Advanced Synthesis of PD-1 Ligand Intermediates for Commercial Scale-Up
The pharmaceutical industry is currently witnessing a paradigm shift in the development of immunomodulators, particularly those targeting the PD-1/PD-L1 pathway, which has emerged as a cornerstone in cancer immunotherapy. Patent CN113493423A introduces a groundbreaking method for synthesizing a key intermediate essential for these life-saving therapies, addressing critical challenges related to purity and metal residue that have long plagued traditional manufacturing processes. This novel approach leverages a strategic sequence of Suzuki coupling and oxidative cyclization to construct the complex biphenyl-benzoxazole scaffold with exceptional precision. By isolating the metal-catalyzed step to the very beginning of the synthesis, the method ensures that the final drug substance is virtually free from heavy metal contaminants, a requirement that is becoming increasingly stringent under global regulatory frameworks. For R&D directors and procurement specialists, this represents a significant advancement in reliable pharmaceutical intermediates supplier capabilities, offering a pathway to higher quality active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex heterocyclic intermediates for immunomodulators has been fraught with inefficiencies, primarily stemming from the reliance on multiple transition metal-catalyzed steps throughout the synthetic route. Traditional methodologies often necessitate palladium or copper catalysis in the late stages of synthesis, which creates a formidable challenge in removing trace metal residues to meet the rigorous parts-per-million standards required for clinical-grade materials. Furthermore, conventional routes frequently involve harsh reaction conditions and unstable intermediates that degrade easily, leading to inconsistent yields and a broad spectrum of difficult-to-separate impurities. These factors collectively drive up the cost reduction in immunomodulator manufacturing efforts, as extensive purification protocols such as preparative HPLC or repeated recrystallizations become mandatory. The cumulative effect is a supply chain that is vulnerable to delays and quality failures, making it difficult to ensure the commercial scale-up of complex pharmaceutical intermediates without compromising on economic viability or regulatory compliance.
The Novel Approach
In stark contrast, the methodology disclosed in CN113493423A revolutionizes the production landscape by strategically positioning the sole metal-catalyzed transformation at the inception of the synthetic sequence. This forward-thinking design allows for the bulk removal of catalyst residues early in the process, ensuring that subsequent transformations proceed in a metal-free environment, thereby safeguarding the integrity of the final molecule. The route utilizes a robust Suzuki coupling reaction facilitated by the S-Phos ligand to construct the biaryl core, followed by a series of highly selective functional group manipulations that avoid the need for further metal intervention. This streamlined workflow not only enhances the overall yield but also drastically simplifies the downstream processing requirements, making it an ideal candidate for high-purity PD-1 ligand intermediate production. The result is a manufacturing process that is both chemically elegant and commercially robust, providing a sustainable solution for reducing lead time for high-purity pharmaceutical intermediates.
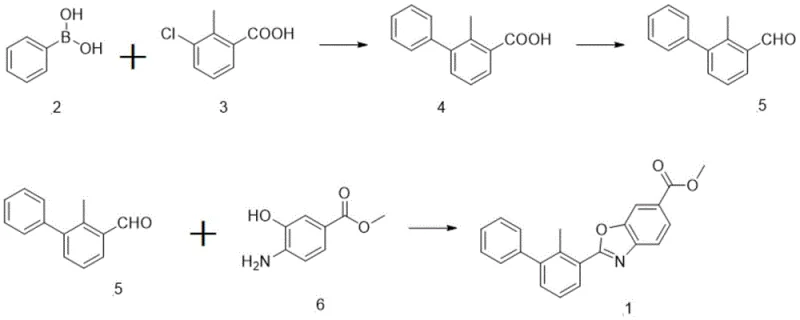
Mechanistic Insights into S-Phos Catalyzed Suzuki Coupling and Oxidative Cyclization
The cornerstone of this synthetic strategy lies in the initial Suzuki-Miyaura cross-coupling reaction, which efficiently joins the phenylboronic acid derivative with the chloro-substituted benzoic acid precursor. The utilization of the S-Phos ligand is critical here, as its bulky and electron-rich nature facilitates the oxidative addition and reductive elimination steps even with sterically hindered substrates, ensuring high conversion rates under mild thermal conditions. Following this, the carboxylic acid moiety is activated and converted into a Weinreb amide, a pivotal intermediate that allows for the controlled reduction to an aldehyde using diisobutylaluminum hydride (DIBAL-H) at cryogenic temperatures. This specific choice of reagent prevents over-reduction to the alcohol, a common side reaction that would derail the synthesis, thereby demonstrating a sophisticated level of impurity control mechanism design. The precision in managing these reactive intermediates is what distinguishes this patent from prior art, ensuring that the aldehyde functionality remains intact for the final cyclization event.
