Advanced Manufacturing of 5-ALA HCl: A Technical Breakthrough for Commercial Scale-up
Introduction to Next-Generation 5-ALA HCl Manufacturing
The pharmaceutical industry's demand for high-purity photosensitizers has never been more critical, particularly for Photodynamic Therapy (PDT) applications in treating actinic keratosis and various epithelial cancers. At the forefront of this chemical evolution is the synthesis of 5-aminolevulinic acid hydrochloride (5-ALA HCl), a molecule whose production complexity has historically constrained supply chains. The technical disclosure found in patent CN102627573B represents a paradigm shift in how this valuable pharmaceutical intermediate is manufactured, moving away from precarious multi-step sequences toward a robust, commodity-chemical-based approach. By leveraging 1,3-dichloroacetone as a foundational building block, this methodology circumvents the reliance on expensive heterocyclic precursors and dangerous reagents that have plagued previous generations of synthesis. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable 5-ALA HCl supplier capable of meeting stringent regulatory and volume requirements without compromising on cost-efficiency or safety standards.
This report analyzes the technical merits of the disclosed invention, contrasting it against seven distinct prior art routes to highlight its superiority in selectivity and yield. The core innovation lies in the strategic combination of a Gabriel reaction followed by a malonate substitution, a sequence that not only simplifies the synthetic topology but also inherently controls impurity profiles through the physical properties of the intermediates. As we delve into the mechanistic details and commercial implications, it becomes evident that this process offers a viable pathway for the commercial scale-up of complex pharmaceutical intermediates, addressing the chronic bottlenecks of purification and raw material availability that often delay time-to-market for new PDT formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-aminolevulinic acid has been fraught with significant technical and economic hurdles, as evidenced by the seven representative routes analyzed in the background of the patent. Early methodologies, such as those utilizing furylmethylamine or tetrahydrofuryl methylamine, depend heavily on precious metal catalysts like palladium or ruthenium for oxidation steps, introducing substantial cost volatility and requiring complex metal scavenging protocols to meet residual metal specifications. Furthermore, routes originating from glycine or succinic anhydride often involve hazardous reagents; for instance, the use of sodium azide presents severe explosion risks during scale-up, while cuprous cyanide introduces acute toxicity concerns that complicate waste management and operator safety. Perhaps most critically, the widely cited route starting from levulinic acid suffers from inherent regioselectivity issues during the bromination step.
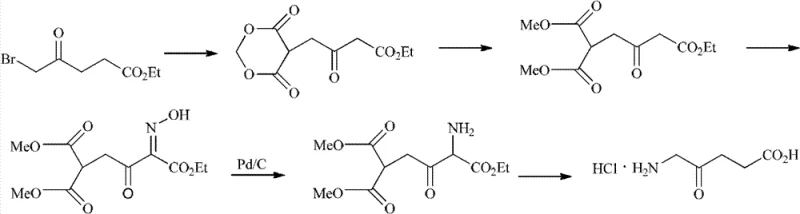
As illustrated in the prior art depicting levulinic acid bromination, the reaction inevitably generates a high proportion of the 3-bromo isomer alongside the desired 5-bromo intermediate. This side reaction cannot be suppressed merely by tuning reaction conditions, necessitating energy-intensive separation techniques such as column chromatography or high-vacuum rectification, which are economically unfeasible for industrial production. Even improved variations of this route that attempt to bypass separation suffer from abysmal overall yields, reported as low as 7%, rendering them commercially non-viable. Additionally, the reliance on specialized starting materials like ethyl 5-bromolevulinate creates supply chain fragility, as these are not readily available commodity chemicals and often require custom synthesis, further elongating lead times and increasing the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these convoluted pathways, the novel approach detailed in CN102627573B utilizes 1,3-dichloroacetone, a readily accessible and inexpensive commodity chemical, to construct the carbon backbone of 5-ALA HCl. This strategy fundamentally alters the economic equation by eliminating the need for precious metal catalysts and explosive reagents, replacing them with classical, high-yielding organic transformations. The process initiates with a Gabriel reaction to install the nitrogen functionality, followed by a nucleophilic substitution with a malonate derivative to extend the carbon chain, and concludes with a unified hydrolysis and decarboxylation step. This streamlined three-step sequence minimizes unit operations and maximizes atom economy, directly addressing the yield and purity deficiencies of legacy methods. By avoiding the formation of regioisomeric byproducts inherent in the bromination of ketones, this route ensures a cleaner reaction profile that simplifies downstream processing.
