Optimized Industrial Synthesis of Ticagrelor Intermediates for Commercial Scale-up
Optimized Industrial Synthesis of Ticagrelor Intermediates for Commercial Scale-up
The pharmaceutical landscape for antiplatelet therapy has been revolutionized by the introduction of Ticagrelor, a potent P2Y12 receptor antagonist marketed as Brilinta. As demand for this critical cardiovascular medication surges globally, the efficiency and sustainability of its supply chain have become paramount concerns for generic manufacturers and innovators alike. The patent CN102675321A discloses a groundbreaking preparation method that addresses the longstanding bottlenecks in Ticagrelor synthesis. This technical insight report analyzes the novel pathway, which leverages advanced protecting group chemistry to streamline the construction of the complex cyclopentyl-triazolo-pyrimidine core. By shifting away from hazardous oxidation protocols toward robust nucleophilic substitutions and controlled cyclizations, this methodology offers a viable path for reliable API intermediate supplier partnerships aiming to secure the global market.
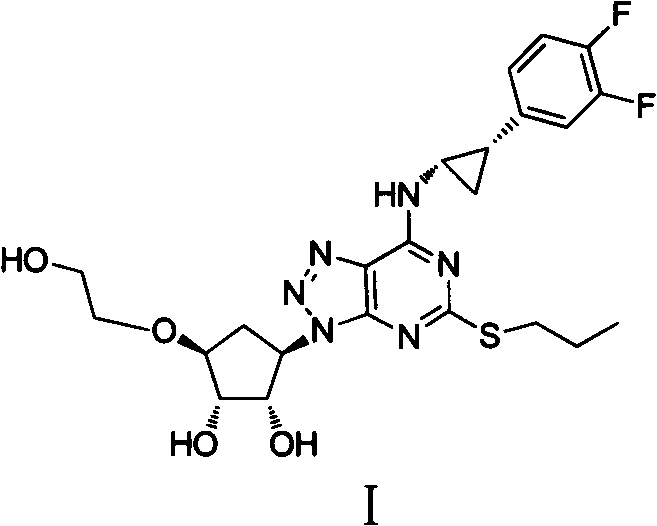
Ticagrelor, chemically defined as (1S,2S,3R,5S)-3-[7-[(1R,2S)-2-(3,4-difluorophenyl)cyclopropylamino]-5-(propylsulfanyl)-3H-[1,2,3]triazolo[4,5-d]pyrimidin-3-yl]-5-(2-hydroxyethoxy)cyclopentane-1,2-diol, presents significant synthetic challenges due to its multiple chiral centers and sensitive functional groups. The disclosed invention provides a comprehensive solution that enhances both the economic and environmental profile of production. For procurement managers and supply chain heads, understanding the nuances of this improved route is essential for evaluating potential partners who can deliver cost reduction in pharmaceutical manufacturing without compromising on the stringent quality standards required for cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as those detailed in patents WO9905143 and WO0192263, have historically plagued the industry with inefficiencies and safety hazards. The original AstraZeneca route, often referred to as Scheme 1 in technical literature, relies heavily on osmium tetroxide for dihydroxylation and DIBAL-H for reductions. These reagents are not only prohibitively expensive but also pose severe toxicity risks and generate substantial quantities of hazardous industrial waste, complicating disposal and regulatory compliance. Furthermore, the extensive step count in these legacy processes inherently accumulates yield losses at each stage, resulting in a low overall recovery rate that drives up the cost of goods sold (COGS). The reliance on iron powder reductions and other archaic techniques further exacerbates the impurity profile, necessitating complex and costly purification workflows that delay time-to-market for generic versions.
