Advanced Iridium Complex Manufacturing: Bridging High-Potency Antitumor Activity with Scalable Production
The pharmaceutical landscape is constantly evolving in the search for more effective and less toxic antitumor agents, and patent CN110950914A represents a significant breakthrough in the field of organometallic oncology therapeutics. This patent discloses a novel iridium complex characterized by a unique coordination environment involving 5-chloro-8-hydroxyquinoline and 7,8-benzoquinoline ligands, offering a promising alternative to traditional platinum-based chemotherapy. The technical disclosure provides a robust synthetic pathway that utilizes readily available starting materials and mild reaction conditions, making it highly attractive for industrial scale-up. For R&D directors and procurement specialists alike, this technology addresses the critical need for compounds that overcome cisplatin resistance while minimizing systemic toxicity. The detailed crystallographic data and biological evaluation included in the patent confirm the structural integrity and potent bioactivity of the final product, establishing a solid foundation for further drug development initiatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemotherapy relies heavily on platinum complexes such as cisplatin, which, despite their efficacy, are plagued by severe limitations including dose-limiting nephrotoxicity, neurotoxicity, and the rapid development of drug resistance in tumor cells. These adverse effects often necessitate treatment discontinuation or dose reduction, severely compromising therapeutic outcomes for patients suffering from cervical and ovarian cancers. Furthermore, the synthesis of next-generation metal-based drugs often involves complex multi-step procedures requiring expensive catalysts, harsh reaction conditions, or difficult purification processes that drive up manufacturing costs. The lack of selectivity in many existing metal complexes means they attack both cancerous and healthy tissues, leading to the myelosuppression and organ damage that characterize current standard-of-care treatments. Consequently, there is an urgent demand for novel metallodrugs that can bypass these resistance mechanisms and offer a safer toxicity profile without sacrificing potency.
The Novel Approach
The synthesis method described in CN110950914A offers a transformative solution by employing a straightforward ligand substitution reaction that proceeds efficiently in ethanol-based solvent systems. By reacting 5-chloro-8-hydroxyquinoline with a 7,8-benzoquinoline iridium dimer, the process generates a stable, neutral iridium(III) complex with remarkable biological properties. This approach eliminates the need for exotic reagents or extreme temperatures, as the reaction can proceed effectively at moderate heating (35-100°C) or even at room temperature over a longer duration. The use of ethanol as a primary solvent not only aligns with green chemistry principles by reducing hazardous waste but also simplifies the downstream processing, as the product precipitates directly from the reaction mixture as high-purity reddish-brown crystals. This streamlined workflow significantly reduces the operational complexity typically associated with synthesizing heteroleptic iridium complexes, thereby enhancing the overall feasibility for commercial production.
Mechanistic Insights into Iridium Coordination and Ligand Exchange
The core of this technology lies in the precise coordination chemistry that stabilizes the iridium center within a distorted octahedral geometry, as evidenced by the single-crystal X-ray diffraction data provided in the patent. The complex features two cyclometalated 7,8-benzoquinoline ligands and one bidentate 5-chloro-8-hydroxyquinoline ligand, creating a rigid structure that likely contributes to its kinetic stability in physiological environments. The presence of the chloro-substituent on the hydroxyquinoline ring is particularly strategic, as it modulates the electronic properties and lipophilicity of the molecule, potentially enhancing cellular uptake and interaction with biological targets. The bond length and angle data confirm the strong chelation of the nitrogen and oxygen donors to the iridium atom, which prevents premature ligand dissociation—a common failure mode for less stable metal drugs. This structural robustness is key to the complex's ability to maintain its integrity until it reaches the intracellular target, thereby maximizing its therapeutic index.
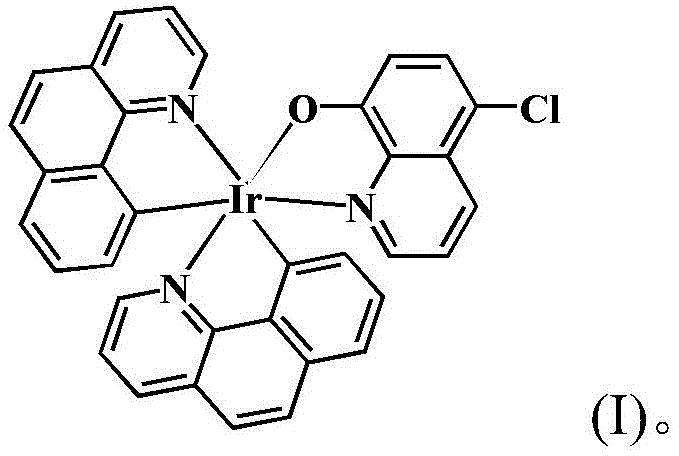
Furthermore, the mechanism of action appears to involve a high degree of selectivity towards malignant cells, distinguishing it sharply from non-selective cytotoxic agents. Biological assays reveal that while the complex exhibits potent inhibition against HeLa cervical cancer cells and cisplatin-resistant SK-OV-3 ovarian cancer cells, it shows negligible toxicity towards normal human liver HL-7702 cells. This differential activity suggests that the complex may exploit specific metabolic differences or transport mechanisms unique to tumor cells, rather than relying solely on DNA intercalation like cisplatin. The stability of the Ir-C and Ir-N bonds under physiological conditions ensures that the drug does not degrade into toxic free metal ions before reaching its site of action. For process chemists, understanding this stability is crucial, as it implies that the manufacturing process does not require stringent exclusion of moisture or air beyond standard pharmaceutical practices, further simplifying the production protocol.