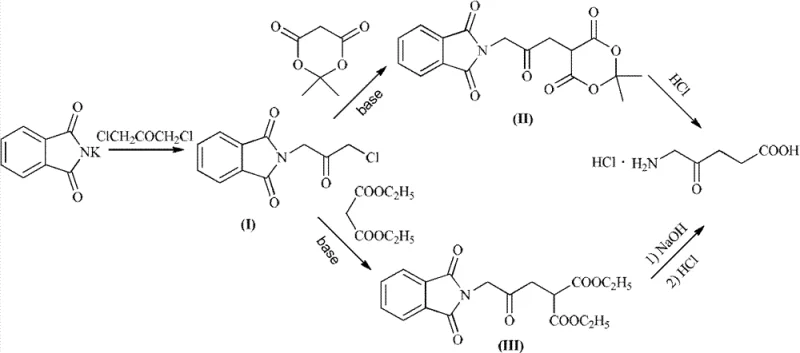
The versatility of this novel approach is further enhanced by the option to use either isopropylidene malonate or diethyl malonate, allowing manufacturers to optimize based on solvent compatibility and cost preferences. The reaction conditions are notably mild, with the initial Gabriel substitution proceeding efficiently at temperatures between 10°C and 30°C, reducing energy consumption compared to high-temperature reflux processes. Moreover, the intermediates generated in this sequence—specifically the phthalimide-protected ketones—are crystalline solids, a physical property that is invaluable for purification. Unlike oily intermediates that require difficult distillations, these solids can be isolated via simple filtration and recrystallization, ensuring that impurities are stripped away at each stage rather than carrying through to the final API. This structural elegance translates directly into operational reliability, making it an ideal candidate for reliable pharmaceutical intermediates supplier networks aiming for consistent quality.
Mechanistic Insights into Gabriel Reaction and Malonate Alkylation
The chemical elegance of this synthesis lies in the precise orchestration of nucleophilic substitutions and hydrolytic cleavages. The first stage involves the reaction of potassium phthalimide with 1,3-dichloroacetone. Mechanistically, the phthalimide anion acts as a potent nucleophile, selectively displacing the chloride at the 1-position of the dichloroacetone. A critical feature of this step, particularly when conducted in an aqueous system, is the solubility differential; the starting materials are water-miscible, but the resulting intermediate (I), 2-(3-chloro-2-oxopropyl)isoindoline-1,3-dione, is water-immiscible and precipitates out of the solution. This precipitation serves as a thermodynamic drive, pulling the equilibrium towards product formation and preventing over-reaction or degradation. The use of excess 1,3-dichloroacetone ensures complete consumption of the more expensive phthalimide, and the unreacted dichloroacetone remains in the aqueous filtrate, allowing for potential recycling and further cost optimization.
Following the isolation of intermediate (I), the second stage employs a classic alkylation of a stabilized carbanion. Under alkaline conditions, using bases such as sodium methoxide, sodium ethoxide, or triethylamine, the active methylene group of the malonate derivative (either diethyl malonate or isopropylidene malonate) is deprotonated to form a nucleophilic enolate. This enolate then attacks the remaining chloromethyl group of intermediate (I) in an SN2 fashion, successfully installing the four-carbon backbone required for 5-ALA. The choice of base and solvent is flexible, accommodating green chemistry principles by allowing the use of alcohols or polar aprotic solvents like DMF. The final transformation is a vigorous acid-catalyzed hydrolysis. Treatment with 6N hydrochloric acid at reflux simultaneously cleaves the phthalimide protecting group to release the primary amine and hydrolyzes the ester functionalities of the malonate. The resulting beta-keto acid intermediate is thermally unstable and spontaneously undergoes decarboxylation, losing carbon dioxide to yield the final 5-aminolevulinic acid structure, which is immediately captured as the stable hydrochloride salt.
How to Synthesize 5-Aminolevulinic Acid Hydrochloride Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the patented route. The process begins with the preparation of the phthalimide intermediate in a controlled aqueous or organic environment, followed by the alkylation step which demands anhydrous conditions if strong bases like sodium hydride are employed. The final hydrolysis is the most energy-intensive step, requiring prolonged reflux to ensure complete decarboxylation and removal of the phthalic acid byproduct.
