutic agent. However, the historical manufacturing pathways for this critical molecule have been plagued by significant safety hazards, primarily due to the reliance on highly toxic and malodorous reagents. Patent CN102219833A introduces a paradigm-shifting methodology that fundamentally alters the risk profile of Caspofungin production. By replacing the conventional thiophenol-based stereochemical selection with safer heterocyclic alternatives like 2-mercaptobenzothiazole or 1-phenyl-5-sulfydryl-tetrazole, this invention offers a route that is not only chemically robust but also industrially viable from an Environmental, Health, and Safety (EHS) perspective. This technical insight report dissects the novel synthetic strategy detailed in the patent, highlighting its potential to redefine supply chain reliability and cost structures for global API manufacturers seeking a reliable Caspofungin supplier.
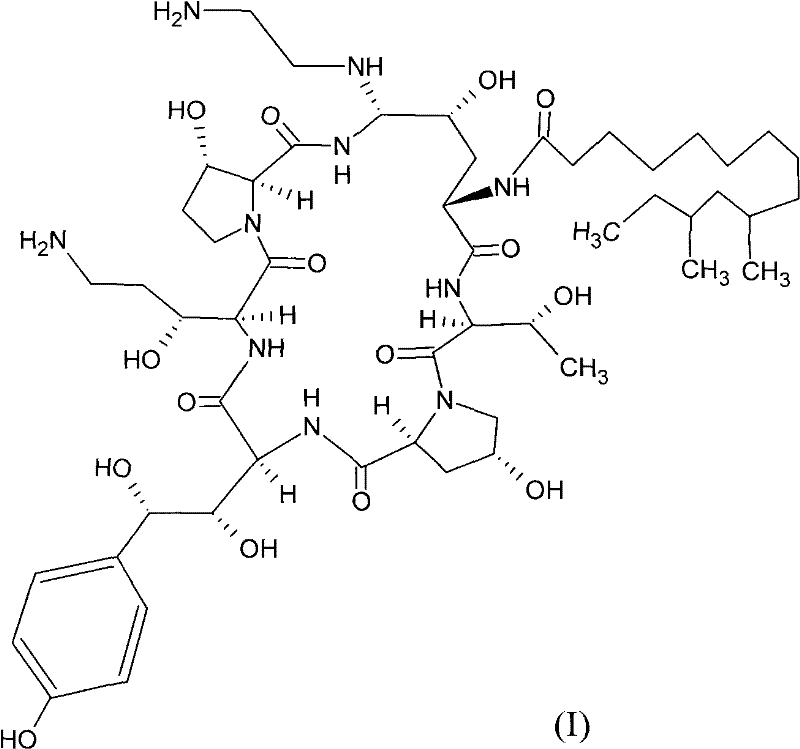
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Caspofungin from Pneumocandin B0 has depended heavily on the use of thiophenol as a stereochemical structure selecting agent. While chemically effective in forming the necessary cyclic boronate esters to protect the sensitive diol side chains, thiophenol presents severe logistical and safety challenges. It is classified as a hypertoxic chemical with an extremely low odor threshold, meaning even minute leaks can create unbearable working conditions and necessitate expensive containment infrastructure. Furthermore, the handling of thiophenol requires rigorous personal protective equipment (PPE) and specialized waste treatment protocols to neutralize its toxicity before discharge. These factors collectively inflate the operational expenditure (OPEX) of traditional manufacturing lines and introduce significant supply chain fragility, as regulatory scrutiny on volatile sulfur compounds continues to tighten globally.
The Novel Approach
The innovation disclosed in CN102219833A circumvents these legacy issues by utilizing solid, less volatile, and significantly less toxic thiol-containing heterocycles. Specifically, the patent details the successful application of 2-mercaptobenzothiazole and 1-phenyl-5-sulfydryl-tetrazole. These agents perform the same critical function of stereoselective protection but without the debilitating odor and acute toxicity profiles of thiophenol. This substitution allows for a much more streamlined operation where reaction vessels do not require the same level of hermetic sealing against vapor escape, and worker exposure risks are drastically minimized. The transition to these safer reagents represents a move towards 'Green Chemistry' principles within the fine chemical sector, aligning production capabilities with modern sustainability mandates while maintaining high chemical fidelity.
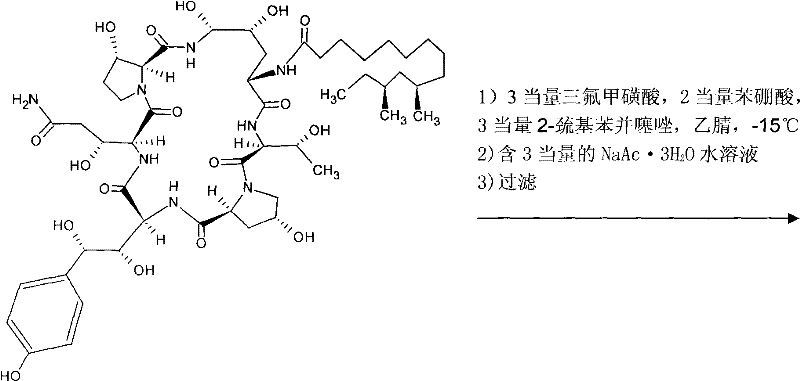
Mechanistic Insights into Boronic Acid-Mediated Stereochemical Protection
The core chemical transformation in this patent revolves around the formation of a cyclic boronate ester, which serves as a temporary protecting group for the vicinal diol system on the hexapeptide backbone of Pneumocandin B0. In the initial step, Pneumocandin B0 is reacted with phenylboronic acid in the presence of the novel stereochemical selecting agent and trifluoromethanesulfonic acid in anhydrous acetonitrile. The reaction is conducted at cryogenic temperatures, specifically maintained between -15°C and 0°C, to ensure kinetic control and prevent epimerization or degradation of the sensitive peptide bonds. The selecting agent coordinates with the boron center, stabilizing the transition state and ensuring that the protection occurs with the correct stereochemistry, which is vital for the biological activity of the final antifungal product. This step typically proceeds with high efficiency, with patent data indicating yields reaching up to 90% and purity levels exceeding 94%.
