Revolutionizing Tildipirosin Production: A High-Yield Hydroiodic Acid Reflux Strategy for Veterinary API Intermediates
The global demand for effective veterinary antibiotics continues to surge, driven by the critical need to manage respiratory diseases in livestock such as pigs and cattle. At the forefront of this therapeutic class is Tildipirosin, a semi-synthetic macrolide derivative known commercially under the trade name Zuprevo. The efficient manufacturing of this potent drug relies heavily on the availability of high-quality intermediates, specifically 20-piperidyl-23-iodo-5-O-mycaminosyl-tylonolide. Recent advancements disclosed in patent CN111393493A present a transformative synthetic methodology that addresses long-standing bottlenecks in the production of this key precursor. By shifting away from complex multi-step activation protocols towards a streamlined hydroiodic acid reflux strategy, this innovation promises to redefine the economic and operational landscape for reliable veterinary drugs supplier networks globally. The structural complexity of Tildipirosin, characterized by its macrocyclic lactone ring and specific piperidine substitutions, demands precise chemical control to ensure bioavailability and safety.  Understanding the molecular architecture is essential for R&D teams aiming to optimize downstream processing and formulation stability.
Understanding the molecular architecture is essential for R&D teams aiming to optimize downstream processing and formulation stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tildipirosin intermediates has been plagued by inefficient routes that impose significant burdens on both cost and environmental compliance. Prior art, such as the method described in WO2008012343, relies on a cumbersome five-step sequence involving piperidine amination, dual hydrolysis, and activation using iodine and pyridine. 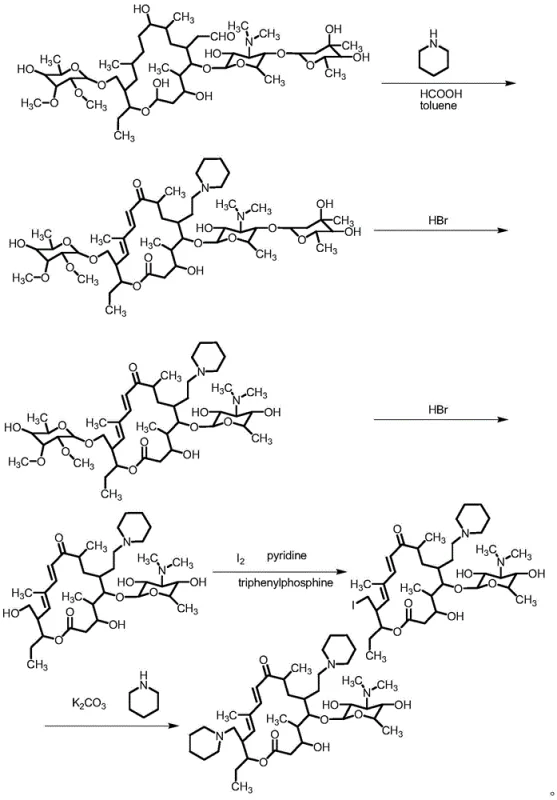 This traditional approach generates substantial volumes of waste liquid containing high concentrations of organic solvents and hazardous byproducts, drastically inflating the cost reduction in veterinary drugs manufacturing. Furthermore, alternative routes like those found in CN102863487 utilize triphenylphosphine and imidazole, reagents known for their unpleasant odors and difficult removal profiles.
This traditional approach generates substantial volumes of waste liquid containing high concentrations of organic solvents and hazardous byproducts, drastically inflating the cost reduction in veterinary drugs manufacturing. Furthermore, alternative routes like those found in CN102863487 utilize triphenylphosphine and imidazole, reagents known for their unpleasant odors and difficult removal profiles.  These methods often require anhydrous conditions and specialized equipment, creating barriers to entry for standard manufacturing facilities. The reliance on column chromatography for purification, as seen in some Japanese patents, further exacerbates the issue by consuming excessive solvents and time, rendering the process unsuitable for the commercial scale-up of complex veterinary antibiotics.
