Advanced Manufacturing of Rosuvastatin Aldehyde Intermediates for Global Pharma Supply Chains
The global demand for lipid-lowering agents continues to drive innovation in the synthesis of key statin intermediates. Patent CN101100459B introduces a transformative methodology for preparing (E)-3-[4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-methylsulfonylamino)pyrimidin-5-yl]-prop-2-en-1-al, a critical building block for Rosuvastatin Calcium. This intellectual property addresses longstanding inefficiencies in prior art by replacing hazardous reagents with safer, more accessible phosphorus-based alternatives. For R&D directors and procurement specialists, this patent represents a pivotal shift towards greener, more cost-effective manufacturing protocols that do not compromise on purity or stereochemical integrity. The disclosed route utilizes standard Wittig or Horner-Wadsworth-Emmons chemistry, leveraging readily available phosphonium salts or phosphonate esters to construct the crucial carbon-carbon double bond with high selectivity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for this specific pyrimidine aldehyde intermediate, such as those disclosed in earlier literature like CN1763015A, suffer from severe operational and safety drawbacks that hinder large-scale commercialization. As illustrated in the reaction scheme below, legacy methods often rely on the preparation of cyanomethylphosphonate esters, which require the use of highly toxic chloroacetonitrile and triethyl phosphite. These precursors are not only difficult to source commercially but also necessitate complex purification steps like molecular distillation to achieve the required purity levels. Furthermore, the subsequent reduction steps typically employ diisobutylaluminum hydride (DIBAL-H) at cryogenic temperatures ranging from -20°C to -50°C. This requirement for deep cooling imposes a massive energy burden on manufacturing facilities and introduces significant safety risks associated with handling pyrophoric reagents on a multi-ton scale.
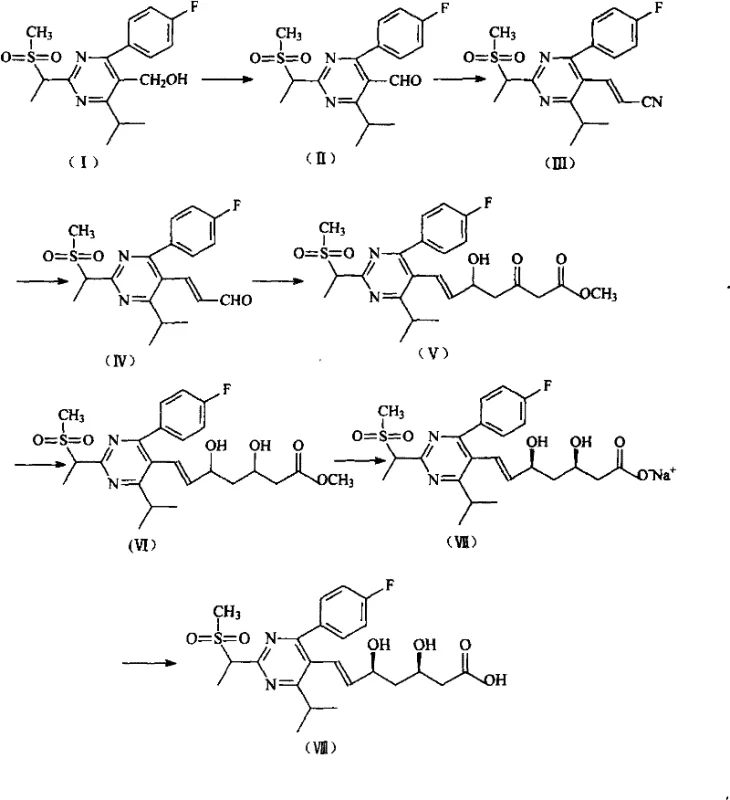
The Novel Approach
In stark contrast, the methodology outlined in CN101100459B circumvents these bottlenecks by employing a direct olefination strategy that operates under much milder conditions. Instead of navigating the treacherous landscape of cyanide chemistry and cryogenic reductions, the novel process reacts a stable phosphonium salt or phosphonate ester directly with the pyrimidine aldehyde precursor in the presence of a base. This one-pot transformation generates a protected acetal or iminium intermediate which is subsequently hydrolyzed under acidic conditions to yield the target aldehyde. The elimination of extreme temperature requirements and toxic starting materials drastically simplifies the engineering controls needed for production. This approach not only enhances operator safety but also significantly reduces the three wastes (waste water, waste gas, and solid waste) associated with the manufacturing process, aligning perfectly with modern environmental compliance standards.
