Advanced Manufacturing of Flurbiprofen Intermediates via Novel Decarboxylative Coupling Technology
Advanced Manufacturing of Flurbiprofen Intermediates via Novel Decarboxylative Coupling Technology
The pharmaceutical industry is constantly seeking more efficient, sustainable, and cost-effective pathways for the production of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), with Flurbiprofen standing out as a critical therapeutic agent for rheumatoid arthritis and osteoarthritis. Patent CN101973869A introduces a groundbreaking methodology that fundamentally reshapes the synthetic landscape for this valuable compound by leveraging advanced organometallic catalysis. This technical insight report analyzes the proprietary decarboxylative coupling strategy disclosed in the patent, highlighting its potential to revolutionize supply chain stability and manufacturing economics for global pharmaceutical partners. By integrating palladium-catalyzed transformations with streamlined downstream processing, this approach offers a robust alternative to legacy synthesis routes that have long plagued manufacturers with excessive step counts and environmental liabilities. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for securing a competitive edge in the generic and branded drug markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Flurbiprofen has been hindered by convoluted reaction sequences that involve multiple protection and deprotection steps, leading to significant yield erosion and increased operational complexity. Traditional routes often rely on Friedel-Crafts acylations or multi-step arylations that require harsh reaction conditions, expensive reagents, and generate substantial quantities of hazardous waste streams that demand costly disposal protocols. These legacy methods frequently suffer from poor atom economy, where a large fraction of the starting material mass ends up as waste rather than incorporated into the final active pharmaceutical ingredient (API). Furthermore, the reliance on stoichiometric amounts of reactive intermediates often necessitates rigorous safety controls and specialized equipment, driving up capital expenditure and limiting the flexibility of production facilities to respond to market demand fluctuations. The cumulative effect of these inefficiencies is a higher cost of goods sold (COGS) and a longer time-to-market for new formulations, creating a strategic vulnerability for supply chain managers tasked with maintaining continuous availability of critical pain management medications.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in patent CN101973869A presents a highly elegant and concise three-step synthesis that dramatically simplifies the manufacturing workflow. The core innovation lies in the direct palladium-catalyzed decarboxylative coupling of 4-halo-2-fluorobiphenyl with cyanoacetate derivatives, a transformation that constructs the critical carbon-carbon bond with exceptional precision and efficiency. 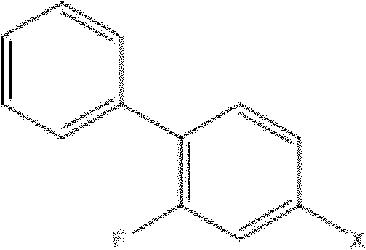
 This reaction bypasses the need for pre-functionalized organometallic reagents like Grignard or organolithium species, which are notoriously sensitive to moisture and air, thereby enhancing process robustness and safety. The subsequent steps involve a straightforward methylation (if required) and a final hydrolysis, both of which can be performed under relatively mild conditions using common industrial reagents. This reduction in synthetic complexity not only accelerates the production timeline but also minimizes the accumulation of impurities, resulting in a crude product profile that is significantly easier to purify to meet stringent pharmacopeial standards.
This reaction bypasses the need for pre-functionalized organometallic reagents like Grignard or organolithium species, which are notoriously sensitive to moisture and air, thereby enhancing process robustness and safety. The subsequent steps involve a straightforward methylation (if required) and a final hydrolysis, both of which can be performed under relatively mild conditions using common industrial reagents. This reduction in synthetic complexity not only accelerates the production timeline but also minimizes the accumulation of impurities, resulting in a crude product profile that is significantly easier to purify to meet stringent pharmacopeial standards.
