Advanced Enzymatic Synthesis of P-Cyanobenzoic Acid for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more sustainable and efficient routes for synthesizing critical intermediates, particularly for hemostatic agents like p-aminomethyl benzoic acid. A significant breakthrough in this domain is documented in patent CN107641622B, which discloses a novel series of nitrilases capable of selectively hydrolyzing terephthalonitrile into p-cyanobenzoic acid with exceptional efficiency. This technology represents a paradigm shift from traditional chemical synthesis, leveraging specific enzymes derived from diverse biological sources such as Pantoea sp., Arabidopsis thaliana, and Acidovorax facilis. By utilizing these biocatalysts, manufacturers can achieve conversion rates exceeding 99 percent under remarkably mild conditions, addressing long-standing challenges related to environmental pollution and reaction severity. For R&D directors and procurement specialists, this patent offers a compelling pathway to secure a reliable pharmaceutical intermediate supplier while drastically simplifying the production workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of p-cyanobenzoic acid has relied heavily on chemical methodologies that pose significant operational and environmental hazards. The Sandmeyer process, which utilizes p-aminobenzoic acid as a starting material, involves the use of highly toxic cyanating reagents and generates substantial hazardous waste streams that require costly disposal protocols. Similarly, ammoxidation processes using p-formylbenzoic acid often demand severe reaction conditions, including high temperatures and pressures, which increase energy consumption and equipment maintenance costs. Furthermore, palladium-catalyzed cyanation routes, while effective, introduce the risk of heavy metal contamination in the final product, necessitating complex and expensive purification steps to meet stringent pharmaceutical purity specifications. These conventional methods also suffer from relatively low atom economy and poor selectivity, often leading to the formation of unwanted by-products that complicate downstream processing and reduce overall yield.
The Novel Approach
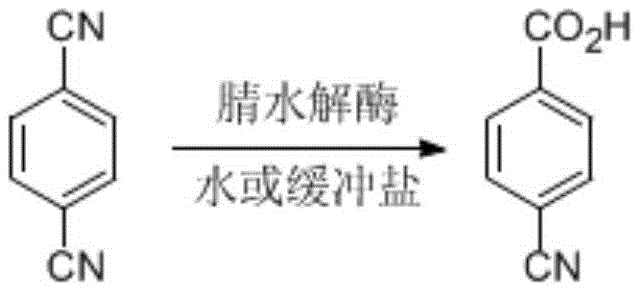
In stark contrast, the biocatalytic approach detailed in the patent utilizes engineered nitrilases to perform a highly selective mono-hydrolysis of terephthalonitrile. This enzymatic route operates in aqueous media at moderate temperatures ranging from 20 to 50 degrees Celsius and a pH between 6.0 and 10.0, effectively eliminating the need for organic solvents and extreme reaction parameters. The use of resting cells from genetically modified E. coli allows for high catalyst loading and stability, enabling the process to handle substrate concentrations as high as 100 g/L, which is a massive improvement over earlier biocatalytic attempts that struggled with concentrations around 25 mmol/L. This novel approach not only ensures a cleaner reaction profile with minimal by-product formation but also significantly reduces the environmental footprint of the manufacturing process, aligning perfectly with modern green chemistry principles and regulatory expectations for sustainable production.
Mechanistic Insights into Nitrilase-Catalyzed Selective Hydrolysis
The core of this technological advancement lies in the specific catalytic mechanism of the disclosed nitrilases, such as N1 derived from Pantoea sp. and N3 from Acidovorax facilis. These enzymes function by attacking the nitrile group of the terephthalonitrile molecule, facilitating the addition of a water molecule to convert one cyano group into a carboxylic acid while leaving the other cyano group intact. This regioselectivity is crucial because over-hydrolysis to terephthalic acid is a common side reaction in less specific systems. The patent describes the cloning and expression of genes encoding these enzymes in E. coli hosts, optimizing codon usage to ensure high-level protein expression. The resulting resting cells act as efficient biocatalysts, where the enzyme's active site precisely orientates the substrate to favor the formation of p-cyanobenzoic acid. This precision minimizes the formation of impurities, thereby reducing the burden on purification units and ensuring a high-purity product stream directly from the reactor.
