Scalable Synthesis of 5-Aminolevulinic Acid Hydrochloride via Metal-Free Nitroacetate Chemistry
Scalable Synthesis of 5-Aminolevulinic Acid Hydrochloride via Metal-Free Nitroacetate Chemistry
The global demand for 5-aminolevulinic acid hydrochloride (ALA HCl) continues to surge, driven by its critical applications in photodynamic therapy for oncology and as a biodegradable agrochemical agent. However, the industrial realization of ALA has historically been hindered by complex synthetic routes involving toxic reagents or prohibitive costs. Patent CN102827015A introduces a transformative approach that addresses these bottlenecks by utilizing a nitroacetate-based nucleophilic addition strategy. This technical insight report analyzes the proprietary methodology disclosed in the patent, highlighting its potential to redefine the supply chain dynamics for this high-value pharmaceutical intermediate. By shifting away from halogenated solvents and precious metal catalysts, this route offers a compelling value proposition for manufacturers seeking to optimize both purity profiles and production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ALA has relied on pathways that are increasingly untenable for modern green chemistry standards and cost-sensitive manufacturing environments. For instance, early methods utilized levulinic acid as a starting material, requiring bromination steps that generate significant amounts of hazardous liquid bromine waste, creating severe environmental compliance burdens. Other routes, such as those employing glycine derivatives, depend heavily on palladium-catalyzed coupling reactions, as illustrated in prior art schemes involving triphenylphosphine palladium chloride.
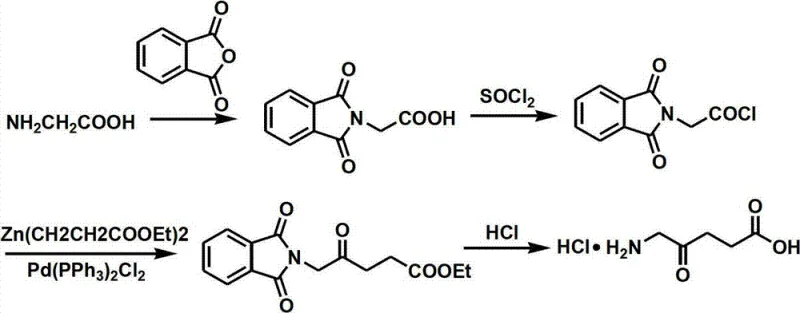
The reliance on such precious metal catalysts not only inflates the raw material costs but also introduces complex purification challenges to remove trace metal residues to meet stringent pharmaceutical specifications. Furthermore, photochemical oxidation routes and those utilizing nitromethane present their own sets of difficulties, including harsh reaction conditions, low overall yields, and the use of highly toxic reagents that complicate worker safety protocols and waste disposal logistics.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN102827015A leverages a direct nucleophilic addition between nitroacetate and succinic anhydride. This reaction constructs the carbon skeleton of ALA in a single, efficient step under strongly alkaline conditions, typically utilizing potassium tert-butoxide. The elegance of this chemistry lies in its simplicity and the avoidance of halogenated intermediates entirely.
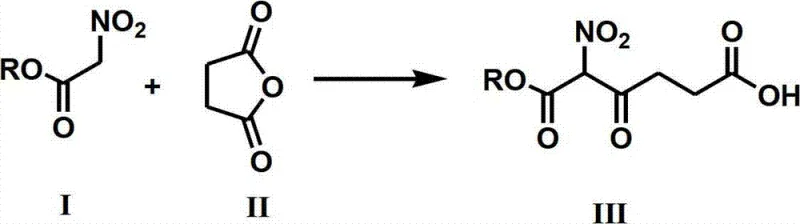
By bypassing the need for bromination or expensive transition metal catalysis, this method significantly streamlines the process flow. The subsequent steps involve a straightforward reduction of the nitro group followed by hydrolysis, both of which can be performed using commodity chemicals like zinc powder and hydrochloric acid. This structural simplification of the synthetic tree directly translates to reduced operational complexity and a lower barrier to entry for large-scale commercial production.
Mechanistic Insights into Nitroacetate-Mediated Carbon Skeleton Construction
The core mechanistic breakthrough of this patent resides in the initial carbon-carbon bond formation. Under the influence of a strong base such as potassium tert-butoxide in a polar aprotic solvent like dimethyl sulfoxide, the nitroacetate ester forms a stabilized carbanion. This nucleophile attacks the electrophilic carbonyl carbon of the succinic anhydride ring, leading to ring opening and the formation of the 5-ester-5-nitro-4-oxo-pentanoic acid intermediate. This step is highly regioselective and proceeds with high efficiency, avoiding the formation of complex byproduct mixtures often seen in radical-based halogenation chemistries.
