Advanced Synthesis and Purification of Tildipirosin for Scalable Veterinary API Manufacturing
Advanced Synthesis and Purification of Tildipirosin for Scalable Veterinary API Manufacturing
The global demand for effective veterinary antibiotics continues to drive innovation in the synthesis of macrolide derivatives, specifically targeting respiratory diseases in livestock. Patent CN112679570A, published in April 2021, introduces a significant technological breakthrough in the manufacturing of Tildipirosin, a semi-synthetic macrolide antibiotic derived from Tylosin. This patent outlines a robust, multi-step synthesis and purification protocol that addresses historical challenges regarding yield stability and impurity control. By utilizing Tylosin phosphate as the starting material and implementing a novel iodination strategy, the disclosed method achieves a final product purity exceeding 99% through simple recrystallization rather than complex chromatography. For R&D directors and procurement specialists, this represents a pivotal shift towards more cost-effective and scalable production methodologies for high-value veterinary intermediates.
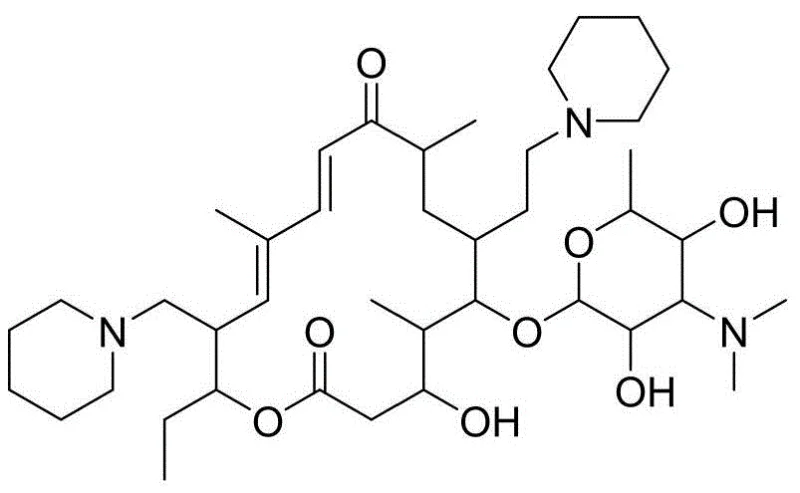
The structural complexity of Tildipirosin, characterized by piperidine substitutions at the C20 and C23 positions alongside a mycaminosaccharide lactone ring, presents unique synthetic hurdles. As illustrated in the molecular structure, the presence of three nitrogen atoms makes the molecule a tribasic entity, requiring precise pH control during isolation. The patented process leverages these chemical properties to facilitate efficient liquid-liquid extraction, avoiding the need for expensive silica gel columns. This approach not only simplifies the operational workflow but also significantly reduces the environmental footprint associated with solvent consumption and waste generation in large-scale manufacturing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tildipirosin has been plagued by inefficient reaction conditions and poor yield profiles, creating substantial bottlenecks for commercial scale-up. Prior art, such as International Patent WO 2008012343, relied on pathways involving amination reduction followed by strong acid hydrolysis and iodide activation. However, these conventional routes often suffered from excessive byproduct formation, resulting in dismal product yields reported as low as 12.2 percent. Furthermore, Chinese patent CN103880903B highlighted the instability of key aldehyde intermediates, which required rigorous temperature control below 0°C to prevent decomposition. Such cryogenic conditions are energy-intensive and difficult to maintain consistently in large industrial reactors, leading to batch-to-batch variability and increased operational costs for manufacturers attempting to secure a reliable veterinary drug intermediate supplier.