- Perform a Gabriel reaction between 1,3-dichloroacetone and potassium phthalimide in an aqueous or organic phase to obtain intermediate (I).
- React intermediate (I) with isopropylidene malonate or diethyl malonate under alkaline conditions to form intermediate (II) or (III).
- Hydrolyze and decarboxylate the intermediate using hydrochloric acid or sodium hydroxide followed by acidification to yield 5-ALA HCl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthesis route offers profound strategic advantages that extend beyond mere technical feasibility. The primary driver for adoption is the drastic simplification of the raw material portfolio. By shifting from specialized, low-volume precursors like furylmethylamine or ethyl 5-bromolevulinate to bulk commodities like 1,3-dichloroacetone and diethyl malonate, manufacturers can leverage established global supply chains that offer greater price stability and availability. This reduces the risk of production stoppages due to raw material shortages, a common vulnerability in the fine chemical sector. Furthermore, the elimination of precious metal catalysts removes a significant cost center associated with both the purchase of the catalyst and the downstream validation required to prove its removal from the final product, thereby streamlining the quality assurance workflow.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the convergence of high-yielding reactions and inexpensive inputs. The Gabriel reaction and malonate alkylation are high-efficiency transformations that minimize waste generation, directly lowering the cost of goods sold. By avoiding the formation of difficult-to-separate isomers, the process eliminates the need for costly chromatographic purification or high-vacuum distillation, which are capital-intensive and slow. The ability to recycle the aqueous mother liquor from the first step further enhances material efficiency, reducing the overall solvent and reagent consumption per kilogram of product. This lean manufacturing approach ensures that the production of high-purity pharmaceutical intermediates remains economically competitive even in fluctuating market conditions.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of non-hazardous, stable intermediates. Unlike routes involving sodium azide, which require special licensing, storage, and handling protocols due to explosion risks, the reagents in this patent are standard laboratory chemicals. This simplifies logistics, reduces insurance premiums, and allows for transportation via standard freight channels without hazardous material surcharges. Additionally, the fact that all key intermediates are isolable solids means that production can be decoupled; intermediates can be manufactured at one site, stored indefinitely without degradation, and shipped to a different facility for the final hydrolysis step. This modularity provides flexibility in capacity planning and mitigates the risk of single-site disruptions affecting the entire supply of 5-ALA HCl.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally robust. The avoidance of heavy metals like ruthenium and palladium aligns with increasingly stringent regulatory guidelines regarding elemental impurities in drug substances, reducing the burden on analytical teams. The aqueous workup in the first step minimizes the volume of organic solvent waste, contributing to a lower environmental footprint. The simplicity of the unit operations—primarily stirring, filtration, and reflux—means that the process can be scaled from pilot plant to multi-ton production with minimal engineering changes. The robustness of the chemistry tolerates minor variations in reaction parameters, ensuring consistent batch-to-batch quality which is critical for maintaining regulatory approval and customer trust in a commercial setting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method is preferred for industrial applications. Understanding these details is crucial for technical teams evaluating process transfer feasibility.
Q: Why is the 1,3-dichloroacetone route superior to levulinic acid bromination?
A: Traditional bromination of levulinic acid produces significant amounts of 3-bromolevulinic acid as a byproduct, which is difficult to separate from the desired 5-bromo isomer without expensive chromatography. The patented 1,3-dichloroacetone route avoids this regioselectivity issue entirely.
Q: Does this synthesis method involve hazardous reagents like sodium azide?
A: No, unlike several prior art methods that utilize explosive sodium azide or highly toxic cuprous cyanide, this process relies on classical Gabriel chemistry and malonate alkylation, significantly improving operational safety.
Q: How does the process handle impurity control during scale-up?
A: The key intermediates (I), (II), and (III) are all solids that can be easily isolated via filtration and recrystallization. This physical state allows for effective purification at each stage, ensuring high purity of the final API intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aminolevulinic Acid Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 5-aminolevulinic acid hydrochloride that meets the rigorous demands of the global pharmaceutical market. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch conforms to the highest standards of quality and safety required for photosensitizer applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this methodology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your project benefits from both technical excellence and commercial viability.
