Optimizing Baricitinib Production: A Novel Reformatsky-Based Synthetic Route for Industrial Scale-up
The pharmaceutical landscape for JAK inhibitors continues to evolve, with Baricitinib standing out as a critical therapeutic agent for rheumatoid arthritis and alopecia areata. As demand surges, the efficiency of its manufacturing process becomes a pivotal factor for global supply chains. Patent CN116375623A introduces a transformative approach to synthesizing Baricitinib and its key intermediates, specifically addressing the bottlenecks of traditional methods. This new methodology leverages a Reformatsky reaction strategy to construct the azetidine core, offering a distinct advantage over legacy Wittig-based routes. By focusing on the synthesis of Intermediate 6, this technology provides a robust foundation for producing high-purity API intermediates suitable for commercial scale-up. The structural integrity of the final molecule, as depicted below, relies heavily on the precise construction of this nitrogen-containing heterocycle.
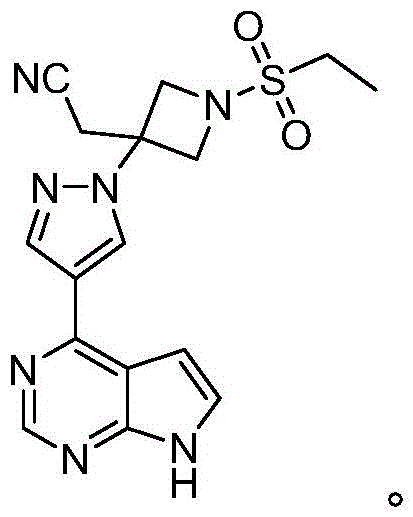
For R&D directors and process chemists, understanding the mechanistic shift from phosphorus-based olefination to zinc-mediated alkylation is crucial. The patent details a pathway that not only simplifies the reaction sequence but also drastically alters the impurity profile. This report analyzes the technical merits of this novel route, evaluating its potential to redefine cost structures and supply reliability for reliable API intermediate suppliers worldwide. The transition from complex protecting group strategies to direct functionalization represents a significant leap in process chemistry optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Baricitinib has relied on routes that introduce significant operational complexity and cost burdens. Prominent prior art, such as the Incyte route disclosed in WO 2009114512, utilizes diethyl cyanomethylphosphonate for the introduction of the acetonitrile moiety via a Wittig-type reaction. While chemically feasible, this approach suffers from severe drawbacks in an industrial setting. The use of expensive phosphonate reagents inflates raw material costs, while the generation of stoichiometric amounts of triphenylphosphine oxide creates a notorious purification challenge. Separating this solid byproduct from the desired intermediate often requires extensive chromatography or recrystallization, leading to yield losses and increased solvent consumption. Furthermore, alternative routes often employ hazardous reagents like sodium hydride for deprotonation steps, posing significant safety risks during scale-up.
Another critical bottleneck in conventional synthesis involves the protection and deprotection of the pyrrolopyrimidine nitrogen. Traditional methods frequently utilize SEM (trimethylsilylethoxymethyl) groups to mask reactive sites. This strategy necessitates additional synthetic steps for both installation and removal, extending the overall cycle time. The removal of SEM groups typically requires harsh acidic conditions or fluoride sources, which can compromise the stability of other sensitive functional groups within the molecule. Consequently, the cumulative yield of these multi-step sequences is often suboptimal, making the final API prohibitively expensive for generic manufacturers. The reliance on high-pressure hydrogenation for removing benzyl-type protecting groups further complicates the equipment requirements, limiting the number of capable contract manufacturing organizations.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN116375623A presents a streamlined alternative that circumvents these historical inefficiencies. The core innovation lies in the synthesis of Intermediate 6, achieved through a Reformatsky reaction between an ethylsulfonyl-protected azetidinone and a halogenated nitrile. This approach eliminates the need for expensive phosphonate reagents entirely, replacing them with cost-effective zinc or samarium iodide catalysts. The reaction proceeds under mild thermal conditions, typically between 0°C and 80°C, avoiding the extreme temperatures or pressures required by older methods. By bypassing the Wittig reaction, the process inherently avoids the formation of triphenylphosphine oxide, resulting in a cleaner crude reaction mixture that is far easier to purify.
