Scalable Manufacturing of Vadadustat Intermediates via Optimized Suzuki Coupling
The pharmaceutical landscape for treating anemia has been significantly reshaped by the development of hypoxia-inducible factor prolyl hydroxylase (HIF-PH) inhibitors, with Vadadustat emerging as a pivotal therapeutic agent. As the industry demands more efficient supply chains for such critical active pharmaceutical ingredients (APIs), the technological breakthroughs detailed in patent CN111320577A offer a transformative approach to synthesizing the core pyridine amide structures. This patent discloses a novel preparation method that addresses the longstanding inefficiencies of legacy synthetic routes, providing a robust framework for the industrial production of high-purity pharmaceutical intermediates. By leveraging a streamlined five-step sequence, manufacturers can now achieve superior product purity and yield while operating under significantly milder reaction conditions. For R&D directors and procurement strategists, understanding the nuances of this improved pathway is essential for securing a reliable Vadadustat intermediate supplier capable of meeting the rigorous demands of global regulatory bodies. The shift from complex, low-yielding processes to this optimized protocol represents a critical evolution in fine chemical manufacturing, ensuring that the supply of this life-saving medication remains stable and cost-effective.
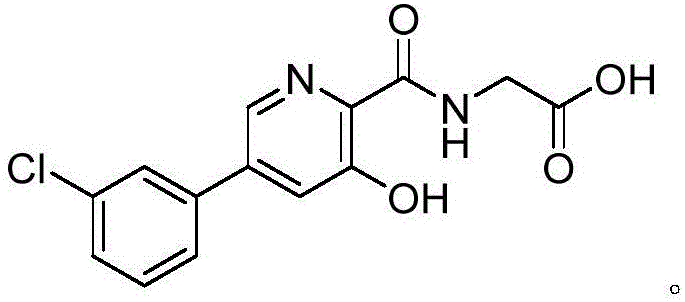
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations presented in CN111320577A, the synthesis of Vadadustat and its precursors was plagued by severe operational bottlenecks that hindered commercial viability. As illustrated in the legacy pathways, such as those disclosed in US20070299086, the conventional synthetic strategy typically involved a cumbersome seven-step sequence that resulted in a dismal overall yield of approximately 9 percent. A major impediment in these older methodologies was the reliance on extreme reaction conditions, including the necessity for microwave irradiation at temperatures reaching 190°C during the initial benzyl alcohol substitution step. Such harsh thermal requirements not only demand specialized and expensive reactor equipment but also pose significant safety risks when attempting to scale the process to multi-kilogram or ton-level batches. Furthermore, the purification protocols associated with these traditional routes were exceptionally labor-intensive, often necessitating up to five separate column chromatography operations to isolate the desired intermediates. This heavy dependence on chromatographic purification is economically unsustainable for large-scale manufacturing, as it consumes vast quantities of silica gel and solvents while drastically extending production lead times. Additionally, some alternative historical routes employed strong acidic conditions that imposed corrosive stress on reaction vessels, further complicating the engineering requirements for plant infrastructure and increasing maintenance costs.
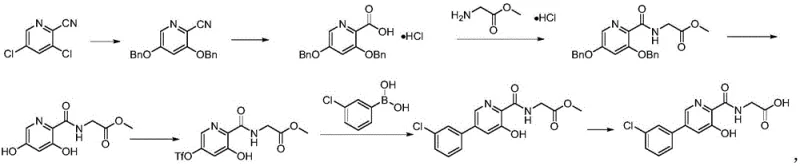
The Novel Approach
In stark contrast to the inefficiencies of the past, the novel approach outlined in the patent data introduces a sophisticated yet practical five-step synthesis that fundamentally re-engineers the construction of the pyridine amide core. This modern methodology replaces the hazardous microwave steps with standard thermal heating under reflux, allowing the reaction to proceed smoothly at much lower and safer temperatures. The strategic design of this route eliminates the need for repetitive column chromatography, substituting it with more scalable unit operations such as liquid-liquid extraction and crystallization, which are far more conducive to continuous processing. By optimizing the order of functional group transformations, specifically the timing of the Suzuki coupling and the installation of the glycine side chain, the new process achieves a dramatic improvement in material throughput. The result is a synthesis that not only delivers high-purity intermediates suitable for direct progression to the next step but also significantly reduces the environmental footprint by minimizing solvent waste. For supply chain managers, this transition from a fragile, low-yielding laboratory curiosity to a robust industrial process means a substantial reduction in the cost of goods sold and a marked increase in supply reliability. The ability to produce complex heterocyclic intermediates without exotic catalysts or extreme conditions underscores the commercial maturity of this technology.
