Revolutionizing Sitagliptin Intermediate Production: A Deep Dive into Enzymatic Catalysis and Commercial Scalability
Revolutionizing Sitagliptin Intermediate Production: A Deep Dive into Enzymatic Catalysis and Commercial Scalability
The pharmaceutical landscape for Type II diabetes treatment has been revolutionized by DPP-IV inhibitors, with Sitagliptin standing as a cornerstone therapy since its initial market entry. Patent CN116217440A introduces a groundbreaking preparation method for the critical Sitagliptin intermediate, specifically the BOC-protected butyric acid derivative known as Compound 1, which serves as the chiral backbone for the final active pharmaceutical ingredient. This innovation addresses the longstanding industry challenge of synthesizing complex fluorinated amino acid derivatives by shifting away from traditional transition metal catalysis toward a highly selective biocatalytic transamination process. By leveraging recombinant aspartase enzymes, the disclosed methodology achieves superior stereochemical control while operating under remarkably mild reaction conditions that avoid the extreme temperatures and pressures often associated with legacy synthetic pathways. For global supply chain stakeholders, this represents a pivotal opportunity to secure a more robust and environmentally sustainable source of high-purity pharmaceutical intermediates that aligns with modern green chemistry mandates.
Sitagliptin, chemically defined as 7-[(3R)-3-amino-1-oxo-4-(2,4,5-trifluorophenyl)butyl]-5,6,7,8-tetrahydro-3-(trifluoromethyl)-1,2,4-triazolo[4,3-a]pyrazine phosphate monohydrate, requires precise stereochemistry at the beta-amino acid position to ensure therapeutic efficacy. The structural complexity involving multiple fluorine atoms and a specific chiral center necessitates a synthesis route that guarantees high enantiomeric excess without introducing toxic contaminants. The patent explicitly targets the preparation of the key intermediate, often referred to as BOC-butyric acid derivative, which is subsequently condensed, hydrolyzed, and salified to form the final drug substance. Understanding the molecular architecture is crucial for R&D directors evaluating the feasibility of adopting this new biocatalytic route over established chemical methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
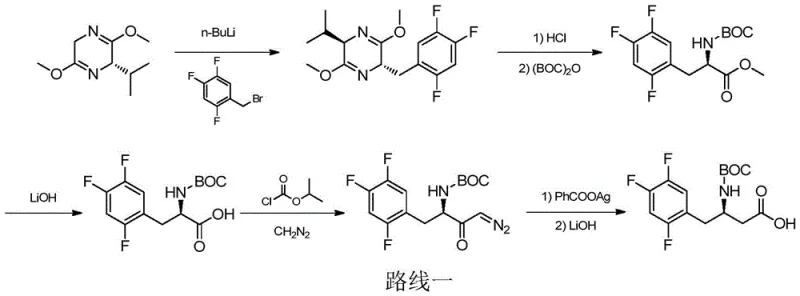
Historically, the synthesis of this critical intermediate has relied heavily on complex chemical catalysis involving expensive and potentially hazardous reagents. Early methodologies, such as those reported by Merck, utilized Schollkopf reagents which required harsh alkylation conditions with n-butyllithium and subsequent Arndt-Eistert reactions, creating significant safety risks and operational difficulties for large-scale manufacturing. Later iterations introduced enantioselective hydrogenation using Ruthenium-BINAP complexes or Rhodium-Josiphos catalysts, which, while effective, introduced the persistent problem of removing trace noble metals from the final product to meet stringent regulatory limits. These transition metal-catalyzed routes often involve multiple protection and deprotection steps, lengthy reaction times, and the use of costly chiral ligands that are difficult to recover, leading to inflated production costs and extended lead times for procurement teams managing global API supply chains.