Mechanistic Insights into Pd-Catalyzed Decarboxylative Coupling
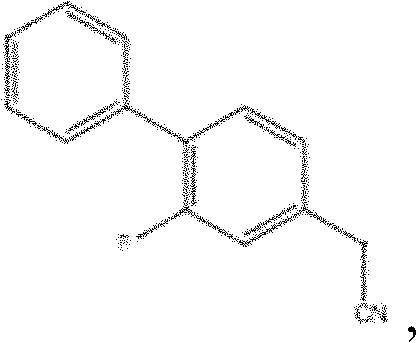
The heart of this technological advancement is the sophisticated catalytic cycle driven by palladium complexes in conjunction with bulky organophosphorus ligands. The mechanism initiates with the oxidative addition of the aryl halide to the low-valent palladium center, followed by the coordination and subsequent decarboxylation of the cyanoacetate derivative. This decarboxylation step is particularly crucial as it generates a nucleophilic carbon species in situ, which then undergoes transmetallation and reductive elimination to forge the desired biaryl linkage while releasing carbon dioxide as the sole gaseous byproduct. The selection of ligands, such as the Xantphos derivatives or bulky biaryl phosphines mentioned in the patent examples, plays a pivotal role in stabilizing the active catalytic species and preventing the formation of palladium black, which would otherwise lead to catalyst deactivation and reduced turnover numbers. 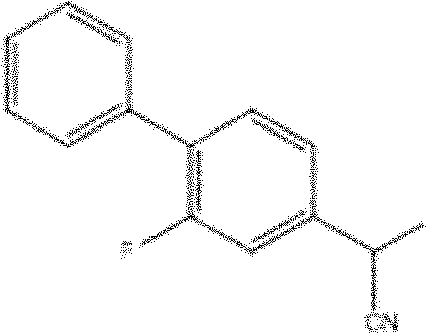
 From an impurity control perspective, this mechanism is inherently cleaner because it avoids the use of excess strong bases or cryogenic temperatures that often promote side reactions like homocoupling or over-alkylation. The patent specifies catalyst loadings as low as 0.1% to 0.4% mol, indicating a highly active system that minimizes the residual metal burden in the final API, a critical quality attribute for regulatory approval. Furthermore, the tolerance of the catalytic system to various functional groups allows for flexibility in substrate selection, enabling manufacturers to optimize raw material sourcing based on regional availability and cost without compromising reaction performance.
From an impurity control perspective, this mechanism is inherently cleaner because it avoids the use of excess strong bases or cryogenic temperatures that often promote side reactions like homocoupling or over-alkylation. The patent specifies catalyst loadings as low as 0.1% to 0.4% mol, indicating a highly active system that minimizes the residual metal burden in the final API, a critical quality attribute for regulatory approval. Furthermore, the tolerance of the catalytic system to various functional groups allows for flexibility in substrate selection, enabling manufacturers to optimize raw material sourcing based on regional availability and cost without compromising reaction performance.
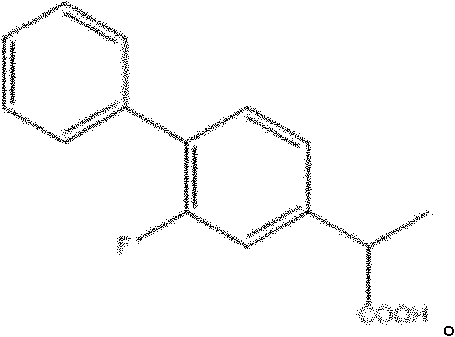
Following the coupling event, the hydrolysis of the nitrile intermediate to the corresponding carboxylic acid is executed with equal attention to process efficiency and safety. The patent details two viable pathways for this transformation: acid-catalyzed hydrolysis using mineral acids like sulfuric or hydrochloric acid, or base-mediated hydrolysis followed by acidification. Both methods proceed through well-understood mechanisms involving the hydration of the nitrile to an amide and subsequent cleavage to the acid, ensuring high conversion rates and minimal formation of difficult-to-remove side products. The ability to choose between acidic or basic conditions provides process chemists with the flexibility to tailor the workup procedure to the specific solubility characteristics of the intermediates, facilitating efficient isolation and crystallization. This level of control over the final step is essential for achieving the high purity levels required for pharmaceutical grade Flurbiprofen, ensuring that the final product meets the rigorous specifications for assay, related substances, and residual solvents mandated by global health authorities.
How to Synthesize Flurbiprofen Efficiently
Implementing this novel synthesis route requires careful attention to reaction parameters and reagent quality to maximize yield and reproducibility on a commercial scale. The process begins with the preparation of the reaction vessel under inert atmosphere to protect the sensitive palladium catalyst from oxidation, followed by the sequential addition of the aryl halide, cyanoacetate salt, ligand, and solvent. Detailed standard operating procedures for temperature ramping, agitation rates, and quenching protocols are essential to ensure consistent batch-to-batch performance and to mitigate any risks associated with exothermic events during the coupling or hydrolysis phases.
- Perform a palladium-catalyzed decarboxylative coupling between 4-halo-2-fluorobiphenyl and cyanoacetate derivatives using organophosphorus ligands.
- If using acetonitrile derivatives, conduct a methylation reaction with methyl halides in the presence of strong base hydrides.
