Scaling High-Purity Tulathromycin Production via Gradient Cooling Crystallization Technology
Scaling High-Purity Tulathromycin Production via Gradient Cooling Crystallization Technology
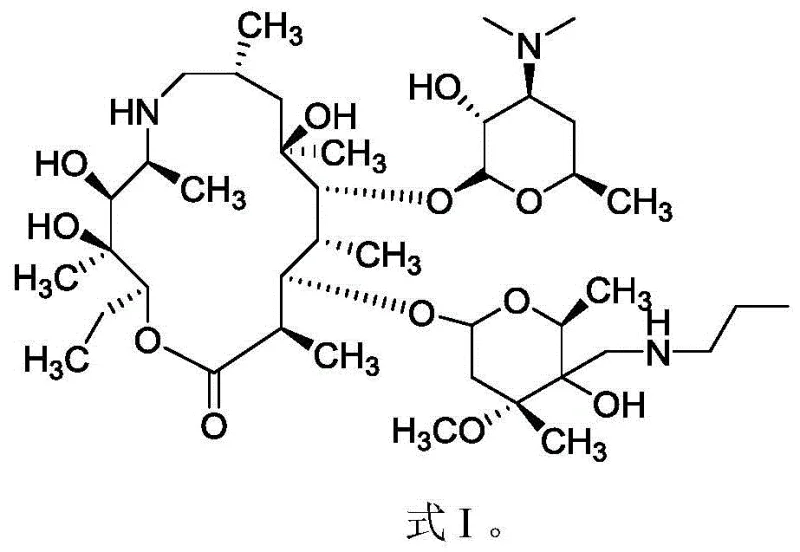
The pharmaceutical landscape for veterinary antibiotics is increasingly demanding higher purity standards, particularly for injectable formulations used in treating severe respiratory diseases in livestock. Patent CN111285910A introduces a transformative refining and purifying process for tulathromycin, a critical macrolide antibiotic used against pathogens like Actinobacillus pleuropneumoniae and Pasteurella. This technology addresses the longstanding industry challenge where crude products typically exhibit purity levels below 98 percent, rendering them unsuitable for high-value injectable applications. By implementing a sophisticated gradient cooling crystallization technique, this method effectively eliminates impurity entrapment, achieving purity levels exceeding 99 percent while maintaining robust yields. For R&D directors and supply chain managers, this represents a pivotal shift towards more reliable and compliant manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of tulathromycin has relied on solvent systems such as dichloromethane mixed with n-heptane or pentane, often involving rapid cooling or room temperature filtration. These traditional approaches suffer from a fundamental thermodynamic flaw: the crystallization rate is rarely controlled with precision, leading to rapid nucleation and crystal growth. When crystals form too quickly, impurities present in the mother liquor become physically trapped or occluded within the growing crystal lattice, a phenomenon known as inclusion. Consequently, despite multiple recrystallization attempts, the final purity often stagnates below the critical 98 percent threshold required for injectable grade materials. Furthermore, the reliance on halogenated solvents like dichloromethane introduces significant environmental and regulatory burdens, complicating waste disposal and increasing the overall cost of compliance for manufacturers.
The Novel Approach
The innovative process detailed in the patent overcomes these deficiencies by utilizing a tailored mixed solvent system comprising isopropyl ether and isoamyl alcohol, coupled with a precise multi-stage gradient cooling profile. Unlike the abrupt temperature drops of conventional methods, this approach gradually reduces the solution temperature through specific intervals, such as cooling from dissolution temperatures down to 25-30°C, then to 5-10°C, and finally to sub-zero conditions. This deliberate slowing of the crystallization kinetics allows the tulathromycin molecules to arrange themselves into a highly ordered lattice, effectively excluding impurity molecules that do not fit the crystal structure. The result is a dramatic improvement in product quality, consistently achieving purity greater than 99 percent, which opens up the lucrative market for injectable veterinary medicines while simultaneously simplifying the solvent recovery process due to the absence of chlorinated compounds.
Mechanistic Insights into Gradient Cooling Crystallization
The core scientific breakthrough of this purification strategy lies in the manipulation of supersaturation levels through thermal control. In standard crystallization, a sharp decrease in temperature creates a high degree of supersaturation instantly, driving a burst of nucleation events that compete for available solute. This chaotic environment fosters the rapid growth of imperfect crystals. In contrast, the gradient cooling method described maintains the system in a metastable zone for extended periods at each temperature plateau. By holding the temperature at intermediate points like 25-30°C and 5-10°C for durations ranging from 0.5 to 5 hours, the process ensures that crystal growth occurs primarily on existing nuclei rather than forming new ones. This Ostwald ripening effect allows smaller, less perfect crystals to dissolve and redeposit onto larger, more stable crystals, thereby enhancing the overall crystallinity and purity of the final bulk solid.