The Novel Approach
In stark contrast, the methodology disclosed in CN112679570A introduces a series of mild reaction conditions that enhance system stability and operational simplicity. The process replaces the unstable aldehyde oxidation steps with a controlled hydrolysis and iodination sequence that operates effectively at temperatures ranging from 20°C to 90°C. A critical innovation lies in the preparation of the iodine reagent; instead of adding solid iodine directly, which causes erratic heat absorption and release, the patent mandates dissolving iodine in an organic solvent and adding it dropwise. This modification ensures a stable thermal profile during the activation of the C23 position. Additionally, the purification strategy eliminates column chromatography entirely, relying instead on pH-adjusted extractions and recrystallization, which drastically simplifies the downstream processing and improves the overall economic viability of the synthesis.
Mechanistic Insights into Iodination and pH-Controlled Extraction
The core chemical transformation in this synthesis involves the activation of the hydroxyl group at the C23 position via an Appel-type reaction mechanism, followed by nucleophilic substitution with piperidine. The patent details a precise protocol where triphenylphosphine and pyridine are added to the hydrolyzed intermediate, followed by the dropwise addition of the iodine solution at 20-60°C. This specific order of addition and temperature control is vital for minimizing side reactions such as elimination or over-iodination. By maintaining the water content of the reaction mixture below 0.1% through normal pressure distillation prior to this step, the process prevents the hydrolysis of the activated iodo-intermediate, thereby preserving the integrity of the macrocyclic ring and ensuring high conversion rates in the subsequent amination step.
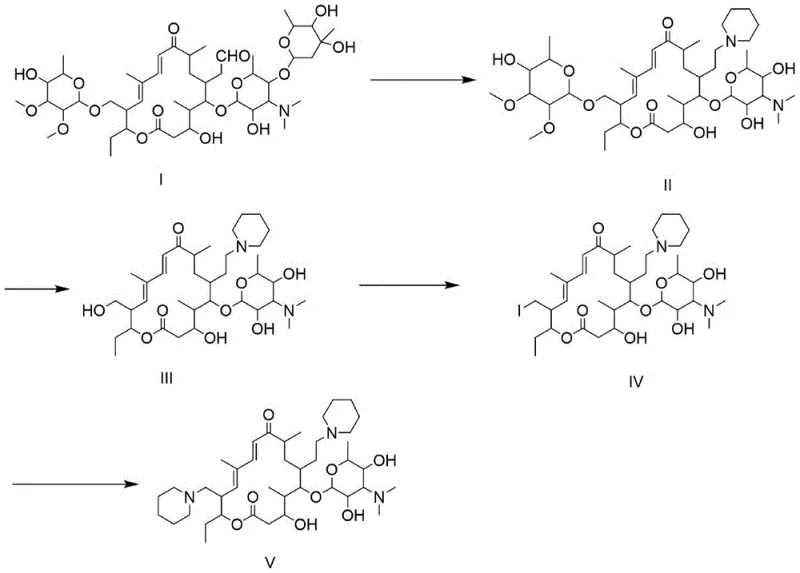
Following the iodination, the second amination reaction introduces the final piperidine moiety at the C23 position. The mechanism relies on the nucleophilic attack of piperidine on the iodo-intermediate, facilitated by an alkali catalyst such as triethylamine or potassium carbonate. The patent emphasizes a sophisticated workup procedure involving multiple pH adjustments to separate the product from unreacted starting materials and phosphine oxide byproducts. By adjusting the pH to neutrality (6.5-7.0) before extracting with organic solvents, and subsequently raising the pH above 13, the process exploits the basicity of the Tildipirosin molecule to partition it selectively into the organic phase. This level of control over the acid-base properties of the intermediates is crucial for achieving the high purity specifications required for veterinary pharmaceutical applications without resorting to costly purification technologies.
How to Synthesize Tildipirosin Efficiently
The synthesis of Tildipirosin described in this patent offers a streamlined pathway that is highly amenable to industrial implementation. The process begins with the reductive amination of Tylosin phosphate, followed by hydrolysis to remove the mycarose sugar, setting the stage for the critical iodination and secondary amination steps. The operational parameters are designed to be forgiving yet precise, allowing for robust manufacturing outcomes. For technical teams looking to implement this route, the key lies in adhering to the specific solvent ratios and temperature gradients outlined in the examples, particularly during the distillation and crystallization phases. The detailed standardized synthesis steps见下方的指南 provide a comprehensive roadmap for replicating these results in a pilot or production environment.
