Revolutionizing Piperazine Production: Continuous Fixed-Bed Catalysis for High-Purity Pharmaceutical Intermediates
Revolutionizing Piperazine Production: Continuous Fixed-Bed Catalysis for High-Purity Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the need for more efficient, scalable, and cost-effective synthetic routes. A pivotal development in this sector is detailed in Chinese Patent CN1413991A, which discloses a robust method for the continuous synthesis of piperazine series compounds using a fixed-bed reactor system. This technology represents a critical advancement for the production of high-value heterocyclic amines, which serve as essential building blocks for a wide array of bioactive molecules, including fluoroquinolone antibiotics and veterinary drugs. By shifting from traditional batch processing to a continuous fixed-bed approach, this methodology addresses long-standing challenges regarding catalyst stability, reaction pressure, and overall process throughput. For R&D directors and procurement strategists, understanding the nuances of this patent is vital for securing a reliable piperazine intermediate supplier capable of meeting the rigorous demands of modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of piperazine and its derivatives has been plagued by significant technical and economic inefficiencies that hinder large-scale optimization. Traditional routes, such as the ammonolysis of dichloroethane or the cyclization of monoethanolamine, often operate under extreme conditions that compromise safety and yield. For instance, early methods utilizing monoethanolamine as a feedstock typically resulted in piperazine yields as low as 15% to 25%, necessitating complex and energy-intensive separation processes to isolate the desired product from coproducts like diethylenetriamine. Furthermore, alternative pathways involving the deamination of diethylenetriamine, while offering improved yields around 80%, rely on expensive raw materials that drastically inflate the cost of goods sold (COGS). Perhaps most critically, many existing catalytic hydrogenation cyclization methods require excessively high reaction pressures, often ranging from 8 to 26 MPa, which imposes severe constraints on reactor design, increases capital expenditure for high-pressure equipment, and elevates operational risks associated with hydrogen handling in batch systems.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN1413991A introduces a streamlined continuous flow process that fundamentally redefines the production economics of piperazine compounds. By employing a fixed-bed reactor configuration, this novel approach enables precise control over residence time and thermal profiles, thereby maximizing the conversion of N-beta-hydroxyethylethylenediamine precursors into the target cyclic structures. The innovation lies not only in the reactor design but also in the sophisticated catalyst formulation, which utilizes a gamma-alumina support doped with specific transition metals such as Nickel, Copper, Chromium, Zinc, and Iron. This system operates effectively at significantly lower pressures, typically between 0 and 10 MPa, and preferably within the 0.5 to 5 MPa range, reducing the mechanical stress on equipment and enhancing overall plant safety. The transition to continuous processing eliminates the stop-start cycles inherent in batch production, leading to a dramatic improvement in production efficiency and consistency, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Fixed-Bed Hydrogenative Cyclization
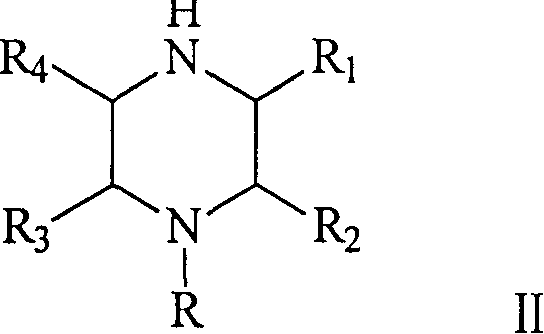
The core of this technological breakthrough resides in the intricate interplay between the supported metal catalyst and the organic substrate under hydrogenative conditions. The reaction mechanism involves the intramolecular cyclization of N-beta-hydroxyethylethylenediamine (Formula I), where the hydroxyl group is eliminated, and a new nitrogen-carbon bond is formed to close the six-membered piperazine ring (Formula II). The gamma-alumina carrier provides a high surface area and appropriate acidity to facilitate the dehydration step, while the dispersed metal species (Ni, Cu, Cr, etc.) activate molecular hydrogen for the necessary reduction steps. The specific combination of metals is crucial; for example, the inclusion of Copper and Chromium alongside Nickel helps to modulate the hydrogenation activity, preventing over-reduction or the formation of unwanted open-chain byproducts. This synergistic effect ensures that the reaction proceeds with high atom economy, converting the linear diamine precursor directly into the cyclic amine with minimal waste generation.
Furthermore, the catalyst design plays a pivotal role in impurity control, which is a primary concern for R&D teams focused on regulatory compliance. In conventional batch processes, hot spots and uneven mixing often lead to the formation of heavy ends or polymeric byproducts that are difficult to remove. The fixed-bed configuration ensures a uniform flow of reactants over the catalyst surface, maintaining a consistent temperature profile throughout the reactor bed. This homogeneity suppresses side reactions, such as the formation of triethylene diamine or other oligomers, resulting in a crude product stream with exceptionally high selectivity. Experimental data from the patent indicates that selectivity for the target piperazine compounds can exceed 90%, significantly reducing the burden on downstream purification units like distillation columns or crystallizers. This level of purity is essential for producing high-purity OLED material precursors or pharmaceutical intermediates where trace impurities can affect the efficacy and safety of the final drug product.
