Scalable Synthesis of Non-Thiazolidinedione Insulin Sensitizers for Global Pharma Supply
The pharmaceutical landscape for Type II diabetes treatment has long been dominated by thiazolidinediones, yet concerns regarding hepatotoxicity have driven an urgent search for safer alternatives. Patent CN1478770A introduces a groundbreaking class of 1,3-dicarbonyl compounds that exhibit potent insulin-sensitizing activity without the structural liabilities of traditional glitazones. These molecules, characterized by a central 1,3-dicarbonyl motif linked to various heterocyclic or aromatic systems, represent a significant evolution in metabolic disease therapeutics. The structural diversity allowed by the general formula enables fine-tuning of pharmacokinetic properties while maintaining efficacy.
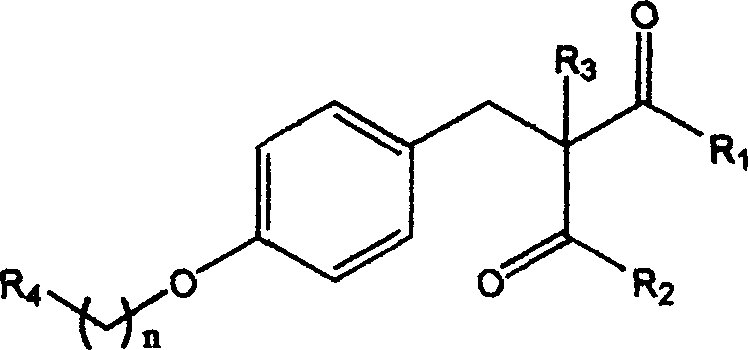
For R&D directors and procurement specialists, this patent offers a robust platform for developing next-generation anti-diabetic agents. The synthesis pathways described rely on readily available starting materials such as substituted benzaldehydes and malonic acid derivatives, ensuring a stable supply chain foundation. By shifting away from the toxicophore associated with liver damage, these compounds address a critical unmet medical need, positioning them as high-value targets for pharmaceutical development pipelines focused on safety and efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional insulin sensitizers, particularly the thiazolidinedione class including troglitazone and rosiglitazone, have faced significant clinical setbacks due to idiosyncratic liver toxicity. The presence of the thiazolidinedione ring is often implicated in these adverse effects, leading to market withdrawals and stringent regulatory scrutiny. From a manufacturing perspective, synthesizing complex heterocyclic rings with high purity can be challenging, often requiring harsh conditions or expensive catalysts that complicate scale-up. Furthermore, the metabolic instability of some conventional scaffolds can lead to unpredictable pharmacokinetic profiles, necessitating extensive and costly reformulation efforts during drug development.
The Novel Approach
The approach detailed in CN1478770A circumvents these issues by utilizing a 1,3-dicarbonyl backbone, which is chemically distinct from the problematic thiazolidinedione ring. This structural modification retains the ability to promote adipocyte differentiation, a key marker for insulin sensitization, as demonstrated in 3T3-L1 cell assays. The synthetic strategy employs straightforward condensation reactions followed by catalytic hydrogenation, avoiding the need for complex ring-closing steps that often limit yield. This simplicity not only enhances the safety profile of the final drug candidate but also streamlines the manufacturing process, making it highly attractive for cost-effective commercial production.
Mechanistic Insights into Condensation and Hydrogenation Strategy
The core synthetic methodology relies on a Knoevenagel-type condensation between a functionalized benzaldehyde, specifically 4-[2-(N-methyl-2-pyridylamino)ethoxy]benzaldehyde, and active methylene compounds like dimethyl malonate. This reaction is typically catalyzed by weak bases such as piperidine acetate in refluxing toluene, facilitating the removal of water to drive the equilibrium towards the benzylidene intermediate. The mechanism involves the formation of an enolate from the dicarbonyl compound, which attacks the carbonyl carbon of the aldehyde, followed by dehydration to form the alpha,beta-unsaturated system. This step is crucial for establishing the carbon skeleton of the target molecule with high regioselectivity.
