Scalable Synthesis of Vadadustat Intermediates: A Technical Breakthrough in HIF-PH Inhibitor Manufacturing
Scalable Synthesis of Vadadustat Intermediates: A Technical Breakthrough in HIF-PH Inhibitor Manufacturing
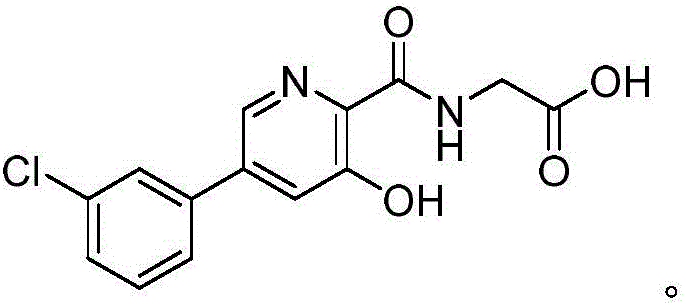
The pharmaceutical landscape for treating anemia associated with chronic kidney disease has been significantly reshaped by the development of Hypoxia-Inducible Factor Prolyl Hydroxylase (HIF-PH) inhibitors. Among these, Vadadustat stands out as a potent oral agent currently advancing through late-stage clinical trials globally. The efficient manufacturing of its core intermediates is critical for ensuring a stable supply chain for this vital medication. Patent CN110903238A introduces a revolutionary preparation method that addresses the longstanding bottlenecks in synthesizing the key pyridone scaffold. This technical insight report analyzes the novel three-step pathway, contrasting it with legacy methods to highlight its superiority in purity, yield, and operational simplicity for industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
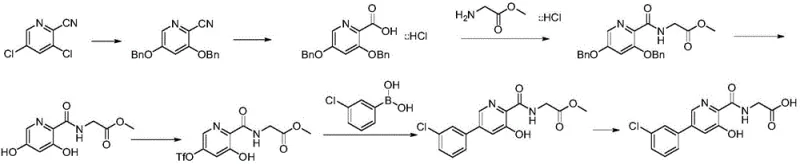
Prior art synthesis routes, such as those disclosed in US20070299086, present substantial challenges for commercial scale-up and cost-effective manufacturing. The traditional approach typically involves a convoluted seven-step synthetic sequence that suffers from an abysmal overall yield of approximately 9 percent. A critical bottleneck in these legacy processes is the reliance on harsh reaction conditions, including microwave irradiation at extreme temperatures of 190°C for benzyl alcohol substitution, which poses significant safety risks and equipment limitations for large reactors. Furthermore, the purification strategy is excessively burdensome, requiring up to five separate column chromatography operations to isolate intermediates of acceptable purity. This dependence on chromatography is economically unsustainable for multi-kilogram or metric-ton production, leading to excessive solvent consumption, prolonged cycle times, and difficult waste management.
The Novel Approach
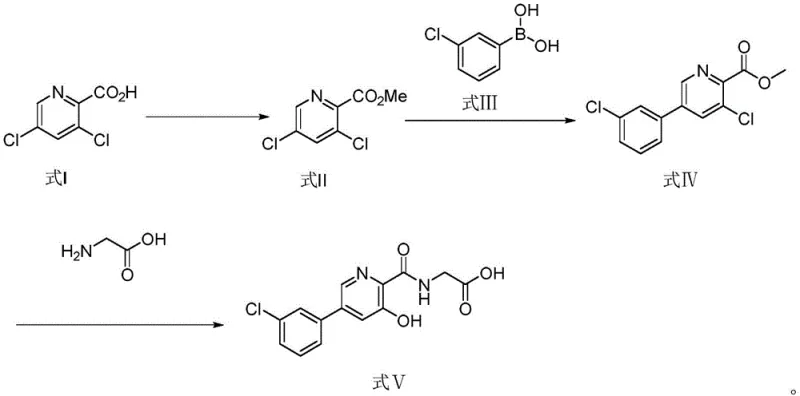
In stark contrast, the methodology outlined in CN110903238A streamlines the synthesis into a robust three-step process that eliminates the need for chromatographic purification entirely. The new route initiates with a straightforward esterification, followed by a highly selective palladium-catalyzed cross-coupling, and concludes with a direct amidation-hydrolysis sequence. By leveraging readily available starting materials like 3,5-dichloro-2-picolinic acid and 3-chlorobenzeneboronic acid, the process achieves high conversion rates under mild thermal conditions (30-100°C for coupling, 80-130°C for amidation). The ability to purify intermediates via simple recrystallization or filtration not only drastically reduces operational complexity but also ensures a consistent impurity profile suitable for pharmaceutical grade standards. This strategic simplification represents a paradigm shift from laboratory-scale curiosity to industrial viability.
Mechanistic Insights into Pd-Catalyzed Suzuki-Miyaura Coupling
The cornerstone of this improved synthesis is the second step: the Suzuki-Miyaura cross-coupling reaction between methyl 3,5-dichloro-2-picolinate and 3-chlorobenzeneboronic acid. Mechanistically, this transformation relies on the oxidative addition of the palladium catalyst (such as PdCl2 or PdCl2(dppf)) into the carbon-chlorine bond at the 5-position of the pyridine ring. The presence of the electron-withdrawing ester group at the 2-position and the chlorine at the 3-position creates a specific electronic environment that favors reactivity at the 5-position over the 3-position, although the 3-chloro substituent remains intact for subsequent functionalization. The use of inorganic bases like potassium carbonate or sodium carbonate facilitates the transmetallation step by activating the boronic acid species, while the aqueous-organic solvent system (e.g., DMF/Water) ensures optimal solubility for both the polar boronic acid and the organic halide.
