Advanced Fixed-Bed Continuous Synthesis of Piperazine Derivatives for Commercial Scale Production
Advanced Fixed-Bed Continuous Synthesis of Piperazine Derivatives for Commercial Scale Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient, scalable, and cost-effective routes for producing critical heterocyclic intermediates. Patent CN1634896A represents a significant technological leap in this domain by disclosing a method for the continuous synthesis of piperazine series compounds using a fixed-bed reactor system. Unlike traditional batch processes that often struggle with thermal management and inconsistent quality, this innovation leverages a specialized composite catalyst system to facilitate the hydrogenation cyclization of N-beta-hydroxyethylethylenediamine and its derivatives. The core breakthrough lies in the ability to operate under relatively moderate pressure conditions while maintaining exceptional conversion rates and selectivity, addressing long-standing pain points in the manufacturing of piperazine-based API intermediates. This technology not only enhances the purity profile of the final product but also aligns with modern green chemistry principles by improving atom economy and reducing energy intensity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of piperazine and its substituted derivatives has been plagued by severe technical and economic bottlenecks. Early methods utilizing monoethanolamine as a starting material, as reported in patents like EP163,253,04, typically suffered from abysmally low yields, often ranging between merely 15% to 25%, necessitating complex and costly separation processes to isolate the target molecule from coproducts like piperazine. Alternative routes employing diethylenetriamine, while offering improved yields around 80%, were economically constrained by the high market price of the raw materials, rendering them unviable for large-scale commodity production. Furthermore, existing technologies based on N-beta-hydroxyethylethylenediamine, such as those described in Japanese patents JP49,11,712 and JP74,125,375, although achieving high yields exceeding 90%, required extremely harsh reaction conditions with pressures soaring between 8 MPa and 26 MPa. These high-pressure batch operations impose massive capital expenditure requirements for specialized pressure vessels and create significant safety hazards, while the batch nature of the process inherently limits throughput and complicates quality consistency across different production lots.
The Novel Approach
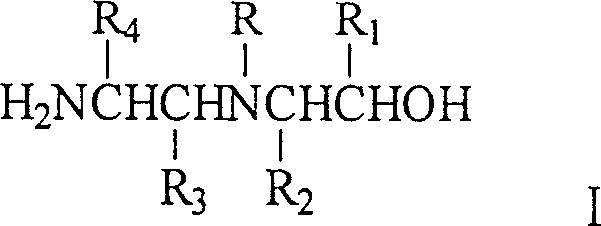
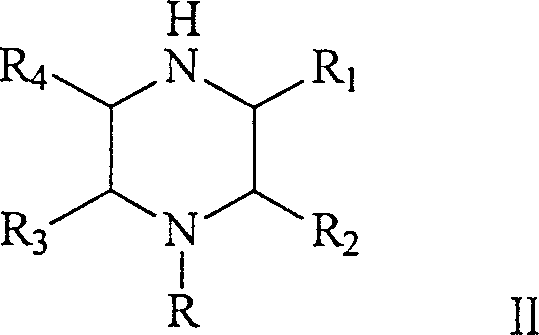
The methodology outlined in patent CN1634896A fundamentally reengineers the production landscape by transitioning from high-pressure batch reactors to a continuous fixed-bed configuration. This novel approach utilizes N-beta-hydroxyethylethylenediamine or its derivatives as the primary feedstock, processed over a sophisticated gamma-Al2O3 supported catalyst containing active components such as Nickel, Copper, Zinc, and Iron. By optimizing the catalyst composition and reactor hydrodynamics, the process achieves high conversion efficiencies and selectivity at significantly reduced pressures, typically ranging from 0 MPa to 10 MPa, with preferred embodiments operating comfortably between 0.5 MPa and 5 MPa. The continuous nature of the fixed-bed system ensures a steady-state operation that eliminates the thermal spikes and mixing inconsistencies common in batch reactors, thereby stabilizing the impurity profile. This shift not only drastically simplifies the engineering requirements for the production facility but also enables a seamless scale-up from pilot studies to multi-ton commercial manufacturing without the linear increase in risk associated with scaling batch pressure vessels.
Mechanistic Insights into Composite Catalyst Hydrogenation Cyclization
The heart of this technological advancement is the unique catalytic system designed to promote the simultaneous deamination and cyclization of the linear diamine precursor into the cyclic piperazine structure. The catalyst employs gamma-alumina as a high-surface-area carrier, which provides thermal stability and mechanical strength essential for long-term continuous operation in a fixed bed. The active phase consists of a synergistic combination of transition metals—specifically Nickel, Copper, Zinc, and Iron—deposited either as elementary substances, composite oxides, or a mixture thereof, with a total active component loading between 10% and 45%. This multi-metal formulation is critical; Nickel and Copper are renowned for their hydrogenation activity, facilitating the reduction of intermediate imines, while Chromium and Iron oxides often act as structural promoters that enhance acidity or basicity required for the dehydration steps inherent in the ring-closing mechanism. The precise balance of these components prevents the over-reduction of the ring or the formation of heavy polymeric byproducts, ensuring that the reaction pathway is directed selectively towards the desired piperazine scaffold.
