Advanced Oxidation Technology for Commercial Scale-up of p-Cyanobenzoic Acid
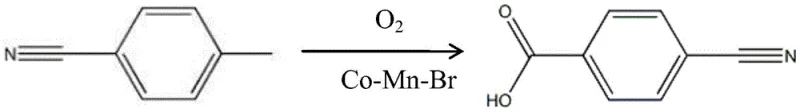
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly for anticoagulant therapies like the Factor Xa inhibitor Betrazan. Patent CN111574403A introduces a transformative preparation method for p-cyanobenzoic acid, a pivotal building block in this therapeutic class. This technology leverages a cobalt-manganese-bromine (Co-Mn-Br) catalytic system to facilitate the aerobic oxidation of p-methylbenzonitrile. Unlike traditional methods that struggle with environmental compliance and operational safety, this novel approach utilizes molecular oxygen as the primary oxidant in a glacial acetic acid solvent. The process is designed to deliver high-purity finished products through a streamlined refining protocol, addressing the urgent demand for scalable and eco-friendly manufacturing solutions in the fine chemical sector. By integrating this advanced oxidation technology, manufacturers can achieve a reliable p-cyanobenzoic acid supplier status, ensuring consistent quality for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of p-cyanobenzoic acid has been plagued by significant technical and environmental hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. One prevalent method involves the oxidation of 4-chloromethyl benzonitrile using concentrated nitric acid; while chemically straightforward, this route generates excessive byproducts and severe nitrogen oxide pollution, making three-waste treatment prohibitively expensive and environmentally unsustainable. Another approach utilizes p-toluic acid with formic acid, which introduces strong corrosivity hazards that complicate equipment maintenance and pose risks to operational safety. Furthermore, ammoxidation methods require extreme high-temperature and high-pressure conditions, necessitating specialized high-cost reactor infrastructure that is often impractical for standard fine chemical facilities. These legacy processes collectively suffer from low atom economy, difficult purification steps, and substantial environmental liabilities, rendering them unsuitable for modern green manufacturing standards.
The Novel Approach
The methodology disclosed in patent CN111574403A represents a paradigm shift by employing a liquid-phase aerobic oxidation catalyzed by a homogeneous Co-Mn-Br system. This route converts p-methylbenzonitrile directly to p-cyanobenzoic acid in a single reaction step, drastically simplifying the process flow compared to multi-step legacy syntheses. The use of oxygen gas as the oxidant not only reduces raw material costs but also produces water as the primary byproduct, significantly mitigating the environmental burden associated with nitric acid oxidation. The reaction conditions are moderate, typically operating between 100°C and 120°C, which lowers energy consumption and equipment stress. Moreover, the patent highlights a sophisticated mother liquor recycling strategy that allows for the reuse of solvents and catalysts, further enhancing the economic viability and sustainability of the process for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Co-Mn-Br Catalyzed Aerobic Oxidation
The core of this technological breakthrough lies in the synergistic action of the cobalt-manganese-bromine catalytic cycle, which facilitates the activation of molecular oxygen for the selective oxidation of the methyl group. In this system, cobalt and manganese acetates act as the primary redox mediators, cycling between different oxidation states to transfer electrons from the organic substrate to oxygen. The bromide ion serves as a crucial promoter, generating reactive bromine radicals that initiate the hydrogen abstraction from the benzylic position of p-methylbenzonitrile. This radical mechanism ensures high selectivity towards the carboxylic acid while preserving the sensitive nitrile functional group, which is often prone to hydrolysis under harsh acidic or basic conditions found in other methods. The precise molar ratios of the catalyst components, specifically a Co:Mn:Br ratio of approximately 2:2:1, are critical for maintaining the stability of the catalytic cycle and preventing the formation of over-oxidized byproducts or tar.
Impurity control is inherently managed through the specificity of the radical oxidation mechanism and the subsequent refining protocol. The reaction is monitored by tracking the residual mass fraction of p-methylbenzonitrile, with oxygen introduction ceased once levels drop below 0.5%, preventing over-oxidation and degradation of the product. Post-reaction, the crude product undergoes a meticulous purification sequence involving dissolution in water, pH adjustment to neutrality using sodium bicarbonate, and activated carbon decolorization. This sequence effectively removes trace metal catalysts, colored impurities, and unreacted starting materials. The final acidification step precipitates the high-purity p-cyanobenzoic acid, ensuring that the final impurity profile meets the stringent specifications required for API intermediate applications. This robust control strategy guarantees batch-to-batch consistency, a key requirement for regulatory compliance in pharmaceutical supply chains.