The Novel Approach
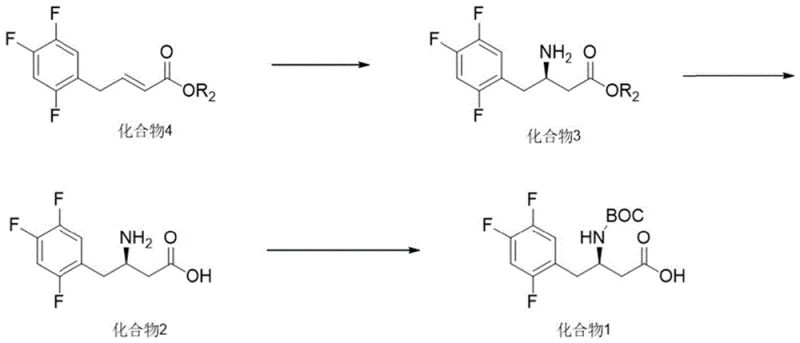
In stark contrast, the method disclosed in patent CN116217440A utilizes a streamlined three-step sequence initiated by an enzymatic transamination reaction that fundamentally alters the economic and environmental profile of the synthesis. The process begins with the conversion of an unsaturated ester precursor (Compound 4) into the chiral amine (Compound 3) using a recombinant aspartase enzyme in the presence of a simple amino donor like isopropylamine. This biocatalytic step proceeds under mild aqueous or semi-aqueous conditions at moderate temperatures, eliminating the need for high-pressure hydrogenation equipment and expensive noble metal catalysts entirely. The subsequent hydrolysis and Boc-protection steps are standard chemical transformations that are easily controlled and scalable, resulting in a final product with high purity and yield suitable for direct progression to the final API synthesis without extensive purification burdens.
Mechanistic Insights into Enzymatic Transamination
The core of this technological breakthrough lies in the stereoselective power of the recombinant aspartase enzyme, which facilitates the transfer of an amino group to the beta-position of the substrate with exceptional precision. The reaction mechanism involves the formation of a Schiff base intermediate between the enzyme-bound pyridoxal phosphate (PLP) cofactor and the amino donor, followed by the transfer of the amino group to the ketone or unsaturated substrate to generate the chiral amine product. This biological catalyst ensures that only the desired (R)-enantiomer is produced, effectively bypassing the need for downstream chiral resolution steps that typically result in a 50% loss of material in racemic syntheses. Furthermore, the enzyme operates efficiently in a biphasic system or with co-solvents like DMSO, allowing for the dissolution of the hydrophobic fluorinated substrate while maintaining the enzymatic activity in the aqueous phase, thus optimizing the reaction kinetics and overall space-time yield.
From an impurity control perspective, the absence of transition metals such as Palladium, Platinum, or Rhodium removes a major category of genotoxic impurities that require rigorous and costly clearance strategies in traditional routes. The enzymatic process generates benign byproducts, primarily acetone when isopropylamine is used as the donor, which can be easily removed during workup, unlike the complex organometallic residues found in metal-catalyzed hydrogenation. The specificity of the enzyme also minimizes the formation of regioisomers or over-reduced byproducts, leading to a cleaner crude profile that simplifies the crystallization of the final Boc-protected intermediate. This high level of selectivity translates directly into reduced solvent consumption and waste generation, aligning the manufacturing process with the principles of green chemistry and sustainability.
How to Synthesize Sitagliptin Key Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this biocatalytic route, emphasizing the importance of pH control and temperature management during the enzymatic step to maximize conversion rates. The process is designed to be robust, tolerating variations in substrate concentration while maintaining high enantiomeric purity, which is critical for ensuring consistent quality in commercial production batches. Detailed operational parameters regarding the ratio of enzyme to substrate, the choice of co-solvents, and the specific workup procedures for isolating the intermediate are essential for R&D teams looking to replicate these results in a pilot plant setting.
- Perform enzymatic transamination on Compound 4 using recombinant aspartase and an amino donor to generate the chiral amine Compound 3.