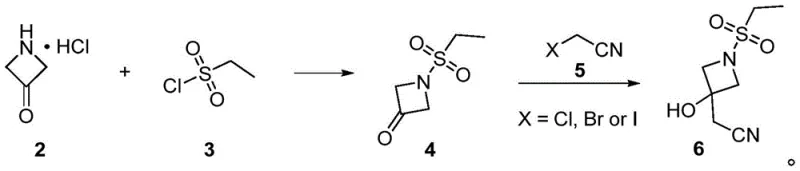
Furthermore, this novel route optimizes the coupling strategy by converting the hydroxyl group of Intermediate 6 into a sulfonate ester prior to coupling with the pyrazole boronic ester. This activation step significantly enhances the leaving group ability compared to a free hydroxyl group, thereby driving the condensation reaction to completion with higher efficiency. The subsequent Suzuki coupling with 4-chloropyrrolopyrimidine proceeds without the need for prior nitrogen protection on the pyrrole ring, shaving off multiple synthetic steps. This reduction in step count directly translates to improved throughput and reduced waste generation. For procurement managers, this means a supply chain less dependent on exotic reagents and more focused on commodity chemicals, enhancing long-term availability and price stability for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Zinc-Mediated Reformatsky Alkylation
The heart of this synthetic advancement is the Reformatsky reaction utilized to construct the quaternary center of the azetidine ring. Mechanistically, this involves the oxidative addition of activated zinc metal into the carbon-halogen bond of the haloacetonitrile, generating an organozinc species in situ. This nucleophilic zinc enolate then attacks the electrophilic carbonyl carbon of the N-ethylsulfonyl azetidin-3-one. Unlike Grignard reagents, which might be too reactive and cause side reactions with the nitrile group or the sulfonamide, the organozinc intermediate offers superior chemoselectivity. The reaction tolerance allows for the presence of the electron-withdrawing sulfonyl group without decomposition, a feat that is challenging with more aggressive organometallics. The use of solvents like tetrahydrofuran or toluene facilitates the formation of the zinc species while maintaining the solubility of the organic substrates.
Impurity control is another area where this mechanism excels. In traditional Wittig pathways, the formation of E/Z isomers and phosphine-related impurities is common. The Reformatsky pathway, however, leads directly to the saturated alkylated product without geometric isomerism issues. Additionally, the subsequent activation of the hydroxyl group in Intermediate 6 to a mesylate or tosylate (Compound 8) is a strategic move to facilitate nucleophilic substitution. The sulfonate group is an excellent leaving group, allowing the pyrazole boronic ester to displace it efficiently under basic conditions. This minimizes the risk of elimination side reactions that could occur if one attempted to couple directly with the alcohol. The final Suzuki coupling employs standard palladium catalysis, such as Pd(PPh3)4, ensuring high fidelity in forming the carbon-carbon bond between the pyrazole and the pyrrolopyrimidine cores.
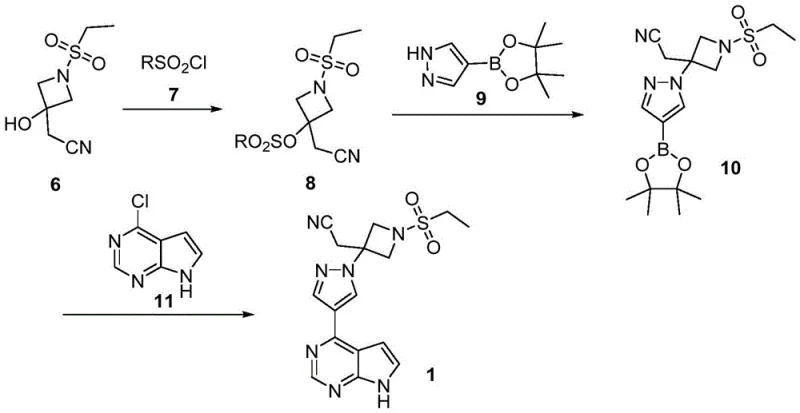
From a quality control perspective, the absence of heavy metal contaminants from phosphine oxides simplifies the purification workflow. The primary impurities are likely to be unreacted starting materials or simple hydrolysis products, which are generally easier to remove via crystallization or extraction. The patent specifies the use of bases like cesium carbonate or potassium carbonate in polar aprotic solvents like DMF for the coupling steps, conditions that are well-understood and easily controlled in a GMP environment. This mechanistic robustness ensures that the process can consistently deliver high-purity API intermediates, meeting the stringent specifications required by regulatory bodies. For technical teams, this predictability reduces the risk of batch failures and ensures a stable supply of clinical-grade material.