Mechanistic Insights into Oxidative Coupling and Suzuki Cross-Coupling
The chemical elegance of this patented route lies in its clever utilization of oxidative amidation and palladium-catalyzed cross-coupling to construct the molecular architecture efficiently. The process initiates with the conversion of methyl 3,5-dihydroxypicolinate into a hydroxamic acid derivative, which serves as a activated acyl donor for the subsequent coupling with glycine methyl ester. In this critical second step, an oxidant such as iodine facilitates the formation of the amide bond under mild alkaline conditions, a transformation that avoids the use of toxic coupling reagents often found in peptide synthesis. Following the installation of the amino acid side chain, the synthesis employs a triflation strategy to convert the 5-hydroxyl group into a triflate leaving group. This activation is paramount for enabling the subsequent Suzuki-Miyaura cross-coupling reaction, where the electron-deficient pyridine ring undergoes nucleophilic attack by the organoboron species. The use of a palladium catalyst, such as PdCl2(dppf), ensures high regioselectivity and conversion rates even in the presence of the sensitive amide and hydroxyl functionalities. This mechanistic pathway demonstrates a deep understanding of electronic effects within the heterocyclic system, allowing for the precise introduction of the 3-chlorophenyl moiety without compromising the integrity of the rest of the molecule. The final hydrolysis step cleanly removes the methyl protecting group to reveal the free carboxylic acid, completing the synthesis of the target API intermediate with exceptional fidelity.
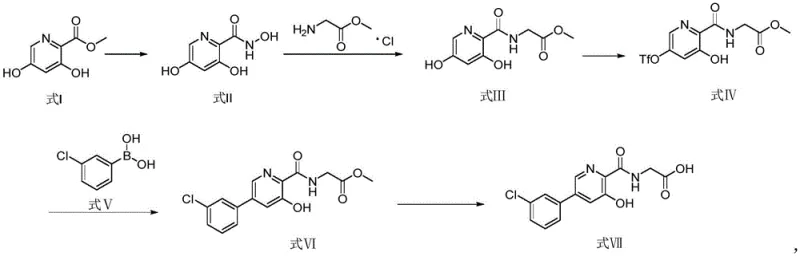
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of difficult-to-remove byproducts. Traditional routes often generated complex mixtures due to the harsh conditions required for C-N or C-C bond formation, leading to polymeric tars or over-alkylated species that were challenging to separate. In the new process, the mild nature of the oxidative coupling step ensures that the glycine side chain is installed cleanly, while the specificity of the palladium catalyst in the Suzuki step prevents homocoupling of the boronic acid. Furthermore, the intermediate isolation strategies, which rely on pH-controlled precipitation and solvent extraction, effectively partition the desired product from inorganic salts and unreacted starting materials. This inherent selectivity reduces the burden on downstream purification units and ensures that the final product meets stringent pharmaceutical specifications regarding residual metals and organic impurities. For quality assurance teams, this predictable impurity profile simplifies the validation process and accelerates the timeline for regulatory filing. The robustness of the catalytic cycle also means that the process is less sensitive to minor fluctuations in reagent quality, providing a wider operating window for plant operators. Ultimately, the mechanistic stability of this route translates directly into batch-to-batch consistency, a critical metric for any commercial API manufacturer.
How to Synthesize Vadadustat Efficiently
The implementation of this synthesis requires careful attention to reaction parameters, particularly during the oxidative coupling and triflation stages, to maximize yield and purity. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for process chemists looking to adopt this technology. It is crucial to maintain strict control over stoichiometry and temperature profiles to ensure the reproducibility of the high yields reported in the literature. Detailed standard operating procedures for each transformation are provided below to facilitate immediate technology transfer.
- React methyl 3,5-dihydroxypicolinate with hydroxylamine hydrochloride and base to form the hydroxamic acid intermediate.
- Perform oxidative coupling with glycine methyl ester hydrochloride using iodine as an oxidant to install the acetamide side chain.
- Protect the 5-position hydroxyl group via triflation using N-phenylbis(trifluoromethanesulfonimide).