How to Synthesize Piperazine Derivatives Efficiently
The implementation of this continuous synthesis route requires a systematic approach to catalyst preparation and reactor operation to fully realize its potential benefits. The process begins with the meticulous preparation of the composite catalyst, ensuring that the active metal components are uniformly distributed on the gamma-alumina support to maximize active site availability. Once the reactor is loaded and conditioned, the feedstock, which may be dissolved in solvents like water, benzene, or triethylamine, is introduced into the hydrogen stream. The operational parameters, including space velocity and temperature, must be tightly regulated to maintain the catalyst in its optimal activity window. For a comprehensive understanding of the specific operational parameters and safety protocols required for this synthesis, the detailed standardized synthesis steps are provided in the guide below.
- Preparation of the composite catalyst involving Ni, Cu, Cr, Zn, or Fe on a gamma-alumina support with total active loading between 10-45%.
- Loading the catalyst into a fixed-bed tubular reactor and establishing a hydrogen atmosphere with pressures ranging from 0 to 10 MPa.
- Feeding the N-beta-hydroxyethylethylenediamine原料 (either gas or liquid phase) at temperatures between 120-380°C with a space velocity of 100-23000 h^-1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the fixed-bed continuous synthesis method described in CN1413991A offers compelling strategic advantages that extend far beyond simple chemical yield improvements. The shift from batch to continuous processing inherently stabilizes the supply chain by enabling uninterrupted production runs, thereby mitigating the risks of supply disruptions caused by batch failures or extended turnaround times between runs. This reliability is crucial for maintaining the continuity of supply for critical pharmaceutical intermediates, ensuring that downstream drug manufacturers can meet their own production schedules without delay. Moreover, the ability to operate at lower pressures and temperatures translates directly into reduced energy consumption and lower maintenance costs for high-pressure vessels, contributing to substantial cost savings over the lifecycle of the production facility.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the elimination of inefficient batch cycles and the optimization of raw material usage. By achieving high conversion rates and selectivity, the process minimizes the loss of valuable starting materials into waste streams, effectively lowering the raw material cost per kilogram of finished product. Additionally, the extended service life of the nickel-copper-chromium based catalysts reduces the frequency of catalyst replacement and regeneration, further driving down operational expenditures. The simplified downstream processing, resulting from a cleaner reaction profile, also reduces the utility costs associated with solvent recovery and product purification, creating a leaner and more cost-competitive manufacturing model.
- Enhanced Supply Chain Reliability: The continuous nature of the fixed-bed process ensures a steady and predictable output of piperazine intermediates, which is vital for just-in-time manufacturing environments. Unlike batch processes that produce material in discrete lots with potential variability, continuous flow systems provide a consistent quality profile that simplifies quality assurance and release testing. This consistency allows supply chain planners to forecast inventory levels with greater accuracy and reduce the need for large safety stocks. Furthermore, the use of readily available raw materials and robust catalyst systems reduces dependency on specialized or scarce reagents, insulating the supply chain from market volatility and ensuring long-term availability of key intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology aligns perfectly with modern green chemistry principles and regulatory expectations. The high efficiency of the reaction reduces the volume of waste generated per unit of product, simplifying waste treatment and disposal compliance. The ability to scale up by simply increasing the number of reactor tubes or the diameter of the fixed bed, rather than building massive batch vessels, offers a modular approach to capacity expansion that requires less capital investment and shorter construction timelines. This scalability ensures that production can be rapidly ramped up to meet surging market demand for piperazine-based drugs without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fixed-bed synthesis technology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on how this method compares to existing industry standards. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this process for their specific manufacturing needs.
Q: What are the primary advantages of the fixed-bed method over traditional batch synthesis for piperazines?
A: The fixed-bed continuous method described in CN1413991A offers superior operational control, significantly longer catalyst life, and higher product yields compared to traditional batch processes which often suffer from high pressure requirements and lower efficiency.
Q: Which catalyst systems are most effective for this cyclization reaction?
A: The patent specifies that composite catalysts utilizing Nickel, Copper, Chromium, Zinc, or Iron on a gamma-alumina carrier provide the optimal balance of activity and stability, with total active ingredient content maintained between 10% and 45%.
Q: How does this process impact the purity profile of the final piperazine intermediate?
A: By optimizing reaction temperature (150-320°C) and pressure (0.5-5 MPa), the process achieves high selectivity (over 90% in examples), effectively minimizing byproduct formation and simplifying downstream purification for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperazine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthetic methodologies in delivering high-quality chemical solutions to the global marketplace. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1413991A are fully realized in practical, industrial settings. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of piperazine intermediate meets the exacting standards required by the pharmaceutical and agrochemical industries. Our infrastructure is designed to support the continuous processing techniques described herein, allowing us to offer a stable and high-volume supply of these vital building blocks.
We invite you to collaborate with us to explore how this innovative fixed-bed technology can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals, ensuring a seamless partnership from pilot scale to full commercialization.
