Advanced Radical C-H Activation for Commercial Scale Production of 2-Substituted Benzothiazoles
The pharmaceutical and agrochemical industries continuously demand more efficient pathways to access nitrogen-containing heterocycles, particularly benzothiazole scaffolds which are ubiquitous in bioactive molecules. Patent CN108358868B introduces a transformative approach to synthesizing 2-substituted benzothiazole compounds, shifting away from classical condensation chemistry toward a direct, copper-catalyzed radical C-H activation strategy. This innovation allows for the one-step coupling of readily available toluene derivatives with o-aminothiophenol, effectively constructing both C-S and C-N bonds simultaneously under oxidative conditions. By utilizing di-tert-butyl peroxide (DTBP) as a radical initiator and methanesulfonic acid as an additive, this method achieves high atom economy and operational simplicity. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the manufacturing of high-purity pharmaceutical intermediates, reducing reliance on unstable aldehyde precursors while maintaining rigorous quality standards essential for regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted benzothiazoles has relied heavily on the condensation of aromatic aldehydes with o-aminothiophenols or the cyclization of o-halothioamides. These traditional routes suffer from inherent thermodynamic and logistical drawbacks that complicate large-scale production. Aromatic aldehydes, often required as key starting materials, are prone to oxidation and polymerization, necessitating strict storage conditions and frequent quality control checks that inflate inventory costs. Furthermore, reactions involving acid anhydrides typically generate stoichiometric amounts of acidic waste, creating substantial environmental burdens and requiring complex neutralization and disposal protocols. The multi-step nature of preparing these specialized precursors also elongates the overall lead time, making the supply chain vulnerable to disruptions in the availability of fine chemical building blocks. Consequently, manufacturers face challenges in achieving consistent batch-to-batch purity, as side reactions associated with aldehyde instability can introduce difficult-to-remove impurities into the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN108358868B leverages the direct functionalization of sp3 C-H bonds in toluene derivatives, bypassing the need for pre-oxidized aldehyde intermediates entirely. This radical-mediated pathway operates under relatively mild thermal conditions (120°C) using commodity chemicals like toluene and its halogenated derivatives as both solvent and reactant. The process is catalyzed by inexpensive copper salts, specifically cuprous oxide, which facilitates the generation of benzyl radicals that subsequently couple with the sulfur and nitrogen centers of o-aminothiophenol. 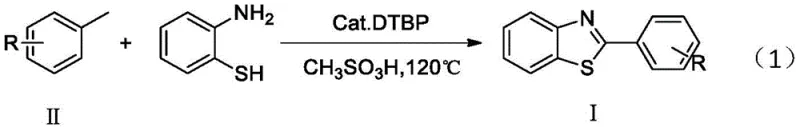 This one-pot transformation not only simplifies the reactor setup by eliminating intermediate isolation steps but also drastically improves the overall mass balance of the process. For a reliable pharma intermediates supplier, adopting this route means accessing a broader range of substituted benzothiazoles from a single, robust platform technology, thereby enhancing flexibility in responding to custom synthesis requests without retooling entire production lines.
This one-pot transformation not only simplifies the reactor setup by eliminating intermediate isolation steps but also drastically improves the overall mass balance of the process. For a reliable pharma intermediates supplier, adopting this route means accessing a broader range of substituted benzothiazoles from a single, robust platform technology, thereby enhancing flexibility in responding to custom synthesis requests without retooling entire production lines.
Mechanistic Insights into Copper-Catalyzed Radical C-H Activation
Understanding the mechanistic underpinnings of this transformation is crucial for process chemists aiming to optimize reaction parameters for commercial scale-up. The reaction proceeds through a distinct radical manifold initiated by the thermal decomposition of DTBP in the presence of the copper catalyst. Initially, DTBP undergoes homolytic cleavage to generate tert-butoxy radicals, which abstract a hydrogen atom from the methyl group of the toluene substrate. This step generates a resonance-stabilized benzyl radical, a key reactive intermediate that drives the subsequent bond-forming events. Simultaneously, the copper catalyst cycles between oxidation states, coordinating with the o-aminothiophenol to form a metal-ligand complex that primes the nucleophilic centers for attack. The convergence of the benzyl radical with this copper-aminothiophenol complex leads to the formation of a C-S bond, yielding a 2-(benzylthio)aniline intermediate. Subsequent intramolecular cyclization and oxidative dehydrogenation, mediated by the excess oxidant, close the thiazole ring to afford the final 2-substituted benzothiazole product. This detailed mechanistic picture confirms that the reaction does not proceed via a benzaldehyde intermediate, as control experiments showed zero yield when benzaldehyde was used as the starting material under identical conditions.