Impurity control in this step is paramount for the quality of the final API. The patent specifies precise molar ratios of base (1.0 to 3.0 equivalents) and catalyst loading (0.005 to 0.5 equivalents) to minimize side reactions such as homocoupling of the boronic acid or dehalogenation. Following the reaction, the workup procedure involves extraction and recrystallization from ethyl acetate and n-hexane, which effectively removes residual palladium species and unreacted starting materials. This rigorous control over the catalytic cycle ensures that the resulting methyl 5-(3-chlorophenyl)-3-chloropyridine-2-carboxylate possesses the high chemical purity required for the subsequent nucleophilic aromatic substitution with glycine, preventing the carryover of genotoxic impurities or heavy metals into the final drug substance.
How to Synthesize 5-(3-chlorophenyl)-3-hydroxypyridine-2-carboxamide Derivatives Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the patented route. The process begins with the acid-catalyzed esterification of 3,5-dichloro-2-picolinic acid in methanol, followed by the critical palladium-catalyzed coupling step which builds the biaryl core. The final transformation involves the displacement of the remaining chloride by glycine under basic conditions, followed by acidification to precipitate the product. For detailed operational parameters, specific solvent volumes, and exact temperature ramps, please refer to the standardized synthesis guide below which encapsulates the critical process parameters defined in the patent examples.
- Esterification of 3,5-dichloro-2-picolinic acid with methanol and sulfuric acid to form methyl 3,5-dichloro-2-picolinate.
- Pd-catalyzed Suzuki coupling with 3-chlorobenzeneboronic acid to introduce the aryl group at the 5-position.
- Nucleophilic substitution with glycine followed by hydrolysis to yield the final hydroxypyridone acetic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers profound strategic advantages beyond mere technical elegance. The primary value driver is the drastic reduction in manufacturing costs achieved by eliminating expensive and time-consuming purification steps. By replacing five rounds of column chromatography with simple crystallization and filtration, the process significantly lowers solvent usage, reduces labor hours, and minimizes waste disposal costs. This efficiency gain translates directly into a more competitive cost structure for the final intermediate, allowing for better margin management in the face of fluctuating raw material prices. Furthermore, the use of commodity chemicals like methanol, sulfuric acid, and potassium carbonate ensures that the supply chain is not dependent on exotic or single-source reagents.
- Cost Reduction in Manufacturing: The economic impact of removing chromatographic purification cannot be overstated in the context of fine chemical production. Chromatography is inherently batch-limited, solvent-intensive, and difficult to automate, whereas crystallization is easily scalable and continuous. By designing a route where intermediates precipitate as high-purity solids, the process enables the use of standard stainless steel reactors without the need for specialized glass-lined vessels required for strong acid conditions found in other routes. This simplification of unit operations leads to substantial capital expenditure savings and lower operating expenses per kilogram of output.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply continuity. The avoidance of microwave reactors and high-pressure hydrogenation equipment means that production can be executed in standard multipurpose pharmaceutical plants without requiring specialized infrastructure. Additionally, the starting material, 3,5-dichloro-2-picolinic acid, is a stable and commercially available building block, reducing the risk of supply disruptions associated with custom-synthesized precursors. The mild reaction temperatures (below 130°C) also reduce the thermal load on facilities, enhancing operational safety and uptime.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The reduction in solvent volume and the elimination of silica gel waste from chromatography columns significantly lower the environmental footprint of the manufacturing process. The ability to recycle solvents like ethyl acetate and methanol further aligns with green chemistry principles. For supply chain heads, this means easier regulatory compliance and a smoother path through environmental audits, ensuring that production schedules are not delayed by waste treatment bottlenecks or regulatory hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within patent CN110903238A, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning tech transfer and procurement teams negotiating supply agreements.
Q: How does the new synthesis route improve yield compared to prior art?
A: The patented method (CN110903238A) reduces the synthetic sequence from 7 steps to just 3 steps, eliminating the need for multiple column chromatography purifications and harsh microwave conditions, thereby significantly improving overall yield and purity.
Q: What are the critical reaction conditions for the Suzuki coupling step?
A: The coupling utilizes PdCl2(dppf) or PdCl2 as a catalyst with bases like K2CO3 in solvents such as DMF or toluene at moderate temperatures (30-100°C), ensuring high regioselectivity at the 5-position of the pyridine ring.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process avoids hazardous high-pressure hydrogenation and extreme microwave temperatures (190°C), utilizing standard reflux and filtration techniques that are easily adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vadadustat Intermediate Supplier
The technical advancements detailed in patent CN110903238A represent a significant opportunity for optimizing the supply chain of HIF-PH inhibitors. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this novel chemistry for your commercial needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of intermediate meets the exacting standards required for API synthesis.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project timelines and budget targets. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact us directly to request specific COA data from our pilot batches and comprehensive route feasibility assessments. Let us collaborate to secure a stable, high-quality, and cost-effective supply of critical pharmaceutical intermediates for the global market.