Following the skeleton construction, the reduction of the nitro group to the primary amine is achieved using zinc powder in an acidic medium, such as acetic acid. This heterogeneous reduction is robust and tolerant of the ketone functionality present in the molecule, ensuring that the carbonyl group remains intact while the nitro group is selectively converted. The final hydrolysis step cleaves the ester moiety and simultaneously forms the hydrochloride salt, yielding the final API intermediate with high purity. The absence of heavy metal catalysts throughout this sequence ensures that the impurity profile is dominated by organic byproducts that are easily removed via standard recrystallization techniques.
How to Synthesize 5-Aminolevulinic Acid Hydrochloride Efficiently
The execution of this synthesis requires precise control over reaction temperatures and stoichiometry to maximize yield and minimize side reactions. The process begins with the careful addition of the base to the nitroacetate solution at low temperatures to prevent decomposition, followed by the controlled addition of succinic anhydride. The subsequent reduction phase demands batch-wise addition of zinc powder to manage the exotherm effectively. Detailed standard operating procedures regarding specific molar ratios, temperature ramps, and workup protocols are essential for reproducibility.
- Perform nucleophilic addition of nitroacetate and succinic anhydride under strong alkaline conditions to form the nitro-keto ester intermediate.
- Reduce the nitro group of the intermediate to an amino group using zinc powder in an acidic solvent.
- Hydrolyze the ester group and form the hydrochloride salt using aqueous hydrochloric acid followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nitroacetate-based route offers substantial strategic advantages over traditional manufacturing methods. The primary driver of value is the drastic simplification of the raw material basket. By eliminating the need for liquid bromine, palladium catalysts, and specialized photochemical reactors, the process reduces exposure to volatile commodity markets for precious metals and hazardous chemicals. This shift creates a more stable and predictable cost structure, insulating the supply chain from sudden price spikes associated with rare earth or noble metal sourcing.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived principally from the substitution of high-cost inputs with commodity chemicals. The removal of palladium and ruthenium catalysts eliminates not only the purchase cost of these metals but also the downstream costs associated with metal scavenging and recovery processes. Furthermore, the use of zinc powder as a reducing agent represents a fraction of the cost of catalytic hydrogenation setups or stoichiometric hydride reagents. The simplified workup, which relies on extraction and recrystallization rather than complex distillation columns, further reduces energy consumption and capital expenditure requirements for plant infrastructure.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically nitroacetates and succinic anhydride, are produced on a massive global scale for various industrial applications. This abundance ensures a secure supply line with multiple qualified vendors, mitigating the risk of single-source bottlenecks. Unlike specialized reagents that may have long lead times or import restrictions due to toxicity classifications, the inputs for this process are readily available in bulk quantities. This accessibility allows for just-in-time inventory strategies and reduces the need for holding large safety stocks of hazardous materials.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is significantly cleaner. The avoidance of liquid bromine and nitromethane reduces the generation of toxic waste streams, simplifying effluent treatment and lowering disposal fees. The process operates under relatively mild conditions without the need for high-pressure hydrogenation or intense UV irradiation, which lowers the operational risk profile of the facility. These factors collectively enhance the scalability of the process, allowing manufacturers to ramp up production from pilot batches to multi-ton commercial runs with minimal regulatory friction or additional environmental permitting hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on the practical implications for industrial adoption.
Q: Why is the nitroacetate route superior to traditional bromination methods for ALA synthesis?
A: Traditional bromination methods often utilize liquid bromine, which poses severe environmental pollution risks and requires specialized handling equipment. The nitroacetate route described in CN102827015A eliminates the need for hazardous halogenating agents, significantly improving operational safety and reducing waste treatment costs.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, this method is distinctively metal-free regarding catalysis. Unlike prior art routes that rely on costly palladium or ruthenium catalysts, this process utilizes inexpensive zinc powder for reduction and common organic bases, drastically lowering the raw material cost profile.
Q: Is the purification process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids complex rectification or multiple recrystallization steps for intermediates. The final product can be purified through a single recrystallization step, which simplifies the downstream processing and enhances overall throughput efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aminolevulinic Acid Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for high-value intermediates like 5-aminolevulinic acid hydrochloride. Our technical team has extensively evaluated the nitroacetate pathway described in CN102827015A and validated its potential for commercial deployment. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical and agrochemical regulators.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
