Advanced Palladium-Catalyzed Synthesis of Polysubstituted Conjugated Dienes for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of Polysubstituted Conjugated Dienes for Commercial Scale-up
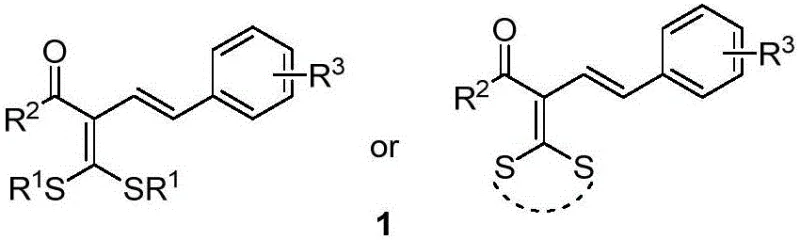
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective methodologies to access complex molecular architectures. A significant breakthrough in this domain is documented in patent CN111302990A, which discloses a novel synthetic route for preparing polysubstituted conjugated diene compounds. These compounds are not merely academic curiosities; they represent a class of vital intermediates possessing unique structural features and special reactivity profiles that make them indispensable for the construction of biologically active molecules. The patent highlights a transformative approach where α-halogenated dithioketals react with styrene derivatives through a free radical olefination reaction. This process is elegantly catalyzed by light-induced transition metal palladium, operating under remarkably mild conditions to construct the conjugated diene framework in a single synthetic step. For R&D directors and procurement specialists alike, this technology signals a shift towards streamlined manufacturing processes that reduce operational complexity while maintaining high standards of purity and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of conjugated diene compounds has been fraught with challenges that hinder large-scale commercial adoption. Traditional methodologies often rely on harsh reaction conditions, requiring elevated temperatures or strong bases that can compromise sensitive functional groups present in the substrate. Prior art, such as the work by Gaunt's group in 2005 utilizing palladium-catalyzed indole and butyl acrylate, or Loh's group's oxidative C-H alkenylation, while scientifically significant, often involve multi-step sequences or expensive reagents that drive up the cost of goods sold. Furthermore, conventional routes may suffer from poor regioselectivity or stereoselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. The reliance on stoichiometric oxidants or toxic heavy metals in older protocols also raises significant environmental and safety concerns, complicating waste disposal and regulatory compliance for manufacturing facilities. These limitations create bottlenecks in the supply chain, extending lead times and increasing the risk of production delays for downstream pharmaceutical applications.
The Novel Approach
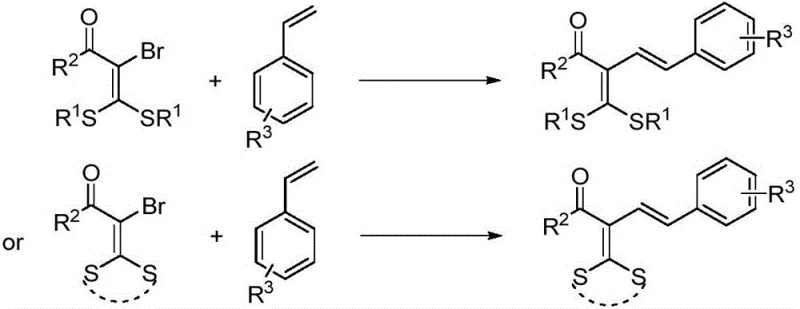
In stark contrast to these legacy methods, the technology described in CN111302990A offers a paradigm shift through its use of a visible-light-induced palladium catalytic system. This innovative strategy utilizes readily available α-halogenated dithioketals and styrenes as starting materials, reacting them under basic conditions to generate the target conjugated diene compound 1. The core of this advancement lies in the activation of the carbon-halogen bond via a radical mechanism facilitated by the palladium catalyst under blue LED irradiation. This allows the reaction to proceed at room temperature, eliminating the need for energy-intensive heating and reducing the thermal stress on the reactants. The generality of this method is demonstrated by its compatibility with a wide range of substituents, including halogens and alkyl groups, ensuring broad applicability across different chemical spaces. By consolidating the synthesis into a one-step procedure with high efficiency, this novel approach drastically simplifies the process flow, offering a clear pathway for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Light-Induced Pd-Catalyzed Radical Alkenylation
To fully appreciate the robustness of this synthetic method, one must delve into the mechanistic intricacies of the light-induced transition metal palladium catalysis. The reaction initiates with the coordination of the palladium catalyst, typically palladium acetate, with a bulky phosphine ligand such as XantPhos. This coordination sphere is crucial for stabilizing the active palladium species and facilitating the single-electron transfer processes required for radical generation. Upon irradiation with 15W blue LED light, the palladium complex interacts with the α-halogenated dithioketal substrate, promoting the homolytic cleavage of the carbon-bromine bond. This generates a reactive carbon-centered radical intermediate alongside a palladium species capable of engaging with the styrene olefin. The subsequent radical addition to the double bond of the styrene, followed by elimination or further catalytic turnover, constructs the new carbon-carbon double bond with high stereocontrol, predominantly favoring the E-isomer. This precise control over the reaction trajectory minimizes the formation of unwanted by-products, thereby enhancing the overall purity of the crude reaction mixture.