The final stage of the synthesis involves an elegant oxidative cyclization between the newly formed aldehyde and methyl 4-amino-3-hydroxybenzoate, mediated by the Dess-Martin periodinane oxidant. This reagent is chosen for its mildness and high selectivity, promoting the condensation and subsequent oxidation to form the benzoxazole ring system without affecting other sensitive functional groups like the ester moiety. The mechanism proceeds through the formation of an imine intermediate which is then oxidatively cyclized, locking the molecular architecture into the desired rigid planar structure essential for biological activity. By avoiding harsh acidic or basic conditions during this closure, the process minimizes the formation of hydrolysis byproducts and racemization risks, which are critical concerns for chiral or sensitive drug candidates. This mechanistic clarity provides R&D teams with confidence in the reproducibility and robustness of the high-purity pharmaceutical intermediates generated through this pathway.
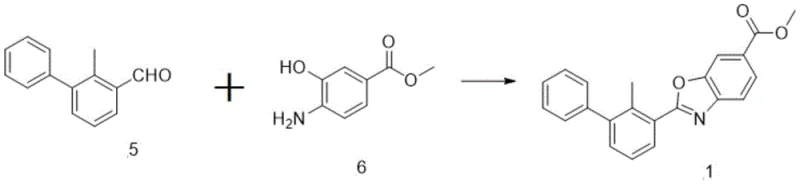
How to Synthesize PD-1 Ligand Intermediate Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to maximize yield and purity, particularly regarding temperature control and reagent stoichiometry during the reduction and oxidation phases. The process begins with the preparation of the biphenyl acid, followed by its conversion to the aldehyde, and concludes with the cyclization to form the target benzoxazole derivative. Each step has been optimized to balance reaction kinetics with operational safety, ensuring that the process is viable for transfer from laboratory bench to pilot plant scales. Detailed standardized synthesis steps are provided below to guide technical teams in replicating these results with high fidelity.
- Perform Suzuki coupling of phenylboronic acid and chloro-methyl-benzoic acid using S-Phos ligand to form the biphenyl backbone.
- Convert the carboxylic acid to an aldehyde via Weinreb amide formation followed by controlled DIBAL reduction at low temperatures.
- Execute oxidative cyclization with methyl 4-amino-3-hydroxybenzoate using Dess-Martin periodinane to finalize the benzoxazole core.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers profound advantages that directly address the pain points of procurement managers and supply chain heads in the pharmaceutical sector. By eliminating the need for multiple metal-catalyzed steps and complex purification sequences, the process inherently lowers the consumption of expensive reagents and reduces the operational burden on quality control laboratories. The simplified workflow translates into a more predictable manufacturing timeline, allowing for better inventory planning and reduced risk of stockouts for critical drug development programs. Furthermore, the use of common solvents and readily available starting materials mitigates the risk of supply chain disruptions associated with specialty chemicals, ensuring a steady flow of materials for continuous production runs.
- Cost Reduction in Manufacturing: The strategic placement of the metal catalysis step at the beginning of the synthesis significantly reduces the cost burden associated with metal scavenging and extensive purification later in the process. By avoiding the use of precious metal catalysts in the final stages, the manufacturer saves substantially on reagent costs and waste disposal fees associated with heavy metal containment. Additionally, the high selectivity of the Dess-Martin oxidation minimizes the formation of byproducts, which means less material is lost during purification, leading to improved overall mass efficiency. These factors combine to create a leaner manufacturing model that drives down the unit cost of the intermediate without sacrificing quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available starting materials such as phenylboronic acid and simple benzoic acid derivatives ensures that the supply chain is resilient against market fluctuations. Unlike routes that depend on exotic or custom-synthesized building blocks, this method leverages commodity chemicals that can be sourced from multiple vendors, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing, further enhances reliability by reducing the likelihood of batch failures. This stability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical partners who rely on just-in-time inventory systems.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable to large-scale reactors without requiring specialized high-pressure or cryogenic equipment beyond standard cooling capabilities. The reduction in metal usage aligns with green chemistry principles, lowering the environmental footprint of the manufacturing process and simplifying compliance with increasingly strict environmental regulations. Waste streams are easier to treat due to the absence of complex metal complexes, facilitating smoother regulatory approvals for manufacturing sites. This environmental compatibility not only reduces liability but also enhances the corporate sustainability profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the patent specifications and practical manufacturing considerations to provide clarity for decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this route for your specific drug development pipeline.
Q: How does this synthesis route minimize metal contamination?
A: The patented method confines metal catalysis to the initial Suzuki coupling step, which is structurally distant from the final active molecule, allowing for effective removal before critical stages.
Q: What purification methods are required for the final compound?
A: The process is designed to minimize impurities, requiring only silica gel column purification at the final stage to achieve high-purity specifications suitable for pharmaceutical applications.
Q: Is this route suitable for large-scale commercial production?
A: Yes, the use of readily available starting materials and robust reaction conditions like room temperature stirring and standard solvent systems supports scalable manufacturing without exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PD-1 Ligand Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the race to bring new immunotherapies to market, and we are fully equipped to support the commercialization of this advanced synthesis route. Our facilities boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in preclinical development or full-scale commercial launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that you can trust us to deliver materials that facilitate smooth regulatory filings and clinical trials.
We invite you to collaborate with our technical team to explore how this synthesis method can optimize your specific supply chain needs and reduce your overall development costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project volume and timeline. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your path to market while ensuring the highest levels of product integrity and supply security.