- Hydrolyze the ester group of Compound 3 under acidic or alkaline conditions to yield the free acid Compound 2.
- React Compound 2 with di-tert-butyl dicarbonate (Boc anhydride) in the presence of a base to obtain the final protected intermediate Compound 1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic route offers substantial strategic benefits that extend beyond mere technical feasibility to impact the bottom line directly. The elimination of noble metal catalysts and complex chiral ligands removes a significant variable cost component, as these materials are subject to volatile market pricing and supply constraints often driven by geopolitical factors affecting mining regions. Additionally, the simplified workflow reduces the number of unit operations required, which lowers capital expenditure on specialized equipment like high-pressure reactors and metal scavenging columns, thereby improving the overall return on investment for manufacturing facilities. The use of readily available starting materials and common solvents further enhances supply chain resilience, reducing the risk of production delays caused by the shortage of exotic reagents.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with biocatalysts leads to a drastic reduction in raw material costs, as enzymes can often be recycled or produced at a fraction of the cost of noble metals like Palladium or Rhodium. Furthermore, the removal of metal scavenging steps and the associated filtration media reduces consumable costs and waste disposal fees, contributing to a leaner and more cost-effective manufacturing process. The higher atom economy of the enzymatic transamination compared to stoichiometric chiral auxiliaries means less raw material is wasted, maximizing the yield per kilogram of input and driving down the cost of goods sold significantly.
- Enhanced Supply Chain Reliability: By relying on fermentation-derived enzymes and commodity chemicals rather than scarce precious metals, manufacturers can secure a more stable and predictable supply of critical inputs for Sitagliptin production. The robustness of the enzymatic process allows for flexible production scheduling and easier scale-up from pilot to commercial volumes without the need for specialized high-pressure infrastructure that often creates bottlenecks in API manufacturing. This flexibility ensures that supply chain heads can respond more agilely to market demand fluctuations, minimizing the risk of stockouts and ensuring continuous availability of this high-volume diabetes medication.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous-based nature of the enzymatic step significantly reduce the environmental footprint of the synthesis, lowering the burden on wastewater treatment facilities and reducing the generation of hazardous organic waste. This alignment with environmental regulations facilitates smoother regulatory approvals and reduces the compliance costs associated with handling toxic heavy metals and volatile organic solvents. The process is inherently safer, operating at near-atmospheric pressure and moderate temperatures, which lowers insurance premiums and operational risks associated with high-energy chemical processes, making it an ideal candidate for sustainable large-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel enzymatic synthesis route for Sitagliptin intermediates. These insights are derived directly from the patent data to provide clarity on the operational advantages and quality improvements offered by this technology. Understanding these details is vital for stakeholders evaluating the transition from traditional chemical synthesis to biocatalytic manufacturing.
Q: How does the enzymatic route improve impurity profiles compared to metal-catalyzed methods?
A: The enzymatic route eliminates the use of transition metals like Palladium, Platinum, and Ruthenium, thereby removing the risk of heavy metal residues in the final API and simplifying the purification process significantly.
Q: What are the cost advantages of using recombinant aspartase for this synthesis?
A: By replacing expensive chiral ligands and noble metal catalysts with biocatalysts, the process drastically reduces raw material costs and avoids the complex waste treatment associated with heavy metal disposal.
Q: Is this enzymatic process suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (40-50°C, atmospheric pressure) using commercially available solvents, making it highly adaptable for multi-ton commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of pharmaceutical intermediate manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to meet the rigorous demands of the global API market. Our state-of-the-art facilities are equipped to handle complex biocatalytic processes, ensuring that the enzymatic route described in patent CN116217440A can be implemented with the highest standards of quality and efficiency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Sitagliptin intermediate meets the exacting requirements of regulatory bodies worldwide, providing our partners with absolute confidence in supply continuity.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits of switching to this enzymatic process for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver high-quality Sitagliptin intermediates reliably and competitively.
