Industrial Scale Synthesis of Optically Active 2-(2-Fluorobiphenyl-4-yl)propionic Acid via Nickel Catalysis
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective routes for producing high-value anti-inflammatory agents. Patent CN108026014B discloses a groundbreaking preparation method for optically active 2-(2-fluorobiphenyl-4-yl)propionic acid, a critical intermediate for non-steroidal anti-inflammatory drugs (NSAIDs). This technology represents a significant leap forward from traditional resolution methods, offering a direct asymmetric synthesis pathway that eliminates the inherent yield losses associated with racemic separation. By leveraging a specialized nickel-catalyzed cross-coupling strategy, this process addresses long-standing challenges in stereocontrol and operational efficiency. For R&D directors and procurement specialists, understanding this methodology is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with reduced environmental impact and optimized production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active 2-(2-fluorobiphenyl-4-yl)propionic acid has relied heavily on the optical resolution of racemic mixtures. Conventional techniques often involve forming salts with optically active amines followed by repeated recrystallization steps. This approach is fundamentally inefficient, with theoretical maximum yields capped at 50% and practical recovery rates often dropping as low as 60% due to solubility losses during multiple purification cycles. Furthermore, alternative asymmetric synthesis routes reported in prior art, such as asymmetric hydrogenation of acrylic acid derivatives, frequently necessitate the use of expensive and toxic rhodium catalysts. Other methods utilizing cobalt or iron catalysts often demand impractical cryogenic reaction conditions ranging from -80°C to -40°C, which impose severe energy burdens and engineering constraints on large-scale manufacturing facilities. These limitations collectively result in high production costs, complex waste streams, and supply chain vulnerabilities.
The Novel Approach
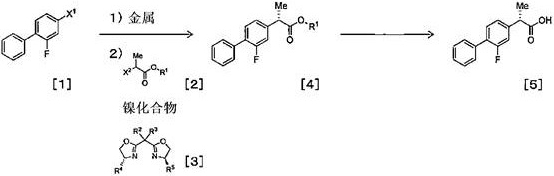
The methodology described in CN108026014B introduces a highly efficient nickel-catalyzed asymmetric Kumada-type coupling reaction that circumvents these historical bottlenecks. Instead of resolving a racemate, this process constructs the chiral center directly during the carbon-carbon bond formation step. The core innovation lies in the use of a catalytic amount of a nickel compound combined with a specific chiral bisoxazoline ligand. This system enables the coupling of an organometallic reagent derived from 4-bromo-2-fluorobiphenyl with an alpha-halo ester under remarkably mild conditions. Unlike previous methods requiring deep freezing, this novel approach operates effectively at temperatures between 0°C and 25°C. This shift not only simplifies reactor requirements but also drastically reduces energy consumption. The ability to utilize readily available starting materials and achieve high stereoselectivity without precious metals makes this a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Coupling
The success of this synthesis hinges on the precise interplay between the nickel catalyst and the chiral ligand environment. The reaction initiates with the formation of an organomagnesium or organozinc reagent from the biphenyl halide precursor. This nucleophilic species then undergoes transmetallation with the nickel catalyst, which is coordinated by the chiral bisoxazoline ligand. The steric bulk and electronic properties of the ligand create a highly defined chiral pocket around the nickel center. When the alpha-halo ester substrate enters this coordination sphere, the ligand dictates the facial selectivity of the oxidative addition and subsequent reductive elimination steps. This ensures that the new carbon-carbon bond is formed with a specific spatial arrangement, yielding the desired (S) or (R) enantiomer with high fidelity. The use of zinc additives further modulates the reactivity of the organometallic species, suppressing side reactions such as homocoupling or beta-hydride elimination that often plague nickel-catalyzed processes.
Impurity control is another critical aspect where this mechanism excels. In traditional Grignard reactions, the high reactivity of the organomagnesium species can lead to indiscriminate attack on various functional groups. However, the transmetallation to zinc or the specific modulation by the nickel-ligand complex softens the nucleophile, enhancing chemoselectivity. The patent data indicates that by optimizing the ratio of the nickel compound to the chiral ligand, manufacturers can minimize the formation of the undesired enantiomer and other structural byproducts. For instance, specific embodiments demonstrate that using a nickel loading as low as 0.03 mol% alongside 0.036 mol% of the chiral ligand still maintains excellent conversion and stereoselectivity. This precision in catalytic turnover not only boosts yield but also simplifies downstream purification, ensuring the final product meets stringent pharmaceutical purity specifications without extensive chromatographic separation.