Mechanistic Insights into Phosphorus-Mediated Olefination
The core of this technological advancement lies in the precise control of the phosphorus ylide formation and its subsequent reaction with the electrophilic carbonyl center. In the first stage, a strong base such as potassium tert-butoxide, sodium hydride, or n-butyllithium deprotonates the phosphonium salt or phosphonate ester to generate a reactive nucleophilic species. This ylide then attacks the carbonyl carbon of the 4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-methanesulfonamido)pyrimidine-5-carbaldehyde. The reaction proceeds through a four-membered oxaphosphetane intermediate (in the case of Wittig) or a similar transition state, which collapses to form the carbon-carbon double bond with high (E)-stereoselectivity. The use of specific protecting groups on the resulting alcohol, such as acetals or imines, prevents over-reaction or polymerization, ensuring that the delicate aldehyde functionality is preserved until the final deprotection step.
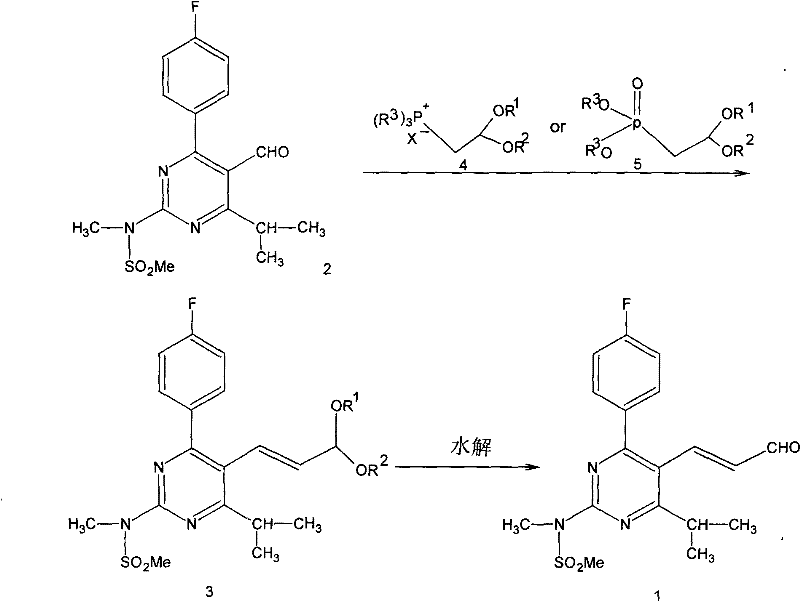
Impurity control is inherently built into this mechanistic pathway due to the stability of the intermediates and the specificity of the hydrolysis step. Unlike reduction methods that can lead to over-reduction to the primary alcohol or incomplete reduction leaving nitrile impurities, the olefination-hydrolysis sequence offers a clean cut-off point. The acidic hydrolysis conditions (pH ≤ 5) are carefully tuned to cleave the acetal or imine linkage without affecting the sensitive sulfonamide group or the fluorophenyl ring. This selectivity minimizes the formation of side products such as des-fluoro analogs or sulfonamide hydrolysis byproducts. Furthermore, the byproduct of the olefination, typically triphenylphosphine oxide, is chemically inert and easily separated from the organic product layer, allowing for potential recycling and further reducing the impurity load in the final API intermediate.