Furthermore, the selection of isopropyl ether and isoamyl alcohol is not arbitrary but is based on a precise matching of solvent polarity to the solute. Tulathromycin possesses complex polar and non-polar regions within its macrolide structure. The binary solvent system provides a solubility profile that is steep enough to allow for crystallization upon cooling but gentle enough to prevent 'oiling out' or amorphous precipitation. The isoamyl alcohol likely acts as a co-solvent that modulates the solvation shell around the tulathromycin molecules, reducing the energy barrier for orderly lattice incorporation. This synergistic solvent effect, combined with the kinetic control of gradient cooling, ensures that impurities remain in the solution phase rather than being mechanically entrapped, resulting in a product that meets the stringent specifications required for global pharmaceutical distribution.
How to Synthesize High-Purity Tulathromycin Efficiently
Implementing this purification protocol requires careful attention to solvent ratios and thermal ramping rates to maximize both yield and purity. The process begins with the dissolution of crude tulathromycin, typically having an initial purity of 90-97 percent, in the optimized solvent mixture at elevated temperatures between 50°C and 60°C. Once a clear solution is obtained, the critical gradient cooling phase commences, requiring precise reactor control to maintain the specified temperature plateaus for the designated stirring times. This step-by-step thermal descent is the key differentiator that separates this high-yield process from lower-quality conventional methods. For a detailed breakdown of the specific operational parameters and equipment requirements, please refer to the standardized synthesis guide below.
- Dissolve crude tulathromycin (90-97% purity) in a mixed solvent of isopropyl ether and isoamyl alcohol at 50-60°C under stirring.
- Execute a multi-stage gradient cooling protocol: cool to 25-30°C, then 5-10°C, and finally -10-0°C, maintaining stirring at each stage to control crystal growth.
- Filter the crystallized product, wash with precooled isopropyl ether, and dry to obtain high-purity tulathromycin with yields exceeding 90%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology offers substantial strategic benefits beyond mere technical specification improvements. The shift away from halogenated solvents like dichloromethane to more benign ethers and alcohols significantly reduces the regulatory overhead associated with hazardous waste disposal and worker safety monitoring. This transition not only aligns with modern green chemistry initiatives but also mitigates the risk of supply chain disruptions caused by tightening environmental regulations on chlorinated solvents. Additionally, the high yield consistency reported in the patent examples, often exceeding 90 percent, implies a more efficient utilization of raw materials, directly translating to improved cost structures and reduced waste generation per kilogram of active pharmaceutical ingredient produced.
- Cost Reduction in Manufacturing: The elimination of expensive and heavily regulated chlorinated solvents drastically simplifies the solvent recovery and waste treatment infrastructure required on-site. By utilizing widely available industrial solvents like isopropyl ether and isoamyl alcohol, manufacturers can achieve significant cost savings in raw material procurement and disposal fees. Furthermore, the high recovery yield minimizes the loss of valuable intermediate material, ensuring that the overall cost of goods sold is optimized without compromising on the stringent quality standards demanded by the veterinary pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of this crystallization process against minor fluctuations in operating conditions ensures consistent batch-to-batch quality, which is critical for maintaining long-term supply contracts with major animal health companies. The use of common, non-restricted solvents enhances supply security, as these chemicals are readily available from multiple global suppliers, reducing the risk of bottlenecks that often plague specialized reagent supply chains. This reliability allows for more accurate production planning and inventory management, ensuring that delivery timelines for critical veterinary antibiotics are met without delay.
- Scalability and Environmental Compliance: The gradient cooling technique is inherently scalable, as the thermal dynamics involved can be effectively managed in large-scale industrial reactors using standard jacketed vessels and chillers. The process avoids the need for complex anti-solvent addition pumps or high-shear mixing equipment, simplifying the engineering requirements for scale-up from pilot plant to commercial tonnage production. Moreover, the improved purity profile reduces the need for downstream reprocessing or additional purification steps, streamlining the manufacturing workflow and reducing the overall environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced purification technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is conventional tulathromycin purification insufficient for injectable applications?
A: Conventional methods often result in purity levels below 98% because rapid crystallization traps impurities within the crystal lattice. This level of purity is generally inadequate for injectable veterinary formulations which require stricter impurity profiles to ensure safety and efficacy.
Q: How does the gradient cooling method improve crystal purity?
A: By deliberately controlling the cooling rate through specific temperature stages, the crystal growth rate is significantly reduced. This slower growth prevents the occlusion of impurities during lattice formation, allowing for a much cleaner crystal structure and final purity exceeding 99%.
Q: What is the advantage of using isopropyl ether and isoamyl alcohol?
A: This specific solvent mixture offers a polarity that matches tulathromycin effectively, preventing overly rapid precipitation. It facilitates the gradient cooling process, ensuring that the solubility changes gradually rather than abruptly, which is critical for high-quality crystal formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tulathromycin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to high-purity manufacturing processes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the gradient cooling crystallization process are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the impurity profiles of complex macrolides, guaranteeing that every batch of tulathromycin meets the exacting standards required for injectable veterinary applications globally.
We invite you to collaborate with us to optimize your supply chain for this critical antibiotic. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our refined process can lower your total landed cost. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can secure your position as a leader in the veterinary pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
