Advanced Manufacturing of High-Purity Sodium Picosulfate via Optimized Sulfamic Acid Sulfonation
The pharmaceutical landscape for laxative agents continues to evolve, driven by the demand for higher purity active pharmaceutical ingredients (APIs) and more sustainable manufacturing processes. A significant breakthrough in this domain is detailed in patent CN113387877A, which outlines a refined preparation method for Sodium Picosulfate, a widely used stimulant laxative. This patent addresses critical bottlenecks in traditional synthesis, specifically targeting the reduction of isomeric impurities and inorganic salt residues that have long plagued industrial production. By shifting from hazardous chlorosulfonic acid esterification to a safer sulfamic acid sulfonation protocol, and optimizing the initial condensation step with a hydrochloric-acetic acid system, this technology offers a robust pathway for producing high-purity intermediates. For global procurement and R&D teams, understanding these mechanistic improvements is essential for securing a reliable supply chain of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Sodium Picosulfate has relied on pathways that introduce significant safety hazards and purification challenges. One common conventional route involves the use of 2,6-dichlorophenol as a starting material to avoid ortho-condensation side reactions. While this improves initial regioselectivity, it necessitates a subsequent dechlorination reduction step, typically employing a nickel-aluminum alloy. This introduces severe operational risks due to the pyrophoric nature of the alloy and generates heavy metal waste streams that are costly to treat. Furthermore, alternative routes utilizing concentrated sulfuric acid for the initial condensation of phenol and pyridine-2-carboxaldehyde often suffer from poor selectivity, generating substantial amounts of the 2,4'-isomer impurity which is structurally similar to the desired 4,4'-product and extremely difficult to separate.
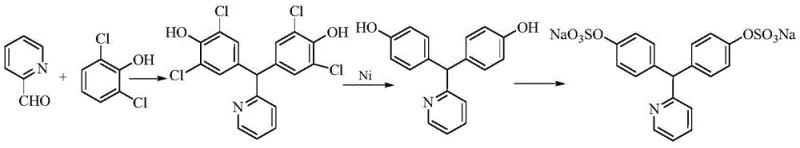
Additionally, the final sulfation step in traditional methods frequently employs chlorosulfonic acid. While effective for esterification, the neutralization of excess chlorosulfonic acid with sodium hydroxide produces massive quantities of inorganic salts, specifically sodium chloride and sodium sulfate. These salts are difficult to completely remove from the highly water-soluble sodium picosulfate product, often requiring repeated desalting procedures that lower overall yield. Moreover, the harsh oxidative conditions associated with these traditional acidic environments can lead to the formation of oxidized impurities, further compromising the quality profile of the final API.
The Novel Approach
The methodology presented in patent CN113387877A represents a paradigm shift towards greener and more efficient chemistry. The core innovation lies in a two-pronged optimization strategy. First, the condensation of phenol and pyridine-2-carboxaldehyde is conducted in a mixed acid system comprising hydrochloric acid and acetic acid. This specific solvent-catalyst combination creates an environment that favors para-substitution while kinetically suppressing the formation of the problematic 2,4'-isomer, achieving impurity levels below 0.5% without the need for complex chromatographic separation. Second, the sulfation step replaces chlorosulfonic acid with sulfamic acid. This reagent choice is transformative; sulfamic acid acts as a mild yet effective sulfonating agent that does not generate corrosive hydrogen chloride gas or excessive inorganic salt byproducts upon neutralization.
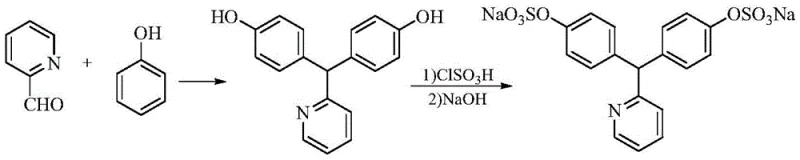
This novel approach streamlines the post-reaction workup significantly. Since unreacted sulfamic acid is insoluble in the organic reaction medium, it can be easily removed by filtration prior to neutralization, drastically reducing the burden on downstream purification. The result is a process that not only enhances safety by eliminating pyrophoric reagents and corrosive gases but also delivers a product with superior purity profiles, characterized by minimal sulfate and chloride residues. This makes the process highly attractive for commercial scale-up where waste disposal costs and operator safety are paramount concerns.
Mechanistic Insights into Acid-Catalyzed Condensation and Sulfamic Acid Sulfonation
To fully appreciate the technical superiority of this method, one must examine the underlying chemical mechanisms governing selectivity and impurity control. In the condensation step, the choice of acid catalyst dictates the electrophilic aromatic substitution pattern on the phenol ring. Traditional sulfuric acid systems can promote scrambling and over-reaction, leading to the thermodynamic formation of the ortho-substituted 2,4'-isomer. The introduction of acetic acid alongside hydrochloric acid likely modulates the proton activity and solvation shell around the reactive iminium intermediate formed from pyridine-2-carboxaldehyde. This modulation sterically and electronically favors attack at the para-position of the phenol, which is less hindered, thereby kinetically trapping the desired 4,4'-bisphenol intermediate before isomerization can occur.