How to Synthesize Rosuvastatin Aldehyde Intermediate Efficiently
The practical execution of this synthesis involves a straightforward sequence that is highly amenable to batch processing in standard stainless steel reactors. The process begins with the activation of the phosphorus reagent in a dry, inert atmosphere using solvents like tetrahydrofuran (THF) or dimethyl sulfoxide (DMSO). Once the ylide is formed, the pyrimidine aldehyde is introduced, and the mixture is stirred at temperatures ranging from -50°C to 50°C depending on the specific base and solvent system employed. Following the formation of the protected intermediate, the reaction mixture is quenched and subjected to acidic hydrolysis using mineral acids like hydrochloric acid or organic acids like oxalic acid. This robust protocol ensures high reproducibility and ease of scale-up from laboratory benchtop to commercial production lines.
- React phosphonium salt or phosphonate ester with a strong base in an organic solvent to generate the active ylide species.
- Add the pyrimidine aldehyde precursor to the reaction mixture to form the protected acetal or iminium intermediate.
- Perform acidic hydrolysis under mild conditions to cleave the protecting group and isolate the final target aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic benefits regarding cost stability and supply continuity. By shifting away from bespoke, hazardous reagents like cyanomethylphosphonate, manufacturers can source raw materials from a broader, more competitive supplier base. The elimination of cryogenic processing steps removes a major bottleneck in plant throughput, allowing for faster batch cycles and reduced utility consumption. Additionally, the ability to recycle triphenylphosphine oxide creates a closed-loop value stream that mitigates raw material price volatility. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of global pharmaceutical markets without the risk of production stoppages due to reagent shortages or safety incidents.
- Cost Reduction in Manufacturing: The replacement of expensive and difficult-to-handle reagents with commodity chemicals significantly lowers the bill of materials. The process avoids the high energy costs associated with maintaining cryogenic temperatures (-20°C to -50°C) required by older methods, leading to substantial savings in utility expenditures. Furthermore, the simplified workup and purification procedures reduce solvent consumption and labor hours, driving down the overall cost of goods sold (COGS) for this critical intermediate.
- Enhanced Supply Chain Reliability: Utilizing widely available phosphonium salts and phosphonate esters reduces dependency on niche chemical suppliers who may have limited capacity. The robustness of the reaction conditions means that production is less susceptible to variations in raw material quality or minor fluctuations in process parameters. This reliability ensures consistent delivery schedules for downstream API manufacturers, minimizing the risk of stockouts that could disrupt the production of finished Rosuvastatin tablets.
- Scalability and Environmental Compliance: The absence of highly toxic cyanide derivatives and pyrophoric aluminum reagents simplifies the permitting process for new manufacturing lines and reduces the liability associated with hazardous waste disposal. The process generates less hazardous waste, aligning with increasingly stringent environmental regulations globally. This green chemistry profile facilitates easier scale-up from pilot plants to multi-ton commercial facilities, ensuring that supply can be rapidly expanded to meet market demand without regulatory hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer. The answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making.
Q: How does this new method improve safety compared to traditional routes?
A: The novel process eliminates the need for highly toxic cyanomethylphosphonate and dangerous cryogenic reductions using diisobutylaluminum hydride (DIBAL-H), significantly reducing operational hazards and waste treatment costs.
Q: What yields can be expected from this synthesis route?
A: Experimental data from the patent indicates consistent yields ranging from 82% to 88%, demonstrating high efficiency and robustness suitable for industrial scale-up.
Q: Is the triphenylphosphine oxide byproduct recyclable?
A: Yes, the process allows for the recovery and recycling of triphenylphosphine oxide, which further contributes to cost reduction and environmental compliance by minimizing solid waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the competitive landscape of cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101100459B are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Rosuvastatin intermediate meets the highest international standards. Our commitment to process optimization allows us to deliver high-quality materials that support your regulatory filings and commercial launch timelines effectively.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most robust and cost-efficient chemical strategy available today.