How to Synthesize Baricitinib Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters, particularly during the metal-mediated steps. The preparation of Intermediate 6 serves as the critical gateway, where the choice of zinc activation and temperature control dictates the success of the alkylation. Following this, the activation of the alcohol and the subsequent coupling reactions must be monitored to prevent over-sulfonylation or hydrolysis. The detailed procedural nuances, including specific molar ratios and workup protocols, are essential for reproducing the high yields reported in the patent literature. For a comprehensive guide on executing these transformations in a pilot or production plant, please refer to the standardized protocol below.
- Sulfonylation of Azetidin-3-one hydrochloride with ethylsulfonyl chloride to form compound 4.
- Reformatsky reaction of compound 4 with halogenated nitrile using Zinc or Samarium Iodide catalyst to yield Intermediate 6.
- Activation of Intermediate 6 via sulfonylation with sulfonyl chloride to create a better leaving group (Compound 8).
- Condensation of Compound 8 with pyrazole boronic ester (Compound 9) to form the coupled intermediate 10.
- Final Suzuki coupling of Compound 10 with 4-chloropyrrolopyrimidine to yield Baricitinib.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for diethyl cyanomethylphosphonate and complex protected pyrrolopyrimidine derivatives, manufacturers can source inputs from a broader base of chemical suppliers. This diversification mitigates the risk of supply disruptions caused by single-source dependencies. Moreover, the removal of hazardous reagents like sodium hydride reduces the regulatory burden associated with handling and storing dangerous goods, lowering insurance and compliance costs. The overall process safety profile is significantly enhanced, making it easier to obtain operational permits for large-scale production facilities.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the replacement of expensive reagents with commodity chemicals. The avoidance of the Wittig reaction removes the cost associated with phosphonate esters and the downstream processing required to remove phosphine oxides. Fewer synthetic steps mean reduced labor hours, lower solvent volumes, and decreased energy consumption for heating and cooling. The higher atom economy of the Reformatsky reaction ensures that a greater proportion of the input mass ends up in the final product, minimizing waste disposal fees. These factors combine to create a substantially lower cost of goods sold (COGS), providing a competitive edge in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as azetidin-3-one hydrochloride and ethylsulfonyl chloride ensures a stable supply chain. These chemicals are produced in large volumes for various industries, reducing the likelihood of shortages. The simplified process flow also shortens the manufacturing lead time, allowing for faster response to market demand fluctuations. By reducing the number of intermediate isolation steps, the risk of material loss during transfer and storage is minimized. This continuity is vital for maintaining consistent inventory levels and meeting Just-In-Time delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of high-pressure hydrogenation make this route highly scalable. It can be easily transferred from laboratory glassware to large stainless steel reactors without significant engineering modifications. The reduction in hazardous waste, particularly the elimination of phosphorus-containing byproducts, aligns with increasingly strict environmental regulations. This green chemistry approach not only reduces the environmental footprint but also simplifies wastewater treatment processes. For companies aiming to meet sustainability goals, this synthesis method offers a clear pathway to greener manufacturing practices without compromising on yield or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. They are derived from the specific advantages and procedural details outlined in the patent documentation. Understanding these aspects helps stakeholders evaluate the feasibility of adopting this technology for their own production lines. The answers reflect the consensus on how this route compares to established industry standards.
Q: Why is the Reformatsky reaction preferred over the Wittig reaction for this synthesis?
A: The Reformatsky reaction avoids the formation of triphenylphosphine oxide, a difficult-to-remove byproduct associated with Wittig reagents. This significantly simplifies purification and improves overall atom economy.
Q: What catalysts are utilized in the novel synthetic route?
A: The process utilizes metal catalysts such as Zinc powder or Samarium Iodide for the Reformatsky step, and Palladium catalysts like Pd(PPh3)4 for the final Suzuki coupling, ensuring high conversion rates under mild conditions.
Q: How does this method improve supply chain reliability for Baricitinib intermediates?
A: By using readily available starting materials like Azetidin-3-one hydrochloride and eliminating hazardous reagents like sodium hydride, the route reduces procurement risks and enhances operational safety for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baricitinib Supplier
The technological potential of this Reformatsky-based synthesis is immense, offering a clear path to more affordable and accessible Baricitinib. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such innovative patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly converted into industrial outputs. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to quality ensures that the complex chemistry involved in constructing the azetidine-pyrrolopyrimidine scaffold is managed with precision and care.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this optimized route, we can help you achieve significant reductions in manufacturing costs while securing a stable supply of high-quality intermediates. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can accelerate the availability of this vital medication to patients worldwide, combining scientific innovation with supply chain excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