- Execute a palladium-catalyzed Suzuki coupling with 3-chlorophenylboronic acid to introduce the aryl moiety.
- Hydrolyze the methyl ester under basic conditions followed by acidification to isolate the final Vadadustat product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The most significant advantage lies in the drastic simplification of the purification workflow, which eliminates the need for expensive and time-consuming column chromatography. By replacing chromatography with crystallization and extraction, manufacturers can achieve substantial cost savings in terms of both consumable materials and labor hours, directly impacting the bottom line. This reduction in processing complexity also shortens the overall cycle time for each batch, allowing facilities to increase their production capacity without the need for capital investment in new equipment. Furthermore, the avoidance of harsh microwave conditions and strong acids reduces the wear and tear on reactor vessels, lowering maintenance costs and extending the lifespan of existing plant infrastructure. These operational efficiencies translate into a more resilient supply chain that is better equipped to handle fluctuations in market demand. The use of readily available starting materials and common catalysts further mitigates the risk of raw material shortages, ensuring a steady flow of intermediates to downstream API producers. Consequently, this technology represents a strategic asset for companies seeking to optimize their manufacturing footprint and reduce the total cost of ownership for their pharmaceutical portfolios.
- Cost Reduction in Manufacturing: The elimination of column chromatography is a game-changer for cost efficiency, as this technique is notoriously expensive and difficult to scale in a commercial setting. By utilizing extraction and crystallization instead, the process significantly lowers the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical production. Additionally, the higher overall yield means that less raw material is wasted, further driving down the cost per kilogram of the final product. The mild reaction conditions also reduce energy consumption, as there is no need for high-temperature microwave reactors or extensive cooling systems for exothermic quenches. These cumulative savings allow suppliers to offer more competitive pricing while maintaining healthy profit margins, creating a win-win scenario for both manufacturers and buyers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more reliable supply of critical intermediates, minimizing the risk of production delays caused by equipment failure or purification bottlenecks. Since the process does not rely on specialized microwave equipment, it can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure means that production can be ramped up quickly across multiple sites if necessary, providing a buffer against supply disruptions. Moreover, the use of stable reagents and catalysts reduces the dependency on scarce or volatile raw materials, stabilizing the supply chain against market fluctuations. For procurement teams, this reliability translates into greater confidence in meeting delivery deadlines and fulfilling long-term supply agreements with pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this process aligns perfectly with modern green chemistry principles, making it easier to obtain regulatory approvals for large-scale production. The reduction in solvent usage and the avoidance of hazardous strong acids minimize the generation of toxic waste streams, simplifying wastewater treatment and disposal. The mild operating conditions also enhance workplace safety by reducing the risk of thermal runaways or pressure build-ups associated with high-temperature reactions. These factors contribute to a lower environmental footprint and reduced compliance costs, which are increasingly important metrics for sustainable manufacturing. As regulatory scrutiny on pharmaceutical manufacturing intensifies, adopting such an eco-friendly and scalable process provides a significant competitive advantage in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Vadadustat synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of the technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing operations.
Q: How does this new route improve upon previous Vadadustat synthesis methods?
A: Unlike prior art methods requiring harsh microwave conditions at 190°C and multiple column chromatography purifications, this patented route operates under mild thermal conditions and utilizes crystallization or extraction for purification, drastically simplifying the workflow.
Q: What is the overall yield advantage of this 5-step process?
A: While conventional routes reported in literature often suffer from cumulative yield losses resulting in single-digit overall yields (e.g., 9%), this optimized pathway maintains high individual step yields, significantly boosting the final output per batch.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the elimination of specialized microwave equipment and the reliance on standard unit operations like filtration and solvent extraction make this chemistry highly amenable to scale-up in multi-ton reactors without requiring exotic infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vadadustat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape, and we are uniquely positioned to leverage the advancements described in CN111320577A. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By adopting this optimized five-step synthesis, we can offer our clients a reliable Vadadustat intermediate supplier partnership that guarantees consistent quality and competitive pricing. Our facility is designed to handle complex heterocyclic chemistry with precision, utilizing advanced reactor systems that can accommodate the specific thermal and mixing requirements of the Suzuki coupling and oxidative amidation steps.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your sourcing strategy. Together, we can ensure a stable and cost-effective supply of this vital anemia treatment intermediate, supporting your mission to bring life-saving therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