- Dissolve tylosin phosphate in organic solvent, add formic acid and piperidine, heat to 60-90°C for reductive amination, then hydrolyze the aqueous layer at 55-75°C.
- Perform iodination by dropwise adding iodine solution to a mixture of triphenylphosphine and pyridine at 20-60°C, ensuring water content is below 0.1%.
- Conduct secondary amination with piperidine and base catalyst at 40-90°C, followed by pH-controlled extraction and double recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for procurement managers and supply chain heads focused on cost reduction in pharmaceutical manufacturing. The elimination of column chromatography is perhaps the most significant economic driver, as it removes the need for large volumes of silica gel and the associated solvent recovery infrastructure. This simplification translates directly into lower variable costs per kilogram of produced API intermediate. Furthermore, the use of common organic solvents such as ethyl acetate, dichloromethane, and acetonitrile ensures that raw material sourcing remains straightforward and resilient against market fluctuations, enhancing the overall reliability of the supply chain for high-purity veterinary drug intermediates.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing energy-intensive cryogenic reactions with mild heating protocols and eliminating expensive chromatographic purification. The ability to control water content via normal pressure distillation rather than high-vacuum systems further reduces capital expenditure on specialized equipment. These efficiencies collectively lower the barrier to entry for commercial scale-up of complex veterinary antibiotics, making the final product more price-competitive in the global animal health market.
- Enhanced Supply Chain Reliability: By utilizing Tylosin phosphate, a widely available fermentation product, as the starting material, the synthesis avoids reliance on scarce or geopolitically sensitive reagents. The robust nature of the reaction conditions, which tolerate a broader range of temperatures compared to prior art, minimizes the risk of batch failures due to minor process deviations. This stability ensures consistent delivery schedules and reduces the lead time for high-purity veterinary drug intermediates, allowing manufacturers to respond more agilely to market demand spikes.
- Scalability and Environmental Compliance: The reliance on liquid-liquid extraction and recrystallization aligns well with green chemistry principles by reducing solid waste generation associated with silica disposal. The process is inherently scalable, as the unit operations involved—mixing, heating, phase separation, and filtration—are standard in modern chemical plants. This facilitates a smoother transition from laboratory benchtop to multi-ton production scales, ensuring that environmental compliance is maintained through efficient solvent recovery and minimized hazardous waste streams.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of the Tildipirosin synthesis route described in patent CN112679570A. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on critical process parameters. Understanding these nuances is essential for process engineers aiming to maximize yield and purity while maintaining operational safety and efficiency in a GMP-compliant environment.
Q: How does this patent improve the stability of the iodination step compared to prior art?
A: Unlike prior methods that added solid iodine directly causing temperature fluctuations, this patent dissolves iodine in solvent first and adds it dropwise. This maintains a stable reaction system between 20-60°C and prevents byproduct formation.
Q: What is the achieved purity of Tildipirosin using this purification method?
A: The method utilizes a two-stage recrystallization process without column chromatography, consistently achieving a final product purity and content of greater than 99%, which is critical for veterinary API standards.
Q: Why is the water content control critical in the intermediate stage?
A: The process controls water content to less than 0.1% via normal pressure distillation before the iodination step. This eliminates the need for complex high-vacuum drying while ensuring the iodination reaction proceeds smoothly without hydrolysis side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tildipirosin Supplier
As the veterinary pharmaceutical sector evolves, the need for partners who can translate complex patent methodologies into commercial reality has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of Tildipirosin meets the highest international standards. We understand the critical nature of supply continuity in the animal health market and have optimized our logistics and production planning to support long-term partnerships with global innovators.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can benefit your bottom line. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of technical excellence and supply chain security.