Furthermore, the choice of base and solvent plays a pivotal role in modulating the reaction kinetics and thermodynamics. The patent specifies the use of cesium carbonate as the base, which effectively neutralizes the acidic by-products generated during the catalytic cycle without inducing side reactions such as hydrolysis of the dithioketal moiety. Benzene is identified as the optimal solvent, providing a non-polar environment that favors the solubility of the organic substrates while supporting the radical propagation steps. The synergy between the photocatalytic activation and the transition metal catalysis ensures that the reaction proceeds efficiently even at room temperature, typically around 25°C. This mildness is particularly advantageous for scaling up the process, as it reduces the risk of thermal runaway and allows for the use of standard glass-lined reactors without specialized high-pressure or high-temperature equipment. The result is a highly reliable process that delivers consistent quality, essential for meeting the stringent specifications of the pharmaceutical industry.
How to Synthesize Polysubstituted Conjugated Dienes Efficiently
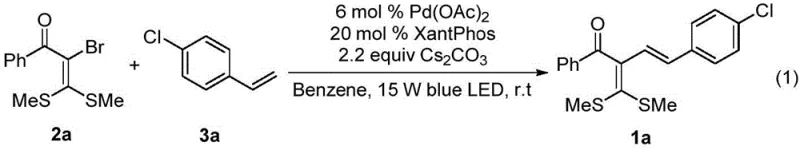
Implementing this cutting-edge synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The protocol outlined in the patent provides a clear blueprint for executing the radical alkenylation reaction. It begins with the careful preparation of the reaction vessel, ensuring an inert atmosphere to prevent quenching of the radical intermediates by oxygen. The precise stoichiometry of the reagents is critical; specifically, a molar ratio of 1:1.5 between the α-halogenated dithioketal and the styrene derivative is recommended to drive the reaction to completion. The catalyst loading is kept low, typically around 6 mol% of palladium acetate, supported by 20 mol% of the XantPhos ligand, which balances cost efficiency with catalytic activity. Following the reaction, standard workup procedures involving solvent removal and silica gel chromatography allow for the isolation of the pure product. For detailed operational instructions, please refer to the standardized guide below.
- Prepare the reaction mixture by weighing alpha-halogenated dithioketal, styrene derivative, palladium acetate catalyst, XantPhos ligand, and cesium carbonate base into a Schlenk flask under nitrogen atmosphere.
- Add benzene solvent to the mixture and stir the reaction under 15W blue LED irradiation at room temperature for approximately 12 hours to ensure complete conversion.