These methods often require anhydrous conditions and specialized equipment, creating barriers to entry for standard manufacturing facilities. The reliance on column chromatography for purification, as seen in some Japanese patents, further exacerbates the issue by consuming excessive solvents and time, rendering the process unsuitable for the commercial scale-up of complex veterinary antibiotics.
The Novel Approach
In stark contrast, the novel methodology introduced in CN111393493A offers a paradigm shift by employing a direct reflux reaction with hydroiodic acid. This approach bypasses the need for separate activation steps using phosphine derivatives, thereby simplifying the entire workflow. The core innovation lies in the direct conversion of 20-piperidyl-5-O-mycaminosyl-tylonolide to the iodo-intermediate using 57% hydroiodic acid in acetonitrile.  This single-step transformation not only reduces the number of unit operations but also significantly lowers the generation of hazardous waste. By eliminating the use of malodorous reagents like triphenylphosphine and pyridine, the new process creates a safer working environment and reduces the load on wastewater treatment facilities. The simplicity of the workup, which involves concentration, pH adjustment, and filtration, ensures that the high-purity veterinary drugs can be produced with minimal downtime between batches, enhancing overall facility throughput.
This single-step transformation not only reduces the number of unit operations but also significantly lowers the generation of hazardous waste. By eliminating the use of malodorous reagents like triphenylphosphine and pyridine, the new process creates a safer working environment and reduces the load on wastewater treatment facilities. The simplicity of the workup, which involves concentration, pH adjustment, and filtration, ensures that the high-purity veterinary drugs can be produced with minimal downtime between batches, enhancing overall facility throughput.
Mechanistic Insights into Hydroiodic Acid Reflux Iodination
The chemical elegance of this synthesis lies in the dual functionality of hydroiodic acid, which acts as both a hydrolyzing agent and an iodinating source under reflux conditions. When 20-piperidyl-5-O-mycaminosyl-tylonolide is subjected to reflux in acetonitrile with 57% HI, the acidic environment facilitates the cleavage of specific protecting groups or functional modifications required for the subsequent substitution. The reaction mechanism likely proceeds through a protonation of the hydroxyl group at the 23-position, followed by a nucleophilic substitution where the iodide ion displaces the leaving group. This direct substitution is highly favorable thermodynamically under reflux temperatures, driving the equilibrium towards the desired iodo-product. The choice of acetonitrile as a solvent is critical, as it provides excellent solubility for the macrolide substrate while maintaining stability under the acidic reflux conditions. This mechanistic pathway avoids the formation of phosphine oxide byproducts, which are notoriously difficult to separate from the final API intermediate, thus ensuring a cleaner reaction profile.
Impurity control is a paramount concern for R&D Directors overseeing the production of veterinary actives. The novel HI reflux method demonstrates superior selectivity, minimizing the formation of di-iodinated byproducts or degradation products that often plague harsher iodination protocols. The patent data indicates that by carefully controlling the molar ratio of hydroiodic acid (between 1:0.55 to 1:0.62 relative to the substrate), the reaction can be tuned to maximize yield while suppressing side reactions. Post-reaction treatment involves adjusting the pH to 11-12 using sodium hydroxide, which neutralizes excess acid and precipitates the product or facilitates its extraction. This pH swing is a robust method for removing acidic impurities and residual starting materials. The resulting crude product exhibits high purity, often exceeding 98% without the need for chromatographic purification, validating the efficacy of this mechanism in producing commercial scale-up of complex veterinary drugs with consistent quality attributes.
How to Synthesize 20-piperidyl-23-iodo-5-O-mycaminosyl-tylonolide Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the dissolution of the tylosin derivative in acetonitrile, followed by the controlled addition of hydroiodic acid. Maintaining the reflux temperature for the specified duration of 4 to 5 hours is crucial to achieve complete conversion, as monitored by HPLC analysis.