From an impurity control perspective, the fixed-bed continuous flow regime offers superior management of residence time distribution compared to batch systems. In batch reactors, prolonged exposure of the product to the catalyst at high temperatures can lead to secondary reactions such as N-alkylation or ring degradation. However, in the continuous fixed-bed process described, the reactants pass through the catalyst zone with a controlled space velocity ranging from 100 h⁻¹ to 23000 h⁻¹. This precise control minimizes the contact time of the newly formed piperazine with the active sites once the cyclization is complete, effectively quenching further unwanted transformations. Additionally, the use of specific solvents or diluents like water, triethylamine, or alcohols helps in moderating the reaction exotherm and solubilizing the intermediates, further suppressing the formation of tarry residues that often foul catalysts in batch processes. This results in a cleaner crude product stream that requires less intensive downstream purification, directly impacting the final cost of goods sold.
How to Synthesize Piperazine Derivatives Efficiently
The synthesis of high-purity piperazine compounds via this patented continuous fixed-bed method involves a streamlined sequence of unit operations designed for maximum efficiency and safety. The process begins with the preparation of the feed solution, where N-beta-hydroxyethylethylenediamine is dissolved in a suitable solvent system, which may include water, benzene, or triethylamine, at a concentration that optimizes reactor throughput while maintaining heat transfer efficiency. This liquid feed is then vaporized or pumped directly into the fixed-bed reactor alongside a stream of hydrogen gas, ensuring intimate contact with the stationary catalyst bed. The reaction proceeds under controlled thermal and pressure conditions, typically between 120°C and 380°C, where the catalytic surface facilitates the intramolecular cyclization and dehydrogenation steps. For a comprehensive understanding of the specific operational parameters and safety protocols required for implementation, the detailed standardized synthesis steps are provided in the guide below.
- Prepare the catalyst by loading Ni, Cu, Zn, or Fe active components onto a gamma-Al2O3 carrier with a total active content of 10-45%.
- Feed N-beta-hydroxyethylethylenediamine or its derivatives into the fixed-bed reactor along with a solvent such as water or triethylamine.
- Maintain reaction conditions at 120-380°C and 0-10MPa pressure under a hydrogen atmosphere to achieve cyclization and deamination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1634896A translates into tangible strategic advantages regarding cost structure and supply reliability. The transition from batch to continuous processing inherently reduces the operational footprint and labor intensity associated with charging, discharging, and cleaning reactors between batches. This operational efficiency leads to a significant reduction in manufacturing overheads, as the plant can run for extended periods without interruption, maximizing asset utilization rates. Furthermore, the ability to operate at substantially lower pressures compared to legacy technologies reduces the energy consumption required for compression and lowers the maintenance costs associated with high-pressure equipment, contributing to a leaner cost base that can be passed down the supply chain.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive high-pressure infrastructure and the optimization of raw material usage. By achieving high selectivity and conversion rates, the process minimizes the loss of valuable diamine precursors to side reactions, thereby improving the overall mass balance and yield per ton of input material. The removal of the need for extreme pressure conditions (dropping from >20 MPa in older methods to <10 MPa) significantly lowers the capital depreciation costs allocated to each unit of product, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Continuous manufacturing processes are inherently more robust against supply disruptions because they maintain a steady inventory of work-in-progress rather than relying on discrete batch completions. The long service life of the gamma-Al2O3 based composite catalyst ensures that production campaigns can run for months without the need for frequent catalyst regeneration or replacement, which are common causes of unplanned downtime in batch facilities. This stability guarantees a consistent flow of finished goods to customers, mitigating the risks of stockouts and enabling just-in-time delivery models that are critical for modern pharmaceutical supply chains.
- Scalability and Environmental Compliance: Scaling a fixed-bed continuous process is generally more predictable and less risky than scaling batch reactors, as it often involves numbering up identical reactor modules rather than increasing vessel size, which introduces new heat and mass transfer challenges. From an environmental standpoint, the higher selectivity of the reaction reduces the volume of hazardous waste streams and solvent emissions, simplifying compliance with increasingly stringent environmental regulations. The process design facilitates easier containment and treatment of effluents, positioning manufacturers as responsible partners in the sustainable production of fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this continuous synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D and technical procurement teams when assessing the feasibility of adopting this route for new or existing product lines.
Q: What are the advantages of the fixed-bed continuous method over batch synthesis for piperazines?
A: The fixed-bed continuous method described in patent CN1634896A offers significant operational advantages, including easier process control, extended catalyst service life, and higher product yields compared to traditional batch processes which often suffer from lower efficiency and higher pressure requirements.
Q: What catalyst system is used in this novel synthesis route?
A: The process utilizes a robust heterogeneous catalyst system where gamma-Al2O3 serves as the carrier. The active components consist of a composite of elementary substances or oxides of Nickel (Ni), Copper (Cu), Zinc (Zn), and Iron (Fe), optimized for high selectivity in hydrogenation cyclization reactions.
Q: How does this technology impact the cost of piperazine intermediate manufacturing?
A: By enabling continuous operation at moderate pressures (0-10MPa) compared to the extreme pressures (8-26MPa) of older methods, this technology reduces energy consumption and equipment stress. Furthermore, the high selectivity minimizes waste generation and downstream purification costs, leading to substantial overall cost reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the fast-paced pharmaceutical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the fixed-bed piperazine synthesis can be seamlessly transferred from the lab to the plant. Our facilities are equipped with state-of-the-art continuous processing units and rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates. We are committed to leveraging advanced catalytic technologies to deliver high-quality piperazine derivatives that meet the exacting standards of global drug manufacturers.
We invite you to collaborate with our technical team to explore how this continuous synthesis method can optimize your specific supply chain requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and security for your piperazine sourcing strategy.
