Advanced Iridium Complexes for Oncology: Technical Breakthroughs and Commercial Scalability
The pharmaceutical landscape is continuously evolving in the search for more effective antineoplastic agents that minimize systemic toxicity while maximizing therapeutic efficacy. Patent CN111072727B introduces a groundbreaking class of iridium complexes constructed from 8-hydroxyquinoline derivatives and 2-phenylpyridine iridium dimers, representing a significant leap forward in organometallic medicinal chemistry. These novel compounds have demonstrated remarkable biological activity against cervical and ovarian cancer cell lines, surpassing the performance of traditional platinum-based drugs like cisplatin in specific assays. Furthermore, the safety profile is exceptionally promising, with toxicity to normal human liver cells remaining minimal, indicated by IC50 values exceeding 80 μM. This dual capability of high potency and low off-target toxicity addresses a critical unmet need in oncology drug development, positioning these molecules as prime candidates for next-generation targeted therapies.
Beyond their therapeutic potential, these iridium complexes offer a unique theranostic advantage through their ability to perform fluorescence localization within tumor cells. Specifically, they target the mitochondria, allowing researchers and clinicians to visualize drug distribution and mechanism of action in real-time. This intrinsic imaging capability eliminates the need for external labeling, simplifying the drug formulation process and reducing potential immunogenicity associated with conjugated dyes. For R&D directors and procurement specialists, this means a streamlined path from bench to bedside, with a single molecule serving both diagnostic and therapeutic roles. The structural versatility of the 8-hydroxyquinoline ligand allows for fine-tuning of physicochemical properties, ensuring that the supply chain can adapt to varying purity and specification requirements for different clinical phases.
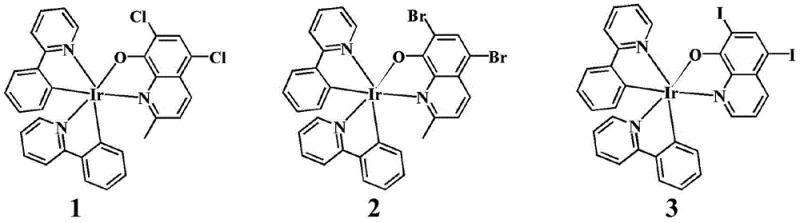
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemotherapy regimens have long relied heavily on platinum-based coordination compounds, which, while effective, are plagued by severe limitations that hinder patient quality of life and treatment compliance. The primary mechanism of these drugs involves binding to double-stranded DNA to inhibit replication, but this action is non-selective, affecting rapidly dividing healthy cells alongside tumor cells. This lack of selectivity leads to notorious side effects such as nephrotoxicity, neurotoxicity, and severe nausea, often necessitating dose reductions that compromise therapeutic outcomes. Moreover, the emergence of drug resistance in tumor cells, particularly in ovarian cancer lines like SK-OV-3/DDP, has rendered many first-line platinum therapies ineffective over time. From a manufacturing perspective, the purification of platinum drugs often requires rigorous removal of residual heavy metals to meet stringent regulatory standards, adding complexity and cost to the production process.
The Novel Approach
The synthetic strategy outlined in patent CN111072727B offers a sophisticated alternative by leveraging the unique electronic and steric properties of cyclometalated iridium(III) centers. Unlike the static binding of platinum drugs, these iridium complexes interact with cellular components through multiple pathways, including mitochondrial disruption, which triggers apoptosis via intrinsic signaling cascades. The synthesis method is remarkably robust, utilizing a straightforward ligand exchange reaction between a pre-formed iridium dimer and functionalized 8-hydroxyquinoline derivatives. This approach avoids the use of harsh reagents or extreme conditions, relying instead on moderate thermal energy in common solvent systems. The result is a series of complexes, such as Ir1 through Ir5, that maintain high structural integrity and purity. This methodological shift not only enhances biological performance but also simplifies the manufacturing workflow, making it an attractive option for cost-conscious procurement teams looking to optimize their API intermediate sourcing strategies.
Mechanistic Insights into Iridium-Carbon Bond Formation and Ligand Coordination
The core of this technology lies in the precise formation of the iridium-carbon and iridium-nitrogen bonds that stabilize the octahedral geometry of the complex. The reaction initiates with the cleavage of the chloro-bridged iridium dimer, which serves as a reactive precursor, allowing the 8-hydroxyquinoline derivative to coordinate through its nitrogen and oxygen atoms. This bidentate coordination creates a rigid chelate ring that enhances the kinetic stability of the complex in physiological environments. The presence of halogen substituents on the quinoline ring, such as chlorine, bromine, or iodine, plays a crucial role in modulating the lipophilicity and electronic density of the metal center. These modifications directly influence the cellular uptake and mitochondrial affinity of the final drug candidate. Understanding this mechanistic nuance is vital for R&D directors, as it provides a clear roadmap for structure-activity relationship (SAR) studies to further optimize potency and pharmacokinetic profiles without altering the fundamental synthetic route.
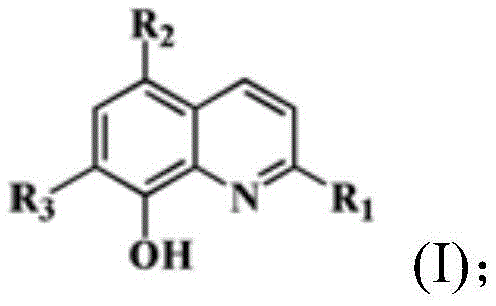
Impurity control is another critical aspect where this novel chemistry excels, particularly concerning the removal of unreacted starting materials and metal residues. The crystallization behavior of these iridium complexes is highly favorable, often precipitating as reddish-brown crystals directly from the reaction mixture upon cooling. This spontaneous crystallization acts as an inherent purification step, significantly reducing the burden on downstream processing units. The patent data indicates that single-crystal X-ray diffraction confirms high structural purity, with well-defined bond lengths and angles that match theoretical predictions. For supply chain managers, this implies a more predictable production timeline with fewer batches rejected due to out-of-specification impurities. The ability to achieve high purity through simple crystallization rather than complex chromatographic separations translates directly into lower manufacturing costs and a more reliable supply of high-purity pharmaceutical intermediates for clinical trials and commercial production.