Furthermore, the stability of these nitrilases under operational conditions contributes significantly to the robustness of the process. The patent data indicates that the enzymes maintain high activity across a broad pH range, allowing for flexibility in buffer selection, such as using sodium phosphate or Tris-HCl systems. The mechanism also benefits from the use of whole resting cells, which protect the enzyme from denaturation and allow for easier separation of the biocatalyst from the reaction mixture via simple centrifugation. This structural integrity of the biocatalyst ensures consistent performance over extended reaction times, typically completing conversions within 2 to 24 hours depending on the substrate load. For technical teams, understanding this mechanism highlights the feasibility of scaling the process without the fear of rapid enzyme deactivation, a common pitfall in other biocatalytic systems.
How to Synthesize P-Cyanobenzoic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or commercial setting. The process begins with the fermentation of the engineered bacterial strains, followed by the preparation of resting cells which are then suspended in a buffered aqueous solution. The substrate, terephthalonitrile, is introduced either as a suspension or dissolved in a co-solvent like DMSO to enhance solubility, allowing the enzymatic reaction to proceed efficiently. Detailed standardized synthetic steps see the guide below.
- Cultivate genetically engineered E. coli strains expressing specific nitrilases (N1-N6) in fermentation media with inducers like IPTG.
- Harvest resting cells via centrifugation and resuspend them in a phosphate buffer solution maintained at pH 6.0-10.0.
- Add terephthalonitrile substrate to the buffer mixture and react at 20-50°C until conversion is complete, followed by acidification and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nitrilase-based technology offers profound strategic advantages that extend beyond mere technical feasibility. The shift from chemical catalysis to biocatalysis fundamentally alters the cost structure and risk profile of producing p-cyanobenzoic acid. By eliminating the reliance on precious metal catalysts like palladium and toxic reagents, companies can insulate themselves from volatile raw material markets and reduce the regulatory burden associated with handling hazardous substances. The high substrate tolerance of the new enzymes means that reactors can be run at much higher concentrations, effectively increasing throughput per batch without requiring larger capital investment in equipment. This intensification of the process leads to significant reductions in solvent usage, water consumption, and waste generation, translating directly into lower operational expenditures and a more sustainable supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes the need for complex and costly metal scavenging steps, which are mandatory in traditional palladium-catalyzed routes to meet pharmaceutical safety standards. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high conversion rates minimize the loss of raw materials to by-products. The simplicity of the downstream processing, involving merely acidification and filtration, further lowers labor and utility costs compared to multi-step chemical extractions. These factors combined create a leaner manufacturing process that delivers substantial cost savings without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Sourcing high-purity pharmaceutical intermediates is often complicated by the limited number of suppliers capable of meeting strict quality controls. This biocatalytic route utilizes readily available biological materials and standard fermentation infrastructure, making it easier to establish multiple production sites or qualify secondary suppliers. The robustness of the engineered strains ensures consistent batch-to-batch quality, reducing the risk of supply disruptions caused by failed batches or off-spec products. Furthermore, the reduced dependency on specialized chemical reagents, which may be subject to export controls or supply shortages, enhances the overall resilience of the supply chain against global market fluctuations.
- Scalability and Environmental Compliance: Scaling up biocatalytic processes is generally more straightforward than scaling exothermic chemical reactions, as the heat generation is manageable and the reaction kinetics are predictable. The aqueous nature of the reaction medium simplifies waste treatment, as the effluent is largely biodegradable and free from heavy metals or persistent organic pollutants. This aligns with increasingly stringent environmental regulations, reducing the risk of fines or shutdowns due to non-compliance. The ability to operate at high substrate concentrations also means that the physical footprint of the production facility can be optimized, allowing for greater production capacity within existing infrastructure constraints.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrilase technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this route into your current manufacturing portfolio.
Q: What are the advantages of this nitrilase method over traditional chemical synthesis?
A: Unlike traditional Sandmeyer or palladium-catalyzed methods which require toxic cyanating reagents and harsh conditions, this enzymatic process operates under mild temperatures (20-50°C) and neutral pH, eliminating heavy metal contamination risks.
Q: What is the substrate tolerance of the disclosed nitrilases?
A: The patented resting cells demonstrate high substrate tolerance, capable of catalyzing concentrations up to 100 g/L or even 200 g/L, which is significantly higher than previous biocatalytic reports that were limited to roughly 25 mmol/L.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes robust engineered E. coli strains and simple downstream processing (acidification and filtration), making it highly scalable for commercial production of hemostatic agent intermediates without complex purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable P-Cyanobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route described in patent CN107641622B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications required for hemostatic agent manufacturing. We are committed to leveraging cutting-edge biocatalytic technologies to deliver superior value to our global partners.
We invite you to collaborate with us to explore the full commercial potential of this innovative synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the next generation of pharmaceutical products.