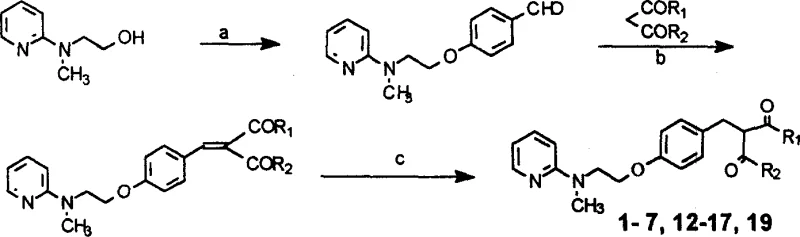
Following the condensation, the unsaturated intermediate undergoes catalytic hydrogenation using 10% palladium on carbon (Pd-C) in a mixed solvent system of methanol and dioxane. This reduction step saturates the double bond, yielding the final 1,3-dicarbonyl derivative with the desired stereochemistry and stability. The use of heterogeneous catalysis allows for easy removal of the catalyst via filtration, minimizing metal contamination in the final product—a critical quality attribute for pharmaceutical intermediates. The patent details various substituents (R1, R2, R3) that can be introduced, allowing for the modulation of lipophilicity and metabolic stability without altering the core synthetic logic.
How to Synthesize 1,3-Dicarbonyl Insulin Sensitizer Intermediates Efficiently
The synthesis of these high-value intermediates follows a logical two-step sequence that balances yield and purity. Initially, the condensation reaction must be carefully monitored to ensure complete conversion of the aldehyde, as residual starting material can complicate downstream purification. The subsequent hydrogenation step requires precise control of hydrogen pressure and catalyst loading to prevent over-reduction or side reactions. Detailed operational parameters, including solvent ratios and temperature profiles, are essential for reproducibility. For a comprehensive guide on executing these steps with optimal efficiency, please refer to the standardized protocol below.
- Condense 4-[2-(N-methyl-2-pyridylamino)ethoxy]benzaldehyde with dimethyl malonate in toluene using piperidine acetate catalyst under reflux.
- Purify the intermediate benzylidene malonate via silica gel column chromatography using petroleum ether and ethyl acetate gradients.
- Perform catalytic hydrogenation using 10% Pd-C in methanol/dioxane mixture to reduce the double bond and obtain the final saturated product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits. The reliance on commodity chemicals such as malonic esters and substituted phenols ensures that raw material sourcing is not bottlenecked by exotic or scarce reagents. This accessibility translates directly into reduced procurement risks and more stable pricing models over the long term. Furthermore, the avoidance of heavy metal catalysts in the final steps, or the use of easily removable heterogeneous catalysts like Pd-C, simplifies the purification workflow, thereby reducing the overall cost of goods sold (COGS) and accelerating time-to-market for new drug applications.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and toxic organometallic reagents often required for constructing complex heterocycles. By utilizing simple condensation and hydrogenation steps, the process reduces energy consumption and waste generation. The high atom economy of the condensation reaction means less raw material is wasted, leading to significant cost savings in large-scale production runs without compromising the quality of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The starting materials, including various alkyl malonates and pyridine derivatives, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification ensures continuity of supply even during market fluctuations. Additionally, the robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling companies to establish regional production hubs to serve local markets more efficiently and reduce logistics lead times.
- Scalability and Environmental Compliance: The processes described operate under relatively mild conditions, avoiding extreme pressures or temperatures that require specialized equipment. This ease of scale-up facilitates the transition from pilot plant to commercial manufacturing with minimal engineering changes. Moreover, the use of standard solvents like toluene and ethanol, which can be recovered and recycled, aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this technology is vital for stakeholders evaluating its potential. The following questions address common inquiries regarding the safety profile, synthetic feasibility, and regulatory implications of these novel compounds. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of these 1,3-dicarbonyl compounds over traditional thiazolidinediones?
A: The primary advantage is the absence of the thiazolidinedione ring structure, which is associated with hepatotoxicity in drugs like troglitazone. These novel compounds maintain insulin-sensitizing activity while potentially offering a safer liver toxicity profile.
Q: What are the key reaction conditions for the synthesis described in the patent?
A: The synthesis typically involves condensation reactions in solvents like toluene or DMF at temperatures ranging from room temperature to reflux (approx. 110°C), followed by catalytic hydrogenation using palladium on carbon (Pd-C) at ambient pressure.
Q: Can these intermediates be scaled for commercial API production?
A: Yes, the synthetic routes utilize standard organic transformations such as Knoevenagel condensation and catalytic hydrogenation, which are well-established and easily scalable from laboratory to industrial manufacturing settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Dicarbonyl Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent chemistry into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. We understand the critical nature of stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,3-dicarbonyl intermediates meets the highest international standards. Our commitment to quality assurance ensures that your downstream synthesis remains uninterrupted and compliant.
We invite you to collaborate with us to leverage these innovative synthetic routes for your next-generation diabetes therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your drug development timeline.