How to Synthesize Iridium Complexes Efficiently
The synthesis of these bioactive iridium complexes is designed for operational simplicity and scalability, making it accessible for both laboratory research and industrial manufacturing. The process begins with the preparation of the 2-phenylpyridine iridium dimer, which can be sourced or synthesized using established literature methods, followed by the stoichiometric addition of the specific 8-hydroxyquinoline derivative. The choice of solvent is critical, with ethanol playing a pivotal role in facilitating the reaction kinetics and product crystallization. Detailed standard operating procedures regarding temperature ramps, stirring rates, and work-up protocols are essential to ensure batch-to-batch consistency. The following guide outlines the standardized synthesis steps derived from the patent examples to assist technical teams in replicating this high-value chemistry.
- Prepare the 2-phenylpyridine iridium dimer precursor and select the appropriate 8-hydroxyquinoline derivative based on desired halogen substitution.
- Dissolve reactants in an organic solvent system containing ethanol, ensuring a volume ratio of ethanol greater than 5 percent for optimal reaction kinetics.
- Heat the mixture to between 35°C and 100°C for 8 to 48 hours to facilitate coordination and crystallization of the target iridium complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel iridium complex synthesis offers tangible benefits that extend beyond mere technical performance. The reliance on commercially available starting materials, such as substituted hydroxyquinolines and iridium salts, ensures a stable raw material supply base that is not subject to the geopolitical volatility often associated with exotic reagents. Furthermore, the reaction conditions are mild, operating at temperatures ranging from 35°C to 100°C, which reduces energy consumption and lowers the safety risks associated with high-pressure or cryogenic processes. This operational simplicity allows for flexible manufacturing scheduling and reduces the need for specialized reactor infrastructure, thereby lowering capital expenditure requirements for scaling up production capacity to meet market demand.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal catalysts or complex purification columns, relying instead on straightforward crystallization for isolation. This simplification of the downstream processing significantly reduces solvent usage and waste generation, leading to substantial cost savings in utility and disposal expenses. Additionally, the high yields reported in the patent examples, often exceeding 80 percent and reaching over 90 percent in optimized conditions, mean that less raw material is wasted per kilogram of final product. These efficiency gains compound over large-scale production runs, offering a competitive pricing structure for the final pharmaceutical intermediate without compromising on quality or purity standards required by regulatory bodies.
- Enhanced Supply Chain Reliability: The robustness of the reaction chemistry ensures high batch consistency, which is critical for maintaining uninterrupted supply to downstream drug manufacturers. The use of common organic solvents like ethanol and water mixtures reduces dependency on specialized or hazardous solvent supply chains that are prone to disruption. Moreover, the reaction time can be adjusted based on production needs, with the option to run reactions at room temperature for longer durations or apply heat for faster turnover. This flexibility allows supply chain planners to optimize inventory levels and respond quickly to fluctuations in demand, ensuring that critical oncology drug pipelines remain stocked with high-quality intermediates.
- Scalability and Environmental Compliance: Scaling this synthesis from gram to kilogram or ton scale is facilitated by the homogeneous nature of the reaction mixture and the absence of gas evolution or exothermic spikes. The environmental footprint is minimized through the use of ethanol, a green solvent, and the reduction of heavy metal waste due to the efficient incorporation of iridium into the final product. This alignment with green chemistry principles supports corporate sustainability goals and simplifies the regulatory approval process for new drug filings. The ability to produce these complex organometallic compounds in an environmentally responsible manner adds significant value to the supply chain, appealing to partners who prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these iridium complexes. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details helps mitigate risks associated with technology transfer and ensures that all stakeholders have a clear expectation of the material's capabilities and limitations.
Q: What is the primary advantage of these iridium complexes over cisplatin?
A: The disclosed iridium complexes exhibit significantly higher biological activity against cervical and ovarian cancer cells while demonstrating extremely low toxicity to normal human liver cells, with IC50 values greater than 80 μM.
Q: Can the synthesis process be scaled for industrial production?
A: Yes, the synthesis utilizes common organic solvents like ethanol and operates under moderate heating conditions (35-100°C), making it highly amenable to commercial scale-up without requiring exotic high-pressure equipment.
Q: Do these complexes offer any diagnostic capabilities?
A: Yes, the complexes possess intrinsic fluorescence properties that allow for mitochondrial localization in tumor cells, enabling simultaneous therapeutic and imaging applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iridium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iridium complexes described in patent CN111072727B for the future of oncology treatment. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including residual metal analysis and chiral purity, which are critical for organometallic pharmaceuticals. We understand the nuances of handling air-sensitive or light-sensitive intermediates and have established protocols to maintain product integrity throughout the manufacturing and logistics process.
We invite you to collaborate with us to leverage this advanced chemistry for your drug development pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and route feasibility assessments that validate the commercial viability of this synthesis. Whether you require custom synthesis of specific halogenated derivatives or large-scale supply of the standard complexes, we are committed to delivering excellence. Contact us today to discuss how we can support your mission to bring safer and more effective cancer therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