- Dissolve 20-piperidyl-5-O-mycaminosyl-tylonolide in acetonitrile within a reaction vessel equipped with stirring capabilities.
- Add 57% hydroiodic acid to the mixture and maintain a reflux condition for approximately 4.5 hours to ensure complete conversion.
- Concentrate the mixture under reduced pressure, adjust pH to 11-12 with sodium hydroxide, and filter to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical feasibility. The elimination of expensive and volatile reagents like triphenylphosphine and iodine directly translates to a more stable and predictable raw material cost structure. By simplifying the process flow and removing the need for column chromatography, manufacturers can significantly reduce solvent consumption and waste disposal fees. This aligns perfectly with global trends towards greener chemistry and sustainable manufacturing practices. The robustness of the HI reflux method also implies a reduction in batch failure rates, ensuring a more consistent supply of critical intermediates for downstream API production. Consequently, this leads to cost reduction in veterinary drugs manufacturing by optimizing the utilization of reactor time and minimizing the inventory holding costs associated with complex multi-step syntheses.
- Cost Reduction in Manufacturing: The replacement of costly activation reagents with commodity-grade hydroiodic acid results in substantial savings on raw material expenditures. Furthermore, the simplified workup procedure reduces the labor and utility costs associated with extended purification steps. The ability to recycle solvents like acetonitrile more efficiently due to the absence of phosphine contaminants further enhances the economic viability of the process. These factors combine to create a leaner manufacturing model that is resilient against market fluctuations in specialty chemical pricing.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often a bottleneck in the pharmaceutical supply chain. This method utilizes readily available starting materials and reagents, reducing the risk of supply disruptions caused by the scarcity of specialized catalysts. The shorter reaction time and simplified isolation steps allow for faster turnaround times, enabling suppliers to respond more agilely to market demand spikes. This reliability is crucial for maintaining the continuity of supply for essential veterinary medicines used in food-producing animals.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in 500L reactors without loss of efficiency. The reduction in hazardous waste generation simplifies regulatory compliance and lowers the burden on environmental treatment infrastructure. By avoiding the use of heavy metals or persistent organic pollutants, the facility can maintain a cleaner environmental footprint, which is increasingly important for maintaining operating licenses and corporate social responsibility goals in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific beneficial effects and experimental data provided in the patent documentation.
Q: What are the primary advantages of the hydroiodic acid reflux method over traditional phosphine-based iodination?
A: The hydroiodic acid reflux method eliminates the need for expensive and odor-intensive reagents like triphenylphosphine and iodine. This significantly reduces waste liquid volume and simplifies the post-treatment process, leading to lower environmental compliance costs and higher overall production efficiency compared to prior art methods.
Q: How does this synthesis route impact the purity profile of the final tildipirosin intermediate?
A: By utilizing a direct one-step hydrolysis and iodination approach, the process minimizes side reactions associated with multi-step activation. Experimental data from the patent indicates that this method consistently achieves product purity levels exceeding 98%, which is critical for meeting stringent veterinary pharmaceutical standards.
Q: Is this process scalable for commercial manufacturing of veterinary antibiotics?
A: Yes, the process is specifically designed for industrial scalability. It avoids complex column chromatography purification steps and uses common solvents like acetonitrile. The simplified workup procedure involving pH adjustment and filtration allows for easy adaptation to large-scale reactor systems ranging from hundreds of kilograms to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tildipirosin Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent to production requires a partner with deep technical expertise and robust manufacturing capabilities. Our team specializes in the commercial scale-up of complex veterinary drugs, leveraging advanced process chemistry to bring innovative synthesis routes like the HI-reflux method to life. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Tildipirosin intermediate meets the highest international standards for veterinary applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that can accelerate your project timelines. Contact us today to discuss how our advanced manufacturing solutions can support your growth in the global veterinary pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