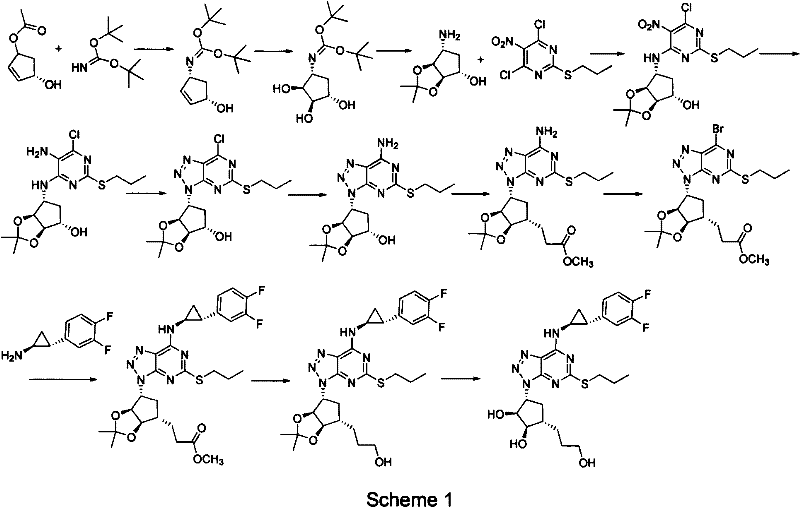
The Novel Approach
In stark contrast, the method described in CN102675321A introduces a strategic redesign of the synthetic trajectory that prioritizes atom economy and operational simplicity. By initiating the synthesis with a protected cyclopentyl amine derivative (Compound VII) and employing a nucleophilic substitution to install the hydroxyethyl side chain early in the sequence, the process effectively shields sensitive hydroxyl groups from the harsh conditions of subsequent steps. This approach eliminates the need for toxic osmium oxidations entirely. Instead, it utilizes readily available alkylating agents and standard bases like potassium tert-butoxide or sodium hydride in common solvents such as THF or toluene. The result is a significantly shortened reaction sequence that minimizes unit operations, thereby reducing solvent consumption, energy usage, and labor costs. This streamlined architecture represents a paradigm shift towards greener chemistry, offering substantial cost savings and a more robust supply chain for high-purity API intermediates.
Mechanistic Insights into Triazole Cyclization and Coupling
The cornerstone of this novel synthesis lies in the efficient construction of the triazolo-pyrimidine fused ring system, a transformation that dictates the final purity and potency of the drug substance. The process involves the reaction of the amino-pyrimidine intermediate (Compound IV) with alkali metal nitrites, such as sodium nitrite or potassium nitrite, under acidic conditions. This diazotization-cyclization cascade is meticulously controlled to ensure the formation of the [1,2,3]triazolo ring with high regioselectivity. The use of acetic acid or dilute sulfuric acid as the reaction medium facilitates the generation of the reactive nitrosonium species in situ, which attacks the primary amine to form the diazonium salt. Subsequent intramolecular cyclization closes the triazole ring, locking the core structure in place. This mechanistic pathway is superior to alternative methods as it avoids the formation of isomeric byproducts that are difficult to separate, thus simplifying downstream purification.
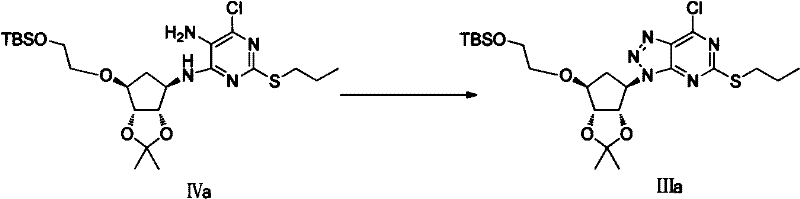
Following the ring closure, the coupling of the chiral cyclopropylamine moiety is executed with precision to maintain stereochemical integrity. The reaction between the chloro-triazole intermediate (Compound III) and (1R,2S)-2-(3,4-difluorophenyl)cyclopropylamine is conducted in alcoholic solvents like ethanol or isopropanol, often with the addition of organic bases such as triethylamine. This nucleophilic aromatic substitution displaces the chlorine atom at the 7-position of the pyrimidine ring. The choice of mild basic conditions and moderate temperatures (room temperature to reflux) ensures that the sensitive cyclopropane ring remains intact and that no racemization occurs at the chiral centers. Impurity control is further enhanced by the stability of the silyl or ether protecting groups on the side chain, which prevent unwanted alkylation or elimination reactions during this critical coupling phase, ensuring a clean reaction profile suitable for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Ticagrelor Efficiently
The execution of this synthesis requires careful attention to reaction parameters to maximize yield and minimize impurities. The process begins with the protection of the starting material, followed by a sequence of coupling, cyclization, and deprotection steps that are amenable to standard reactor setups. The versatility of the method is highlighted by its compatibility with various protecting groups, including tert-butyldimethylsilyl (TBS), triethylsilyl (TES), methoxymethyl (MOM), and tetrahydropyranyl (THP), allowing manufacturers to select the most cost-effective option for their specific infrastructure. Detailed operational guidelines regarding stoichiometry, temperature profiles, and workup procedures are critical for reproducibility. For R&D teams looking to implement this technology, the following standardized synthesis steps outline the critical path from raw materials to the final deprotected intermediate, ensuring a smooth transition from bench scale to pilot plant operations.