How to Synthesize p-Cyanobenzoic Acid Efficiently
Implementing this synthesis route requires precise control over reaction parameters and a disciplined approach to process execution to maximize yield and purity. The patent outlines a clear operational framework where p-methylbenzonitrile is charged into a glass-lined reactor with glacial acetic acid and the specific Co-Mn-Br catalyst mixture. The detailed standardized synthesis steps involve heating the mixture to an optimal temperature of 110°C while introducing oxygen at a controlled rate, followed by a specific workup procedure involving pH swings and filtration. Adhering to these parameters is essential for replicating the high yields reported in the patent examples. For a comprehensive breakdown of the exact reagent quantities, addition rates, and safety protocols, please refer to the standardized guide below.
- Mix p-methylbenzonitrile with glacial acetic acid solvent and a Co-Mn-Br catalyst system in a reaction vessel equipped with oxygen inlet.
- Heat the mixture to 100-120°C and introduce oxygen gas at a constant speed until the starting material is consumed.
- Filter the reaction mixture, refine the crude product via pH adjustment and decolorization, and recycle the mother liquor for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers compelling strategic benefits that extend beyond simple chemical conversion. The shift from corrosive, high-waste oxidants to a catalytic aerobic system fundamentally alters the cost structure of production. By eliminating the need for expensive and hazardous reagents like concentrated nitric acid or formic acid, the process reduces raw material procurement costs and minimizes the logistical complexities associated with handling dangerous goods. Furthermore, the ability to recycle the reaction mother liquor creates a closed-loop system that drastically reduces solvent consumption and waste disposal fees. These operational efficiencies translate into substantial cost savings and a more resilient supply chain, ensuring that high-purity pharmaceutical intermediates can be sourced reliably without the volatility associated with waste treatment compliance.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the efficiency of the catalytic system and the recycling of the mother liquor. By recovering and reusing the acetic acid solvent and the cobalt-manganese catalyst, the consumption of fresh raw materials per kilogram of product is significantly lowered. Additionally, the use of oxygen, which is readily available and inexpensive compared to stoichiometric oxidants, further drives down variable costs. The simplified one-step reaction also reduces labor hours and energy consumption associated with multi-step isolation and purification, leading to a leaner manufacturing cost base that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory constraints surrounding hazardous chemical transport and waste disposal. This method mitigates those risks by utilizing safer reagents and generating less hazardous waste, thereby reducing the likelihood of production stoppages due to environmental compliance issues. The moderate reaction conditions also mean that the process can be executed in a wider range of standard chemical reactors, increasing the pool of qualified manufacturing partners. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, as production is less susceptible to equipment bottlenecks or specialized infrastructure requirements.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is streamlined by the inherent safety and simplicity of the aerobic oxidation process. Unlike high-pressure ammoxidation, this method does not require specialized high-pressure vessels, allowing for easier scale-up in existing facilities. The reduction in nitrogen oxide emissions and corrosive waste aligns with increasingly strict global environmental regulations, future-proofing the supply chain against tightening green policies. This environmental stewardship not only protects the manufacturer from regulatory fines but also enhances the brand value for downstream pharmaceutical clients seeking sustainable sourcing options.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of the Co-Mn-Br catalytic system over traditional nitric acid oxidation?
A: The Co-Mn-Br system utilizes molecular oxygen as the oxidant, which significantly reduces the generation of nitrogen oxide waste gases and corrosive byproducts associated with nitric acid methods, leading to a cleaner production process and easier waste treatment.
Q: How does the mother liquor recycling process impact overall yield and cost?
A: Recycling the reaction mother liquor allows for the recovery of unreacted solvents and catalysts. Data indicates that reusing the mother liquor can further improve product yield and purity while substantially reducing raw material consumption and waste disposal costs.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process operates at moderate temperatures (100-120°C) and atmospheric or low pressure using oxygen, avoiding the extreme high-pressure conditions of ammoxidation methods, which makes it highly scalable and safer for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Cyanobenzoic Acid Supplier
The technological potential of the Co-Mn-Br catalyzed oxidation route for p-cyanobenzoic acid is immense, offering a pathway to high-purity intermediates essential for next-generation anticoagulants. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of p-cyanobenzoic acid meets the exacting standards required for pharmaceutical synthesis. We are committed to delivering consistent quality and supply stability, positioning ourselves as a strategic partner for your long-term growth.
We invite you to explore how this advanced synthesis method can optimize your supply chain and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can support your production goals with efficiency and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