To rigorously validate the radical nature of this mechanism, the patent data presents compelling evidence through radical trapping and kinetic isotope effect experiments. When the radical scavenger TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) was introduced to the reaction mixture, the yield of the desired product decreased progressively with increasing amounts of TEMPO, eventually dropping to zero at 0.9mmol loading. 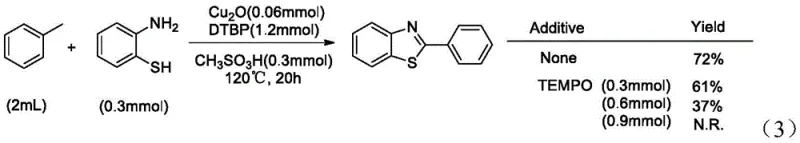 This complete inhibition confirms that free radical species are indispensable intermediates in the catalytic cycle. Furthermore, to identify the rate-determining step, a competition experiment was conducted using a mixture of toluene and deuterated toluene. The resulting kinetic isotope effect (KIE) value of KH/KD = 6.8 indicates a primary isotope effect, unequivocally demonstrating that the cleavage of the benzylic C-H bond is the slowest, rate-limiting step in the entire sequence.
This complete inhibition confirms that free radical species are indispensable intermediates in the catalytic cycle. Furthermore, to identify the rate-determining step, a competition experiment was conducted using a mixture of toluene and deuterated toluene. The resulting kinetic isotope effect (KIE) value of KH/KD = 6.8 indicates a primary isotope effect, unequivocally demonstrating that the cleavage of the benzylic C-H bond is the slowest, rate-limiting step in the entire sequence. 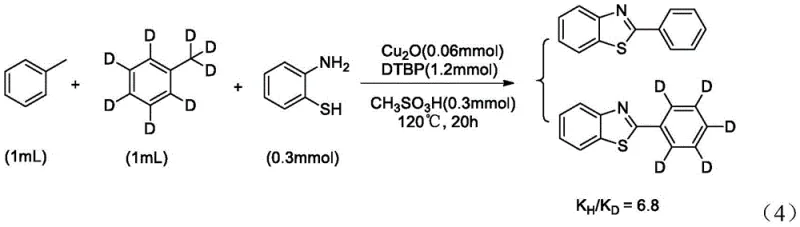 This insight is invaluable for engineering the reactor; it suggests that factors influencing C-H abstraction efficiency, such as temperature precision and radical initiator concentration, are the critical levers for maximizing throughput and minimizing reaction time in a manufacturing setting.
This insight is invaluable for engineering the reactor; it suggests that factors influencing C-H abstraction efficiency, such as temperature precision and radical initiator concentration, are the critical levers for maximizing throughput and minimizing reaction time in a manufacturing setting.
How to Synthesize 2-Substituted Benzothiazole Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling, making it highly suitable for translation from laboratory bench to pilot plant. The standard protocol involves charging a reaction vessel with cuprous oxide (0.06mmol), o-aminothiophenol (0.3mmol), di-tert-butyl peroxide (1.2mmol), and methanesulfonic acid (0.3mmol) directly into the toluene substrate (2mL), which serves as both reactant and solvent. The mixture is then heated to reflux at 120°C for approximately 20 hours to ensure full conversion. Following the reaction, the workup procedure is straightforward: the mixture is cooled, diluted with ethyl acetate, and washed with aqueous solutions to remove inorganic salts and acidic byproducts. The organic layer is dried and concentrated, with final purification achieved via standard chromatographic techniques.
- Combine 0.06mmol cuprous oxide catalyst, 0.3mmol o-aminothiophenol, 1.2mmol di-tert-butyl peroxide (DTBP), and 0.3mmol methanesulfonic acid into 2mL of toluene or its derivative solvent.
- Heat the reaction mixture to reflux at 120°C and maintain stirring for 20 hours to ensure complete radical conversion and cyclization.