How to Synthesize Iridium Complex Efficiently
The synthesis protocol outlined in the patent is designed for reproducibility and ease of execution, making it an ideal candidate for technology transfer from the laboratory to pilot plant scales. The procedure involves dissolving the iridium dimer precursor and the hydroxyquinoline ligand in a specific ratio of ethanol and co-solvents, followed by controlled heating to drive the equilibrium towards the desired heteroleptic product. Monitoring the reaction progress can be easily achieved using thin-layer chromatography (TLC), allowing operators to determine the optimal endpoint without sophisticated instrumentation. Once the reaction is complete, the product crystallizes out of the solution, enabling a simple filtration and drying step that yields the final API intermediate in high purity. For a detailed breakdown of the specific molar ratios, solvent volumes, and temperature profiles required to replicate this synthesis, please refer to the standardized guide below.
- Combine 5-chloro-8-hydroxyquinoline and 7,8-benzoquinoline iridium dimer in a reaction vessel containing an organic solvent mixture where ethanol is the primary component.
- Heat the reaction mixture to a temperature between 35°C and 100°C, or allow it to react at room temperature for an extended period to facilitate ligand exchange.
- Upon completion, cool the mixture to precipitate reddish-brown crystals, then filter and dry the solid product to obtain the target iridium complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the manufacturing route for this iridium complex offers substantial advantages in terms of cost efficiency and raw material availability. The reliance on ethanol as the primary solvent drastically reduces raw material costs compared to processes requiring specialized anhydrous solvents or expensive chlorinated hydrocarbons. Additionally, the ability to run the reaction at relatively low temperatures (as low as 35°C) translates to significant energy savings during the heating phase, lowering the overall utility burden on the manufacturing facility. The high yields reported, often exceeding 90% under optimized conditions, mean that less starting material is wasted, directly improving the cost of goods sold (COGS) for the final active pharmaceutical ingredient. These factors combine to create a highly economical production model that can withstand market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The elimination of complex purification steps, such as column chromatography, is a major driver for cost reduction in antitumor drug manufacturing. Since the product precipitates directly from the reaction mixture as crystals, the need for extensive solvent exchanges or silica gel treatments is removed, saving both time and consumable costs. Furthermore, the use of stoichiometric amounts of reagents minimizes excess waste, and the recovery of the ethanol solvent through distillation allows for a closed-loop system that further enhances economic efficiency. This streamlined downstream processing ensures that the final product meets stringent purity specifications without incurring the high operational expenses typically associated with organometallic synthesis.
- Enhanced Supply Chain Reliability: The starting materials, specifically 5-chloro-8-hydroxyquinoline and iridium trichloride derivatives, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor variations in temperature or mixing rates, ensuring consistent batch-to-batch quality even when scaling up to multi-kilogram quantities. This reliability is critical for maintaining a steady supply of the intermediate for downstream drug formulation, preventing delays in clinical trial material production or commercial launch timelines. The simplified logistics of handling non-hazardous solvents like ethanol also reduce regulatory burdens and transportation costs.
- Scalability and Environmental Compliance: The process is inherently scalable due to its homogeneous nature and the absence of gas-evolving steps or highly exothermic reactions that pose safety risks at large scales. The use of ethanol, a green solvent, aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and waste disposal. By avoiding heavy metal catalysts that require complex scavenging steps, the process generates a cleaner waste stream that is easier and cheaper to treat. This environmental compatibility not only reduces compliance costs but also enhances the sustainability profile of the final pharmaceutical product, a key consideration for modern healthcare stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel iridium complex, based on the detailed disclosures within the patent documentation. Understanding these aspects is vital for partners looking to integrate this technology into their existing oncology drug pipelines or research portfolios. The answers provided reflect the specific experimental data and optimization parameters established during the patent filing process.
Q: What distinguishes this iridium complex from traditional cisplatin drugs?
A: Unlike cisplatin, which often causes severe nephrotoxicity and drug resistance, this novel iridium complex demonstrates significantly higher selectivity. It exhibits potent activity against cervical (HeLa) and ovarian (SK-OV-3/DDP) cancer cells while maintaining extremely low toxicity (IC50 > 80 μM) towards normal human liver cells.
Q: Is ethanol strictly required for the synthesis of this complex?
A: Yes, the patent data indicates that ethanol is a critical component of the solvent system. While co-solvents like DMSO, water, or acetone can be used, the target compound is only generated when ethanol is present, preferably constituting at least 5% to 20% of the total solvent volume.
Q: What are the typical yields and reaction times for this process?
A: The process is highly efficient, with reported yields ranging from approximately 80% to over 92%. Reaction times vary based on temperature; heating at 35-100°C allows completion within 8 to 48 hours, whereas room temperature reactions may require up to 5 days.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iridium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this novel iridium complex in advancing the next generation of cancer therapies, and we are fully equipped to support its development from gram-scale research to commercial production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of the iridium complex meets the exacting standards required for clinical and commercial applications. Our commitment to excellence extends beyond mere manufacturing; we act as a strategic partner dedicated to optimizing your supply chain and accelerating your time to market.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis route can reduce your overall project costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this high-potency antitumor agent into your portfolio. Let us help you navigate the complexities of organometallic drug production with confidence and precision.