Following the protection step, the intermediate undergoes a reduction sequence to convert the amide or related functionality into the requisite amine found in Caspofungin. This is achieved through a silylation followed by reduction using a borane-tetrahydrofuran (THF) adduct. The use of borane-THF is a standard yet powerful reduction method that operates effectively under mild conditions (-5°C to 0°C). The subsequent workup involves careful acidification and purification, often utilizing nanofiltration membranes to concentrate the product while removing boron residues. The final step involves the displacement of the protecting group and side-chain modification using ethylenediamine (referred to as quadrol in the text context of substitution). This nucleophilic substitution is performed under strictly controlled pH conditions (5.0-5.5) and low temperatures (<20°C) to preserve the integrity of the macrocyclic lactone ring, ultimately yielding the target Caspofungin with high purity (up to 97%) and excellent overall yield.
How to Synthesize Caspofungin Efficiently
The synthesis of Caspofungin via this safer route involves a precise sequence of protection, reduction, and substitution steps that demand rigorous process control. The protocol begins with the suspension of Pneumocandin B0 and the safer selecting agent in acetonitrile, cooled to -15°C before the addition of activators. Following the formation of the protected intermediate, the process moves to a reduction phase in THF using borane complexes, requiring careful moisture exclusion. Finally, the deprotection and amination are completed using ethylenediamine under buffered aqueous conditions. For the detailed standardized operating procedures, stoichiometry, and specific workup parameters required for GMP manufacturing, please refer to the technical guide below.
- React Pneumocandin B0 with phenylboronic acid and a safe stereochemical selecting agent (2-mercaptobenzothiazole) in acetonitrile at -15°C to form the protected intermediate.
- Reduce the protected intermediate to the corresponding amine using a borane-THF adduct following silylation, maintaining strict temperature control.
- Perform the final substitution reaction with ethylenediamine (quadrol) under controlled pH and temperature to yield the target Caspofungin compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN102219833A offers tangible strategic benefits beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of EHS compliance. By eliminating thiophenol, facilities can avoid the substantial costs associated with scrubbing toxic sulfur vapors and managing hazardous waste streams. This directly translates to cost reduction in Caspofungin manufacturing, as the overhead for safety monitoring, specialized PPE, and insurance premiums is significantly lowered. Furthermore, the use of solid or less volatile reagents improves the reliability of raw material logistics, as storage and transport requirements are less stringent compared to liquid thiols.
- Cost Reduction in Manufacturing: The replacement of thiophenol with 2-mercaptobenzothiazole eliminates the need for expensive odor-control infrastructure and specialized toxic waste disposal services. This qualitative shift in reagent profile allows for a leaner operational model where capital expenditure on safety engineering is reduced, and the overall cost of goods sold (COGS) is optimized through improved process safety and reduced downtime associated with hazard management.
- Enhanced Supply Chain Reliability: Sourcing safer reagents reduces the risk of supply disruptions caused by regulatory bans or transport restrictions on hazardous materials. The reagents specified in this patent are commercially available and stable, ensuring a continuous flow of materials for production. Additionally, the high yields reported in the patent embodiments (consistently above 75-90% per step) indicate a robust process that minimizes material loss, thereby securing a more predictable output volume for downstream formulation.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common solvents like acetonitrile and THF which are easily recovered and recycled in standard distillation units. The absence of persistent toxic sulfur contaminants simplifies the effluent treatment process, making it easier for manufacturing sites to meet increasingly strict environmental discharge limits. This environmental compatibility ensures long-term operational continuity without the threat of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this safer Caspofungin synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN102219833A, providing a factual basis for evaluating the technology's fit within your existing production portfolio.
Q: Why is the new stereochemical selecting agent preferred over thiophenol?
A: The new agents, such as 2-mercaptobenzothiazole, are non-toxic and lack the severe foul odor associated with thiophenol, significantly improving workplace safety and reducing environmental compliance costs.
Q: What are the typical yields for this Caspofungin synthesis route?
A: According to the patent data, the process achieves high efficiency with yields around 90% for the protection step, 78% for the reduction, and 86% for the final substitution, ensuring robust material throughput.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the elimination of highly toxic reagents and the use of standard solvents like acetonitrile and THF make this route highly scalable and compliant with modern industrial safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caspofungin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the long-term viability of pharmaceutical supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one described in CN102219833A can be seamlessly transferred from the laboratory to the plant. We are committed to delivering high-purity Caspofungin and its intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to optimize your antifungal API supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance your product's market competitiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