How to Synthesize 2-(2-Fluorobiphenyl-4-yl)propionic Acid Efficiently
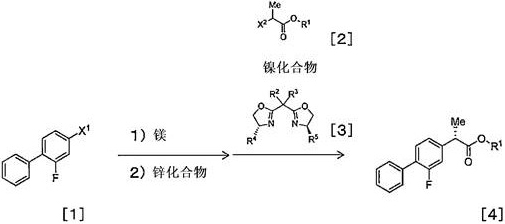
Implementing this synthesis requires careful attention to the preparation of the organometallic reagent and the maintenance of an inert atmosphere to prevent catalyst deactivation. The process generally begins with the activation of magnesium metal and the slow addition of the aryl bromide in a dry ether solvent like THF. Once the Grignard reagent is formed, it may be treated with a zinc salt to generate the corresponding organozinc species, which offers enhanced compatibility with the ester functionality. The coupling step involves the slow addition of this reagent to a mixture containing the alpha-halo ester, the nickel catalyst, and the chiral ligand at controlled low temperatures. Following the coupling, the resulting ester intermediate is subjected to acidic hydrolysis to reveal the free carboxylic acid. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility.
- Preparation of Organometallic Reagent: React 4-bromo-2-fluorobiphenyl with magnesium in THF to form a Grignard reagent, optionally followed by transmetallation with zinc chloride or bromide.
- Asymmetric Coupling: React the organometallic reagent with an alpha-halo ester (e.g., tert-butyl 2-bromopropionate) in the presence of a catalytic amount of a nickel compound (e.g., NiCl2·DME) and a chiral bisoxazoline ligand at 0°C to 25°C.
- Hydrolysis and Purification: Convert the resulting optically active ester intermediate into the final acid form using acidic conditions (e.g., formic acid) followed by recrystallization to achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this nickel-catalyzed protocol offers tangible strategic benefits beyond mere technical elegance. The most immediate impact is seen in the raw material and catalyst cost structure. By eliminating the need for expensive rhodium or palladium catalysts and replacing them with abundant nickel salts, the direct material cost is significantly reduced. Furthermore, the drastic reduction in catalyst loading—down to trace levels in optimized runs—means that the cost contribution of the chiral ligand, often a pricey component, is minimized per kilogram of product. This efficiency translates directly into a more competitive pricing model for the final intermediate, allowing pharmaceutical companies to better manage their COGS (Cost of Goods Sold) without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling requirements represents a massive operational expenditure saving. Conventional asymmetric syntheses often require maintaining reactors at -78°C for extended periods, demanding specialized refrigeration equipment and high energy inputs. By shifting the operating window to 0°C to 25°C, this process allows for the use of standard chilled water or even ambient temperature control in some phases. Additionally, the high atom economy of the coupling reaction and the avoidance of resolution steps (which discard half the material) mean that less raw material is required to produce the same amount of active ingredient. The simplified purification process, driven by high selectivity, reduces solvent consumption and waste disposal costs, further enhancing the overall economic viability of the production line.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for exotic catalysts or resolving agents introduces significant risk. This method relies on commodity chemicals like nickel chloride and magnesium, which are widely available from multiple global vendors, ensuring supply continuity even during market fluctuations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supply or environmental conditions, leading to more consistent batch-to-batch quality. For supply chain planners, this reliability reduces the need for excessive safety stock and minimizes the risk of production delays caused by catalyst shortages or failed batches due to sensitive reaction parameters.
- Scalability and Environmental Compliance: Scaling cryogenic processes from the lab to multi-ton production is notoriously difficult and hazardous. The mild thermal profile of this nickel-catalyzed route facilitates seamless scale-up from pilot plants to commercial manufacturing suites. The reduced use of heavy metals aligns with increasingly strict environmental regulations regarding residual metals in pharmaceutical products. Lower catalyst loading simplifies the metal scavenging steps required before the API can be formulated, reducing the chemical footprint of the manufacturing process. This alignment with green chemistry principles not only aids in regulatory approval but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of what partners can expect when adopting this technology. Understanding these nuances is vital for making informed decisions about process validation and vendor qualification.
Q: What is the typical catalyst loading for this nickel-catalyzed process?
A: The process utilizes extremely low catalyst loadings, with the nickel compound used at 0.03 to 1.00 mol% and the chiral ligand at 0.036 to 1.20 mol% relative to the substrate, significantly reducing heavy metal residue concerns.
Q: What optical purity can be achieved with this method?
A: The method consistently produces the target compound with high enantiomeric excess, typically achieving greater than 90% ee, and up to 99% ee after optimization and recrystallization steps.
Q: Does this process require cryogenic temperatures?
A: No, unlike conventional asymmetric methods requiring -60°C to -80°C, this novel protocol operates efficiently at much milder temperatures ranging from 0°C to 25°C, facilitating easier industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Fluorobiphenyl-4-yl)propionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of air- and moisture-sensitive nickel catalysis, ensuring that the high optical purity and yield demonstrated in the lab are faithfully reproduced at scale. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical intermediates, including comprehensive analysis of enantiomeric excess and residual metal content.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to a secure, scalable, and cost-efficient source of this critical intermediate. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation that reflects the economic advantages of this next-generation manufacturing process.