- Cool to room temperature, dilute with ethyl acetate, wash sequentially with saturated saline and NaHSO3 solution, dry over anhydrous Na2SO4, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this novel synthetic route offers profound advantages that directly address the pain points of cost volatility and supply chain fragility often encountered in fine chemical manufacturing. By replacing specialized, moisture-sensitive aldehyde reagents with bulk commodity toluenes, the raw material cost base is significantly lowered, and the risk of supply disruption due to niche precursor shortages is virtually eliminated. The elimination of pre-functionalization steps reduces the total number of unit operations required, which translates to lower energy consumption and reduced labor overhead per kilogram of product. Moreover, the use of a heterogeneous or easily removable copper catalyst system simplifies the purification workflow, potentially reducing the need for expensive scavenging resins or complex recrystallization sequences that often bottleneck production schedules. These efficiencies collectively contribute to a more resilient supply chain capable of meeting just-in-time delivery commitments for downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost, low-stability aldehyde intermediates with inexpensive toluene derivatives that are available in massive global volumes. Since the reaction utilizes the solvent as a reactant, there is no need for additional diluents, which maximizes reactor volumetric efficiency and reduces solvent recovery costs. Additionally, the high atom economy of the radical coupling means less waste is generated per unit of product, lowering the expenses associated with waste treatment and environmental compliance. The avoidance of protecting group chemistry further streamlines the synthesis, removing the cost of extra reagents and the time required for protection-deprotection cycles.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like toluene, chlorotoluene, and fluorotoluene ensures that the supply chain is anchored in petrochemical feedstocks with established, multi-vendor availability, rather than relying on single-source fine chemical suppliers. This diversification of raw material sources mitigates the risk of production stoppages caused by upstream shortages. The robustness of the reaction conditions (120°C, ambient pressure) also means that the process can be run in standard glass-lined or stainless steel reactors without requiring specialized high-pressure equipment, allowing for greater flexibility in choosing contract manufacturing organizations (CMOs) or internal production facilities.
- Scalability and Environmental Compliance: The simplicity of the one-pot procedure facilitates seamless scale-up from gram to ton quantities without the need for extensive process re-engineering. The reaction generates minimal hazardous byproducts compared to traditional methods involving acid anhydrides or halogenated cyclization precursors, aligning well with green chemistry principles and increasingly stringent environmental regulations. The ability to tolerate various functional groups, including halogens, without additional protection steps reduces the overall chemical footprint of the synthesis. This environmental compatibility not only reduces regulatory hurdles but also enhances the corporate sustainability profile of the manufacturing operation, a key metric for modern pharmaceutical procurement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this radical coupling technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent literature to provide clarity for potential partners.
Q: Why is the radical mechanism preferred over traditional aldehyde condensation for benzothiazole synthesis?
A: Traditional methods often rely on unstable aromatic aldehydes or harsh acid anhydrides, leading to difficult post-treatment and lower atom economy. The radical pathway described in CN108358868B utilizes stable, commodity toluene derivatives, bypassing the need for pre-functionalized aldehyde intermediates and significantly simplifying the supply chain.
Q: What is the rate-determining step in this copper-catalyzed process?
A: Kinetic Isotope Effect (KIE) studies indicate a KH/KD ratio of 6.8, confirming that the cleavage of the benzylic C-H bond on the toluene substrate is the rate-determining step. This insight is critical for optimizing reaction temperature and catalyst loading during commercial scale-up.
Q: Can this method tolerate halogenated substrates for further functionalization?
A: Yes, the protocol demonstrates excellent compatibility with halogenated toluenes such as 4-fluorotoluene (80% yield), 4-chlorotoluene (77% yield), and 4-bromotoluene (63% yield). This retention of halogen handles is vital for downstream cross-coupling reactions in complex API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Benzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from academic innovation to industrial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN108358868B can be reliably translated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-substituted benzothiazole meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance extends beyond simple compliance; we proactively monitor impurity profiles to ensure consistency that supports your regulatory filings.
We invite you to engage with our technical procurement team to discuss how this advanced radical synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient manufacturing route for your specific target molecules. We encourage you to contact us today to obtain specific COA data for our benchmark batches and to receive comprehensive route feasibility assessments that will help you make informed decisions about your future sourcing strategies.