- Upon completion, remove volatile components under reduced pressure and purify the crude residue via silica gel column chromatography using petroleum ether and ethyl acetate to isolate the target diene.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The primary benefit lies in the significant simplification of the supply chain for raw materials. The starting materials, α-halogenated dithioketals and substituted styrenes, are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of supply disruption associated with exotic or proprietary reagents. Moreover, the elimination of harsh reaction conditions translates into lower energy consumption and reduced wear and tear on manufacturing equipment, contributing to substantial cost savings in facility operations. The high atom economy of the reaction, coupled with the ability to achieve high yields without extensive purification steps, means that less raw material is wasted, further driving down the cost per kilogram of the final intermediate. This efficiency is crucial for maintaining competitive pricing in the volatile market of pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily due to the replacement of expensive and complex multi-step sequences with a single-step catalytic process. By utilizing a low loading of palladium catalyst and avoiding the need for cryogenic conditions or high-pressure reactors, the capital expenditure and operational expenditure are drastically reduced. The use of common solvents like benzene and inexpensive bases like cesium carbonate ensures that the variable costs remain low. Additionally, the high selectivity of the reaction minimizes the formation of impurities, which reduces the burden on downstream purification processes such as recrystallization or preparative HPLC. This streamlined workflow allows manufacturers to produce high-purity conjugated dienes at a fraction of the cost associated with traditional methods, enhancing profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and scalability of this synthetic method. The reaction conditions are mild and tolerant to minor variations in temperature and mixing, making the process highly reproducible across different batch sizes, from grams to tons. The reliance on stable, shelf-stable reagents means that inventory management is straightforward, with no need for specialized storage conditions like deep freezing or strict moisture control. Furthermore, the short reaction time of approximately 12 hours allows for rapid turnover of batches, enabling manufacturers to respond quickly to fluctuating demand from downstream clients. This agility is a key differentiator in a market where lead times can often be a bottleneck for drug development timelines.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is facilitated by the use of standard photochemical reactors that are increasingly available in modern chemical plants. The absence of toxic heavy metal waste streams, beyond the recoverable palladium catalyst, aligns well with green chemistry principles and stringent environmental regulations. The ability to recycle the palladium catalyst further enhances the sustainability profile of the process. Moreover, the final products can be further derivatized into valuable heterocycles like thiophenes, adding value to the supply chain. This versatility ensures that the manufacturing capacity can be flexibly allocated to produce a variety of high-value intermediates, optimizing asset utilization and ensuring long-term supply continuity for partners.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent. These answers address common concerns regarding reaction scope, optimization, and downstream processing. Understanding these technical nuances is essential for integrating this method into existing production workflows and ensuring that the expected performance metrics are met. The following insights are derived directly from the exemplary embodiments and comparative examples documented in the intellectual property, providing a reliable foundation for decision-making.
Q: What are the optimal reaction conditions for this palladium-catalyzed alkenylation?
A: The optimal conditions involve using palladium acetate (6 mol%) with XantPhos ligand (20 mol%) and cesium carbonate (2.2 equiv) in benzene solvent, irradiated by 15W blue LED at room temperature.
Q: Can this method tolerate various functional groups on the styrene substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating substituents such as fluoro, chloro, trifluoromethyl, and methyl groups on the aromatic ring of the styrene.
Q: What is the potential downstream application of these conjugated diene products?
A: These functionalized conjugated diene structures serve as versatile precursors that can undergo further transformations, such as iodine-mediated cyclization to form valuable thiophene derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Conjugated Diene Compound Supplier
The technological potential of light-induced palladium catalysis for synthesizing conjugated dienes represents a significant opportunity for innovation in the fine chemical sector. At NINGBO INNO PHARMCHEM, we pride ourselves on being at the forefront of adopting such advanced synthetic methodologies to serve our global clientele. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped with the necessary photochemical reactors and rigorous QC labs to handle sensitive catalytic processes, guaranteeing that every batch meets stringent purity specifications required for pharmaceutical applications. We understand the critical nature of timeline and quality in drug development, and our team is committed to delivering excellence at every stage of the manufacturing process.
We invite you to explore how this efficient synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data for our conjugated diene intermediates and to discuss route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable source of high-quality chemical building blocks, backed by decades of expertise in process chemistry and a commitment to sustainable manufacturing practices. Let us help you accelerate your development timeline with our superior supply capabilities.