Furthermore, the control of oxidation impurities is a critical quality attribute for Sodium Picosulfate. In traditional strong acid environments, the electron-rich phenolic rings are susceptible to oxidative degradation, leading to quinone-like structures or polymerized tars. The use of sulfamic acid in the second step operates under milder conditions (40-50°C) compared to the exothermic violence of chlorosulfonic acid addition. Sulfamic acid reacts to form the sulfate ester directly without generating strong oxidizing species. This mechanistic gentleness ensures that the delicate bis-phenolic structure remains intact, preventing the formation of the oxidized impurities often seen in older patents. The result is a cleaner reaction profile where the primary impurities are limited to trace mono-sulfated species, which are far easier to remove via crystallization than complex oxidative degradation products.
How to Synthesize Sodium Picosulfate Efficiently
The implementation of this patented technology requires precise control over reaction parameters to maximize yield and purity. The process begins with the careful preparation of the reaction vessel for the condensation step, ensuring that the molar ratio of phenol to aldehyde is maintained between 2.0:1.0 and 2.5:1.0 to drive the reaction to completion while minimizing self-condensation of the aldehyde. Following the isolation of the intermediate, the sulfonation step demands strict temperature control to prevent hydrolysis of the forming sulfate ester. The detailed standardized operating procedures, including specific solvent volumes, addition rates, and crystallization protocols necessary to achieve the reported 99.8%+ purity, are outlined below.
- Condense phenol and pyridine-2-carboxaldehyde in a hydrochloric acid and acetic acid mixture at 40-50°C to form the bisphenol intermediate.
- React the purified intermediate with sulfamic acid using morpholine or N-methylpyrrolidone as a catalyst in pyridine solvent.
- Neutralize with sodium methoxide, followed by crystallization and recrystallization from aqueous ethanol to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates into tangible operational efficiencies and risk mitigation. The shift away from specialized starting materials like 2,6-dichlorophenol to commodity chemicals like phenol significantly stabilizes the raw material supply base. Phenol is a high-volume petrochemical derivative with a robust global supply network, reducing the risk of shortages that can plague niche chlorinated intermediates. Furthermore, the elimination of the nickel-aluminum reduction step removes a major bottleneck in production scheduling, as this step often requires specialized equipment and extended safety protocols that slow down batch turnover times.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the workup and purification stages. By utilizing sulfamic acid, the generation of inorganic salts is minimized, which drastically reduces the volume of wastewater requiring treatment and the energy consumption associated with repeated recrystallizations or desalting steps. Additionally, the higher selectivity of the condensation step means less starting material is wasted on forming inseparable isomers, effectively increasing the mass efficiency of the process. The removal of expensive and hazardous catalysts like nickel alloys further lowers the direct material costs and the associated costs of hazardous waste disposal.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of stable, non-hazardous reagents that are easier to transport and store. Sulfamic acid is a stable solid, unlike chlorosulfonic acid which requires specialized corrosion-resistant tankers and strict handling protocols. This simplifies logistics and reduces the likelihood of supply disruptions due to regulatory transport restrictions. Moreover, the robustness of the HCl-Acetic acid condensation system allows for wider operating windows, making the process more forgiving to minor variations in raw material quality, thus ensuring consistent batch-to-batch availability for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is inherently more scalable. The absence of pyrophoric reagents and corrosive gases aligns with modern green chemistry principles, facilitating easier permitting for plant expansion. The reduction in heavy metal usage (nickel) and inorganic salt waste simplifies compliance with increasingly stringent environmental regulations regarding effluent discharge. This future-proofs the manufacturing site against regulatory tightening, ensuring long-term continuity of supply without the need for costly retrofits to waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and quality implications of this novel synthesis method. These answers are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on how this process outperforms legacy methods in terms of impurity control and operational safety.
Q: How does the new sulfamic acid method improve purity compared to chlorosulfonic acid?
A: The use of sulfamic acid eliminates the formation of excessive inorganic salts like chlorides and sulfates that typically result from chlorosulfonic acid neutralization, thereby simplifying purification and reducing residue levels below 0.04%.
Q: What is the advantage of using an HCl-Acetic acid system for condensation?
A: This specific acid mixture significantly suppresses the formation of the difficult-to-remove 2,4'-isomer impurity, controlling it to below 0.5%, whereas traditional sulfuric acid catalysis often yields over 2.0% of this isomer.
Q: Does this process avoid the use of hazardous nickel-aluminum alloys?
A: Yes, unlike older routes starting with dichlorophenol which require dangerous dechlorination using nickel-aluminum alloys, this method uses readily available phenol, removing the need for heavy metal reduction steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Picosulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN113387877A are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of Sodium Picosulfate meets the highest international pharmacopoeia standards, free from the isomeric and inorganic impurities that compromise drug efficacy.
We invite pharmaceutical companies seeking to optimize their laxative portfolios to engage with our technical procurement team. By leveraging our mastery of this optimized sulfamic acid route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance the quality and reliability of your supply chain.