- Complete the synthesis via hydrolysis of the nitrile intermediate using either strong acid or strong base conditions to yield Flurbiprofen.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages that extend far beyond simple technical feasibility. The primary value proposition lies in the drastic simplification of the supply chain, as the process relies on readily available commodity chemicals such as 4-halo-2-fluorobiphenyl and simple cyanoacetates, rather than exotic or custom-synthesized building blocks that are prone to supply disruptions. This shift towards commoditized raw materials significantly enhances supply security and reduces the risk of price volatility associated with niche intermediates, allowing for more accurate long-term budget forecasting and contract negotiation. Moreover, the reduction in the number of unit operations directly translates to lower utility consumption and reduced labor hours per kilogram of product, driving down the overall manufacturing cost structure without the need for aggressive margin compression.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the use of ultra-low catalyst loadings fundamentally alter the cost equation for Flurbiprofen production. By removing the need for stoichiometric organometallic reagents and cryogenic cooling infrastructure, manufacturers can achieve substantial savings in both raw material procurement and capital equipment maintenance. The high atom economy of the decarboxylative coupling ensures that a greater proportion of purchased raw materials is converted into saleable product, minimizing waste disposal fees and maximizing resource utilization efficiency. Additionally, the simplified purification requirements reduce the consumption of chromatography media and solvents, further contributing to a leaner and more cost-effective manufacturing operation that can withstand competitive pricing pressures in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes directly to improved supply chain resilience and reliability. Because the reaction conditions are less sensitive to moisture and oxygen compared to traditional Grignard-based routes, the risk of batch failures due to environmental factors is significantly mitigated. This reliability allows for tighter production scheduling and reduced safety stock requirements, freeing up working capital and warehouse space. Furthermore, the use of common solvents like toluene, DMF, or mesitylene ensures that solvent supply chains remain stable even during periods of global petrochemical market fluctuation. The ability to source key starting materials from multiple qualified vendors reduces single-source dependency, providing procurement teams with greater leverage and flexibility in managing their supplier portfolios.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant leap forward in sustainable manufacturing practices. The generation of only inorganic salts and carbon dioxide as byproducts aligns perfectly with increasingly stringent global environmental regulations and corporate sustainability goals. The absence of heavy metal waste streams and toxic organic solvents simplifies wastewater treatment and reduces the facility's environmental footprint. This green chemistry profile not only facilitates regulatory approvals in environmentally conscious markets but also enhances the brand reputation of the manufacturer as a responsible corporate citizen. The scalability of the process is further supported by the use of standard reactor types and moderate temperature ranges, allowing for seamless technology transfer from pilot plant to multi-ton commercial production scales without the need for specialized high-pressure or low-temperature equipment.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of answers based on the detailed disclosures within the patent literature. These responses cover critical aspects ranging from catalyst recovery to impurity profiling, providing clarity for technical teams evaluating the feasibility of this route for their specific production needs. Understanding these nuances is vital for making informed decisions about process adoption and for structuring effective technical agreements with manufacturing partners.
Q: What are the environmental benefits of this new Flurbiprofen synthesis route?
A: The process generates only inorganic salts and carbon dioxide as byproducts, eliminating toxic organic waste streams and aligning with strict green chemistry standards for pharmaceutical manufacturing.
Q: How does the palladium catalyst loading impact production costs?
A: The patent specifies an ultra-low catalyst loading of 0.1% to 0.4% mol, which significantly reduces raw material expenses and simplifies downstream metal removal processes compared to traditional stoichiometric methods.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method utilizes widely available raw materials, operates under manageable temperatures (100°C-160°C), and avoids hazardous reagents, making it highly scalable for industrial API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flurbiprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route and are fully equipped to support your Flurbiprofen supply needs with unmatched expertise and capacity. Our state-of-the-art facilities boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercial launch. We maintain stringent purity specifications across all our batches, supported by rigorous QC labs equipped with the latest analytical instrumentation to guarantee that every gram of material meets the highest industry standards for safety and efficacy. Our commitment to quality is matched only by our dedication to customer partnership, as we work collaboratively to optimize processes and resolve any technical challenges that may arise during the development lifecycle.
We invite you to engage with our technical procurement team to discuss how this innovative manufacturing technology can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient synthesis route. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your timeline and budget constraints, ensuring that your supply chain remains robust, compliant, and competitive in the evolving global pharmaceutical landscape.