- Perform nucleophilic substitution on the protected cyclopentyl amine precursor using alkylating agents like 2-bromoethoxy-tert-butyldimethylsilane under basic conditions.
- Execute catalytic hydrogenation to remove Cbz protecting groups, followed by coupling with 4,6-dichloro-2-(propylthio)-5-aminopyrimidine.
- Conduct triazole ring closure using alkali metal nitrites in acidic media, followed by amine coupling and final acid-mediated deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates directly into tangible business value and risk mitigation. The elimination of hazardous reagents like osmium tetroxide removes a significant bottleneck related to specialized handling, storage, and waste disposal, which are often major cost drivers in API manufacturing. Furthermore, the shortened synthetic route reduces the number of isolation and purification steps, leading to faster batch cycle times and increased throughput capacity. This efficiency gain allows suppliers to respond more agilely to market fluctuations, ensuring reducing lead time for high-purity pharmaceutical intermediates. The robustness of the chemistry also implies a lower risk of batch failures, providing a more predictable and reliable supply of critical materials for downstream formulation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the replacement of expensive and toxic reagents with commodity chemicals. By avoiding the use of DIBAL-H and osmium catalysts, the raw material costs are drastically lowered. Additionally, the higher overall yields achieved through the protective group strategy mean that less starting material is required to produce the same amount of final product. The use of common solvents like ethanol and ethyl acetate further simplifies solvent recovery and recycling systems, contributing to substantial cost savings in utility and waste management expenses without the need for exotic infrastructure investments.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available raw materials and reagents. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents used in this route, such as sodium nitrite, triethylamine, and palladium on carbon, are globally sourced commodities. This diversification of the supply base mitigates the risk of disruptions. Moreover, the simplified process flow reduces the complexity of logistics and inventory management, allowing for leaner operations and ensuring a continuous flow of materials to meet the rigorous demands of the global pharmaceutical market.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. The absence of heavy metal oxidants significantly reduces the toxic load of the effluent, making wastewater treatment more straightforward and compliant with increasingly strict environmental regulations. The process is designed for scalability, utilizing reaction conditions that are easily transferable from laboratory glassware to large-scale stainless steel reactors. The thermal profiles are moderate, avoiding extreme cryogenic or high-pressure conditions that require specialized equipment, thus facilitating a smoother technology transfer and faster ramp-up to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ticagrelor synthesis method. These insights are derived directly from the experimental data and process descriptions within the patent, providing a clear understanding of the operational realities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios or for sourcing intermediates from external partners.
Q: How does this novel route improve upon previous Ticagrelor synthesis methods?
A: Unlike prior art methods utilizing toxic osmium tetroxide or DIBAL-H which generate significant hazardous waste, this optimized route employs safer reagents and shorter reaction sequences, significantly reducing environmental impact and processing costs.
Q: What protecting group strategies are utilized to ensure high yield?
A: The process utilizes robust protecting groups such as tert-butyldimethylsilyl (TBS), triethylsilyl (TES), or methoxymethyl (MOM) on the hydroxyethyl side chain. This strategic protection prevents side reactions during the harsh triazole cyclization and coupling steps, ensuring superior regioselectivity and overall yield.
Q: Is this synthesis method suitable for large-scale GMP production?
A: Yes, the method relies on standard industrial solvents like ethanol, THF, and toluene, and avoids cryogenic conditions or exotic catalysts. The robust nature of the intermediates and the high yields demonstrated in embodiments make it highly scalable for commercial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing pathways for life-saving medications like Ticagrelor. Our team of expert process chemists has extensively analyzed the methodology disclosed in CN102675321A and possesses the technical capability to optimize and scale this route for industrial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate or API meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in advanced organic synthesis can drive value and security for your pharmaceutical projects.
